 Dementias are diseases that destroy the brain. Much is not understood about the causes of the various dementias, but, it is known that clogged arteries cause small and large strokes and vascular dementia. Misfolded proteins occur in many types of dementia and may be part of the cause. Genetics plays some role. Now, recent research has found that inflammation can cause several of the most frequent dementias and can increase others. In fact, the new understanding of inflammation makes a direct link in the previously mysterious relation of amyloid plaques and tau neurofibrillary tangles — the two hallmarks of Alzheimer’s. disease. This post describes the new understanding of the important relationship between inflammation and dementia.
Dementias are diseases that destroy the brain. Much is not understood about the causes of the various dementias, but, it is known that clogged arteries cause small and large strokes and vascular dementia. Misfolded proteins occur in many types of dementia and may be part of the cause. Genetics plays some role. Now, recent research has found that inflammation can cause several of the most frequent dementias and can increase others. In fact, the new understanding of inflammation makes a direct link in the previously mysterious relation of amyloid plaques and tau neurofibrillary tangles — the two hallmarks of Alzheimer’s. disease. This post describes the new understanding of the important relationship between inflammation and dementia.
A previous post, Five Secrets of Brain Health, describes simple lifestyle changes that can help avoid both clogged arteries and inflammation to decrease the chances of dementia. This post describes the new research showing how important inflammation is in causing Alzheimer’s and other degenerative brain diseases.
Previous posts have demonstrated constant communication and interactions between immune cells and the brain. Immune cells are active in every aspect of ordinary brain function. So, it is not surprising that inflammation and abnormal immune activity would be significant in degenerative brain diseases like Alzheimer’s, Frontotemporal dementia, Parkinson’s, ALS, Huntington’s and others. Research now shows that immune signaling and microglia are critical to the development of these brain diseases.
Immune and Brain Systems
 There is really no way to separate the immune and brain functions. In a previous post, it was shown that T cell lymphocytes, ordinarily, travel in the CSF and are critical for cognition. They send signals to increase and decrease cognition based on levels of infection. Another post described how immune molecules are utilized as basic signals for neurons to find their place in the brain.
There is really no way to separate the immune and brain functions. In a previous post, it was shown that T cell lymphocytes, ordinarily, travel in the CSF and are critical for cognition. They send signals to increase and decrease cognition based on levels of infection. Another post described how immune molecules are utilized as basic signals for neurons to find their place in the brain.
The post, Are Microglia the Most Intelligent Brain Cell, describes the many varied functions of microglia. These immune cells are vital to all aspects of maintenance of neuronal and astrocyte networks. Microglia are critical in the development and pruning of synapses from the fetus to adulthood. Also, astrocytes are very involved in signaling with immune cells and they produce and manage every aspect of synapses.
Inflammation is the critical response by the innate and adaptive immune systems to any perceived threats, based on recognizing patterns of molecules from microbes and other abnormal molecules. This reaction includes responding to the mis-folded proteins that are part of dementia. Another factor is neuro-inflammation that was described in a post as inflammation pathways that are completely controlled by neurons. These pathways are used as one of the many forms of neuroplasticity, although to a lesser extent that a full blown inflammatory reaction.
Cleaning the Brain of Misfolded Proteins to Avoid Dementia
 Cleaning of the brain occurs in two major ways. Both help avoid dementia by getting rid of the particles that form toxic molecular clumps in or near the damaged neurons.
Cleaning of the brain occurs in two major ways. Both help avoid dementia by getting rid of the particles that form toxic molecular clumps in or near the damaged neurons.
The first is the function of microglia and other immune cells gobbling up the debris.
The second is the newly discovered brain cleaning mechanism called the “glia lymphatic” or “glymphatic system” and consists of a convective flow of the salty fluid between the brain cells from arteries and cerebral spinal fluid to veins. A very surprising discovery even more recently is that during sleep, the neurons shrink to allow the extra cellular fluid space increase by 60%. Also, brain waves from sleep seem to trigger a more rapid cleaning flow.
Microglia Are Both Critical Brain Cells and Immune Cells
 The many elaborate functions of microglia, and their many different shapes, were described in the previous post, Are Microglia the Most Intelligent Brain Cells. This post shows how microglia and other immune cells react to mis folded proteins that cause degenerative brain disease.
The many elaborate functions of microglia, and their many different shapes, were described in the previous post, Are Microglia the Most Intelligent Brain Cells. This post shows how microglia and other immune cells react to mis folded proteins that cause degenerative brain disease.
Unlike other full time immune cells, microglia are full time brain cells, as well as, full time immune cells. Microglia have many different functions, including patrolling a region of neurons and astrocytes and checking constantly on the viability of synapses. But, they can quickly become immune powerhouses.
Normally, microglia eat particles that make dementia. However, when they react to too many mis-folded proteins and nucleic acid aberrations, they do not focus on their brain surveillance duties as well. They begin to secrete powerful cytokines that can stimulate inflammation. The signaling stimulates a large structure called the inflammasome that leads to even stronger reactions. When microglia continue to be activated by mis-folded proteins, they present neurons with a large amount of inflammatory signals that can damage the neuronal circuits.
Where Has the Brain’s Immune Privilege Gone?
 Early neuroscientists thought the brain avoided all immune cells, unless the blood brain barrier was broken—a theory called immune privilege. Now, it is clear that a large number of immune cells are routinely in the brain and can identify specific microbe molecular patterns that trigger inflammation signaling. But, mis-folded proteins, also, trigger these same pattern recognition receptors. This occurs with mis-folded proteins, peptide clumps, and nucleic acids that are in the wrong place.
Early neuroscientists thought the brain avoided all immune cells, unless the blood brain barrier was broken—a theory called immune privilege. Now, it is clear that a large number of immune cells are routinely in the brain and can identify specific microbe molecular patterns that trigger inflammation signaling. But, mis-folded proteins, also, trigger these same pattern recognition receptors. This occurs with mis-folded proteins, peptide clumps, and nucleic acids that are in the wrong place.
It must be recognized that the other brain cells, including vessel lining endothelial cells, (see post on choroid plexus cells), astrocytes and neurons all can respond to abnormal patterns of microbes and toxins and send immune signals. These cells have cytokine receptors that are normally part of any inflammatory response to microbe invasion.
Most research in this area has been done for Alzheimer’s dementia, but, there is, also, now evidence of immune involvement in Parkinson’s dementia, frontotemporal dementia (FTD), amyotrophic lateral sclerosis (ALS) and Huntington’s disease.
Immune Function in the Brain
 Microglia come from a line of immune macrophages, first emerging from the embryonic yolk sac at days 8 to 10 and from then on, become the critical immune brain cells. (Please see the post, Are Microglia the Most Intelligent Brain Cell). Because they are determined so early in fetal life, they settle in the brain before the blood brain barrier is built, and live there for their entire lives. In certain infections, the immune system sends comrades to help—macrophages from the same lineage.
Microglia come from a line of immune macrophages, first emerging from the embryonic yolk sac at days 8 to 10 and from then on, become the critical immune brain cells. (Please see the post, Are Microglia the Most Intelligent Brain Cell). Because they are determined so early in fetal life, they settle in the brain before the blood brain barrier is built, and live there for their entire lives. In certain infections, the immune system sends comrades to help—macrophages from the same lineage.
As the previous post describes, microglia take on many different shapes with different functions. They have a form with many arms that are constantly moving and tapping structures such as synapses, astrocyte networks, neuronal networks, and blood vessels. This is critical for building, maintaining and pruning synapses and for neuronal plasticity.
Microglia have many different receptors, as well as transport channels, for neurotransmitters, hormones, modulators, cytokines and recognition of abnormal patterns. They produce many factors that stimulate synapses and new brain cells, including the critical BDNF. When they find abnormal synapses, they eat them.
Immune Triggers in Brain
 Specific patterns trigger large immune responses from microglia. During evolution, we developed a large number of different pattern recognition receptors for all kinds of microbes. It is possible that the microglia confuse the patterns from abnormal proteins and clumps with patterns from microbes because they are very similar. The patterns that resemble amyloid pieces on their surface are called curli fibers. Response to these microbe patterns could be the reason that our immune system is so reactive to amyloid-?.
Specific patterns trigger large immune responses from microglia. During evolution, we developed a large number of different pattern recognition receptors for all kinds of microbes. It is possible that the microglia confuse the patterns from abnormal proteins and clumps with patterns from microbes because they are very similar. The patterns that resemble amyloid pieces on their surface are called curli fibers. Response to these microbe patterns could be the reason that our immune system is so reactive to amyloid-?.
When a receptor is triggered, the microglia turns to protect the brain by fighting microbes that are not there. Recently, it has been found that multiple triggers usually activate the abnormal behavior in the microglia, not just one. These triggers include:
- Amyloid-? aggregates – in Alzheimer’s
- ?-synuclein, – in Parkinson’s
- Mutant huntingtin (HTT) – in Huntington’s
- Mutant super- oxide dismutase 1 (SOD1) – in amyotrophic lateral sclerosis
- S100A9–S100A8 complex (also known as MRP14–MRP8) – in inflammation,
- cancer and psoriasis
- Chromogranin A– in cancers and diabetes
Most neurodegenerative diseases produce an increased amount of various powerful interleukin (IL) cytokines, secreted by microglia, triggered by these patterns.
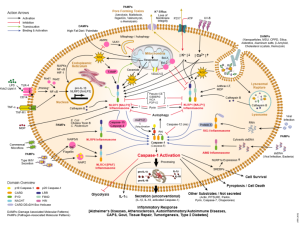
When an infection is triggered, one of the early events is the creation of an inflammasome, a large structure made of multiple proteins—receptor proteins, adaptor proteins and the enzyme caspase. Interleukin is a family of powerful cytokines that are triggered in this process. The cytokine that is first produced is not active. Caspase in the inflammasome cuts the interleukin and activates it. The inflammasome has the ability to sense many of these abnormal proteins.
Microglia, also, produce substances that can attack microbes called, reactive oxygen (ROS). This is a byproduct of metabolism that if not controlled will kill cells. Anti oxidants detoxify ROS. Microglia, also, produce, nitric oxide (NO) and other substances that can kill microbes. But, while trying to kill microbes, these various secretions from the microglia slow the creation of new brain cells and decrease the movement of materials along the neuron’s axons.
 Activated microglia, responding to an immune pattern receptor, change their shape by pulling in their arms. Unfortunately, these arms are the critical way that microglia explore the brain territory that each is responsible for. By not having arms, they are not able to touch synapses and find those that need to be pruned, as well as many other functions for neurons and astrocytes.
Activated microglia, responding to an immune pattern receptor, change their shape by pulling in their arms. Unfortunately, these arms are the critical way that microglia explore the brain territory that each is responsible for. By not having arms, they are not able to touch synapses and find those that need to be pruned, as well as many other functions for neurons and astrocytes.
The secreted NO and cytokines, also, decrease neuroplasticity—a major feature of degenerative brain disease. The brain inflammation that is triggered by the microglia stops neurotrophic factors that hurt both neurons and astrocytes. Astrocytes were shown in previous posts to be critical in all aspects of creating and maintaining synapses, as well as having their own large important network of communication that we are just beginning to understand.
The inflammation, also, damages the microglia, which are triggering it. As the microglia secretes more cytokines, both these cytokines and the microbe’s lipopolysaccharide (LPS), make the microglia less competent—less able to eat the mis-folded and clumped proteins, which started the entire problem to begin with.
Alzheimer’s
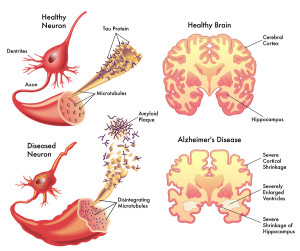 There has been no way to connect the mechanisms of the two basic simultaneous events occurring in Alzheimer’s—the destruction wrought by amyloid plaques and by tau based fibrillary tangles. Recent research, now, connects them through complex immune pathways that cause inflammation; these pathways exist in Alzheimer’s and other brain diseases.
There has been no way to connect the mechanisms of the two basic simultaneous events occurring in Alzheimer’s—the destruction wrought by amyloid plaques and by tau based fibrillary tangles. Recent research, now, connects them through complex immune pathways that cause inflammation; these pathways exist in Alzheimer’s and other brain diseases.
In Alzheimer’s, these two processes destroy neurons in different ways.
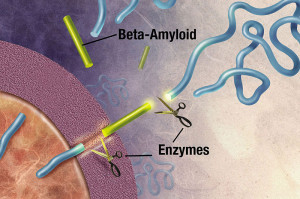 The first is the formation of larger and larger clumps of amyloid-?. Too much amyloid-? can occur by mutations that affect the enzymes that produce it. The enzyme gamma secretase cuts the amyloid precursor protein (APP). The APP is a large protein that sits in the membrane of the neuron near the synapse. It is not clear what it does, but it appears to be important for the synapse. The cut of APP can occur in several places producing amyloid-? particles in various sizes –mainly 40 or 42 amino acids long. The 42-sized amyloid-? is the size that seems to form clumps. Specific mutations of this large protein (the mutations that cause hereditary Alzheimer’s) produce more 42 and therefore, more clumps. These form the very destructive plaques.
The first is the formation of larger and larger clumps of amyloid-?. Too much amyloid-? can occur by mutations that affect the enzymes that produce it. The enzyme gamma secretase cuts the amyloid precursor protein (APP). The APP is a large protein that sits in the membrane of the neuron near the synapse. It is not clear what it does, but it appears to be important for the synapse. The cut of APP can occur in several places producing amyloid-? particles in various sizes –mainly 40 or 42 amino acids long. The 42-sized amyloid-? is the size that seems to form clumps. Specific mutations of this large protein (the mutations that cause hereditary Alzheimer’s) produce more 42 and therefore, more clumps. These form the very destructive plaques.
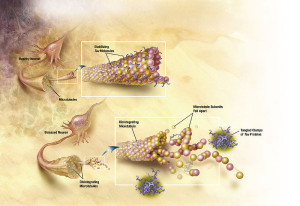 The second mechanism is the breakup of microtubules in neurons that ordinarily form scaffolding for structures and are tracks along which transport occurs. This breakup of the microtubules occurs because a protein tau that usually holds the microtubule structure together changes its shape. This alteration occurs because of a metabolic modification that abnormally attaches phosphates, excessively, to an amino acid in tau. Because of this, the tau is unable to bind with the microtubule and they disintegrate. The abnormal tau forms clumps in form of a fibril, which are insoluble, and these fibrils with the broken microtubules form the neuro fibrillary tangle, which eventually destroys the neuron.
The second mechanism is the breakup of microtubules in neurons that ordinarily form scaffolding for structures and are tracks along which transport occurs. This breakup of the microtubules occurs because a protein tau that usually holds the microtubule structure together changes its shape. This alteration occurs because of a metabolic modification that abnormally attaches phosphates, excessively, to an amino acid in tau. Because of this, the tau is unable to bind with the microtubule and they disintegrate. The abnormal tau forms clumps in form of a fibril, which are insoluble, and these fibrils with the broken microtubules form the neuro fibrillary tangle, which eventually destroys the neuron.
Now, the third way that destruction of neurons occurs appears to be inflammation.
Inflammation in Alzheimer’s
 There is a lot of evidence now for the importance of inflammation in either starting or furthering Alzheimer’s. Blood infections, obesity and lack of physical activity all appear to increase Alzheimer’s and all are involved in producing inflammation. People who have severe blood infections have increases in the dangerous cytokine tumor necrosis factor (TNF). Sepsis leads to damage to the hippocampus and cognitive problems. Severe periodontal disease is, also, correlated with more Alzheimer’s. Considering the aging population is more prone to developing Alzheimer’s, those that are wanting to combat the destruction of brain neurons through aging will want to take care of their health in other areas discussed, the big issue is having poor oral health, poor oral health is linked to other ailments and illnesses as well as Alzheimer’s, meaning dental hygiene is of utmost importance to senior citizens, many seniors exhibit poor oral health due to the lack of funding for dental health care, for this reason, dental plans are in place for seniors, however these dental plans are often only available in specific states. Websites like Let’s Say Thanks offer comparisons so it’s easier for you to pick the best one out of the countless options available on the market, aiming to improve the elderly populations’ dental hygiene.
There is a lot of evidence now for the importance of inflammation in either starting or furthering Alzheimer’s. Blood infections, obesity and lack of physical activity all appear to increase Alzheimer’s and all are involved in producing inflammation. People who have severe blood infections have increases in the dangerous cytokine tumor necrosis factor (TNF). Sepsis leads to damage to the hippocampus and cognitive problems. Severe periodontal disease is, also, correlated with more Alzheimer’s. Considering the aging population is more prone to developing Alzheimer’s, those that are wanting to combat the destruction of brain neurons through aging will want to take care of their health in other areas discussed, the big issue is having poor oral health, poor oral health is linked to other ailments and illnesses as well as Alzheimer’s, meaning dental hygiene is of utmost importance to senior citizens, many seniors exhibit poor oral health due to the lack of funding for dental health care, for this reason, dental plans are in place for seniors, however these dental plans are often only available in specific states. Websites like Let’s Say Thanks offer comparisons so it’s easier for you to pick the best one out of the countless options available on the market, aiming to improve the elderly populations’ dental hygiene.
Alzheimer patients have increased inflammation markers. It was thought at first that when tissue is destroyed in the brain, it causes inflammation. But, now there is reason to think that inflammation is part of the reason for the early destruction.
Gene networks are found in Alzheimer’s related to immune and microglia activity. Especially important genes appear to be related to the microglial’s ability to eat the debris that causes Alzheimer’s. Abnormal genes in Alzheimer’s are, also, related to complement and other immune receptors. One particular immune receptor was noted to stop microglial cells from eating amyloid-? and Alzheimer’s microglia have more of this receptor. There are others that increase amyloid-? in the membrane causing more reactivity.
There are other data showing inflammation’s effect in Alzheimer’s:
- Studies show that in experimental animals where inflammosomes cannot form then there is no buildup of amyloid plaques.
 Amyloid-? suppresses plasticity of the synapse, and the microglia’s ability to clear the amyloid.
Amyloid-? suppresses plasticity of the synapse, and the microglia’s ability to clear the amyloid.- Disrupting some of the cytokines improves amyloid-? removal and stimulates the microglia to switch back to their shape that fights inflammation, rather than the shape that promotes it.
- IL-1?, TNF and other cytokines that cause inflammation affect Alzheimer progression.
- Cytokine receptors on astrocytes and stimulation of them increases astrocytes uptake of amyloid-?.
- Microglia need APOE apolipoprotein E to eat amyloid-?.
Other factors produced by microglia, such as reactive oxygen species (ROS) and nitrogen, are critical to forming amyloid-? clumps. The nitrogen attaches to amyloid-? abnormally and this increases clumping. This reaction occurs in the center of the amyloid plaques making it even worse. Amyloid-? with nitrogen attached decreases synaptic plasticity even more.
Other immune cells in the microglial myeloid line are, also, attracted to the Alzheimer brain and contribute to the inflammation.
The New Relation of Tau and Amyloid-? Through Inflammation
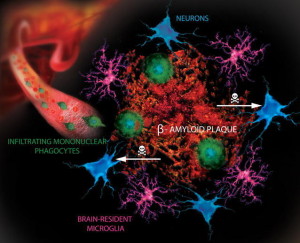 In Alzheimer’s disease, amyloid-? plaques appear to grow for many years before the tau tangles. This amyloid-? buildup is related to inflammation.
In Alzheimer’s disease, amyloid-? plaques appear to grow for many years before the tau tangles. This amyloid-? buildup is related to inflammation.
Later neurofibrillary tangles start developing. Now, research shows that microglia induced inflammation appears, also, to be critical in the development of these tangles. The same immune responses from the pattern recognition, also, stimulate abnormal tau. Microglia have cytokines that decrease the tau tangles and those that will increase them. It appears that the amyloid-? stimulated microglia then involve tau, which further destroys neurons.
Inflammation through microblia appears to be the missing link between the two different critical Alzheimer processes.
Frontotemporal Dementia
Frontotemporal dementia (FTD) is the second most common type of dementia after Alzheimer’s. This form of dementia kills neurons in the frontal and temporal regions of the brain. These neurons, also, have protein clumps inside. Inflammation is, also, an important part of this disease formation with increased levels of TNF and other cytokines. Microglia in these regions are activated into the inflammation shape. Some of the same receptors appear in both FTD and Alzheimer’s. A particular protein progranulin is critical for the formation of FTD. In FED, there is decreased progranulin, which alters the microglia function and increases inflammation.
Parkinson’s Disease and Lewey Body Dementia
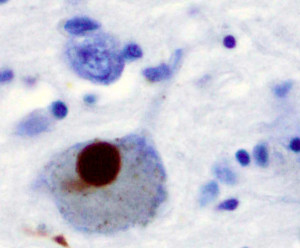
In Parkinson’s disease, specific dopamine producing neurons, related to movement, die. This process, also, involves activated microglia with many different cytokines. As in Alzheimer’s, the astrocytes are involved.
The protein clumps in Parkinson’s neurons are made of a different protein— ?-synuclein (the clumps are known as Lewey bodies). ?-synuclein is a molecule in the pre synaptic neuron that is critical for forming the sacs (vesicles) that carry neurotransmitters. As in Alzheimer’s, there are a variety of genes related to inflammation that have mutations in Parkinson’s. But, unlike Alzheimer’s these genetic alterations are rare (5%). Neuro inflammation appears to be critical.
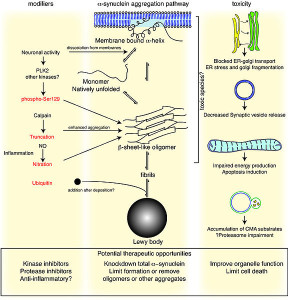
?-synuclein can be secreted from neuronal cells in sacs. It is released by dying neurons.
?-synuclein activates microglia and eventually causes a toxicity that kills neurons. A similar process occurs with the reactive oxygen and nitrogen species secreted by microglia to kill microbes. They affect gene networks and alter proteins. In this process, ?-synuclein is given an attached nitrogen (just like amyloid-?), which increases clumping and the formation of Lewey bodies, killing cells. Microglia increase this fixation of nitrogen on the ?-synuclein with the result being neuronal death.
But, just like amyloid, once ?-synuclein clumps are formed, they induce strong cytokines triggering inflammation. While the cause of the clumps is not clearly from inflammation, as it appears to be in Alzheimer’s, once they form, a massive inflammation effect increases the disease.
Amyotrophic Lateral Sclerosis (ALS or Lou Gehrig’s Disease)
This disease kills upper and lower motor neurons causing paralysis. There is now plenty of evidence that inflammation contributes to the disease, along with other toxicities that kill the neurons. Again, microglia are activated in these regions. A recent study showed that cytokines from microglia are necessary to kill the neurons.
The clumps in this case are from mutant super- oxide dismutase 1 (SOD1)., which comes from microglia and astrocytes. These clumps, while secreted from microglia, also, activate microglia and stimulate neuronal death. Cytokines and ROS and nitrogen (as in the other cases) are involved in the neuronal death. Microglia that produce SOD1 were impaired and couldn’t function as regular microglia. They could not clean up debris. Studies now show that general infections increase the ALS process.
Huntington’s Disease
This is a genetic disease with an abnormal gene that makes an abnormal protein huntingtin. The abnormal mutated protein is less stable and kills neurons through a mechanism that is not clear. It kills medium spiny neurons of the striatum, a center for movement and habitual learning.
As with the other neurodegenerative diseases, Huntington’s, also, has abnormal and increased amounts of microglia and astrocytes. The microglia produce a variety of inflammatory cytokines. Very early in the disease, microglia are activated to cause inflammation. These microglia have an abnormal accumulation of iron. In fact, Huntington’s has an abnormal amount of iron and the microglia are trying to clear it.
Inflammation and Dementia
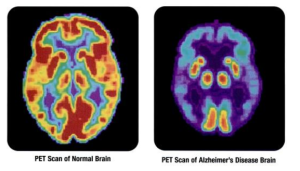 Very complex pathways of inflammation, involving microglia, appear to be critical players in the creation of brain damage in a variety of the most common degenerative brain diseases.
Very complex pathways of inflammation, involving microglia, appear to be critical players in the creation of brain damage in a variety of the most common degenerative brain diseases.
Knowing that debris is cleaned and inflammation is decreased through the Five Secrets of Brain Health, these simple lifestyle changes are highly recommended.
