 It has been thought that the brain doesn’t have typical immune responses—it has been called “immune privileged.” With the discovery of the dual role of microglia as supportive glia brain cells and resident immune cells, this view changed somewhat. Then it was discovered that T cells populate the CSF and signal to the neurons, microglia, and other brain cells. Recently, lymph drainage from the brain has been discovered in several places.
It has been thought that the brain doesn’t have typical immune responses—it has been called “immune privileged.” With the discovery of the dual role of microglia as supportive glia brain cells and resident immune cells, this view changed somewhat. Then it was discovered that T cells populate the CSF and signal to the neurons, microglia, and other brain cells. Recently, lymph drainage from the brain has been discovered in several places.
Now, the picture is much more complex. It appears that the many different compartments of the brain that are protected by various strong barriers for cell entry each have different complex immune function—blood brain barrier, blood-CSF barrier, tight junctures between border cells, strong basement lamina, pericyte layers, astrocyte end feed layers. All of these cells are involved in elaborate conversations with each other, immune cells, and microbes. Please see previous posts on T cells in the CSF and guardians of the blood brain barrier. This post summarizes the current understandings and questions about the many different compartments and how they interact for immune function—dura, subarachnoid, pia, choroid cells, CSF, brain tissue, fluid between brain cells called interstitial fluid or ISF or IS, blood vessels, and lymphatics.
New and exciting ways to image individual cells has allowed unprecedented observation of small numbers of immune cells that have very important activity in the brain. Recently, lymph vessels were discovered in the dura mater, which surrounds the brain. The ways that the immune system operates in the brain is based on the unusual complex anatomy of these many compartments and barriers to brain entry. Barriers consist of multiple different types of cells, endothelial, epithelial, and glia. The lymphatic system is unique and drains to the lymph nodes of the neck and other places
Expanding Information on Immunity in the Brain
 The term immune privilege originally referred to an organ where a transplant or graft did not produce an immune reaction. This is generally true of the brain. The usual reactions to microbes don’t occur, such as innate immune responses. The typical response to microbe molecular patterns and damaged cell patterns picked by special receptors don’t occur in the same way, where there is a rapid entry of immune and blood cells, and myelomonocytic cells. Not understanding the subtleties of the brain, it was not possible to understand how it worked until recently.
The term immune privilege originally referred to an organ where a transplant or graft did not produce an immune reaction. This is generally true of the brain. The usual reactions to microbes don’t occur, such as innate immune responses. The typical response to microbe molecular patterns and damaged cell patterns picked by special receptors don’t occur in the same way, where there is a rapid entry of immune and blood cells, and myelomonocytic cells. Not understanding the subtleties of the brain, it was not possible to understand how it worked until recently.
Previous posts show that T cells provide many functions in the brain both under normal circumstances and during infections. It was recently learned that grafts into the ventricles are rejected, and newly described drainage into the neck lymph nodes was discovered. It is odd to note that the lymph tissues in the dura draining to the subarachnoid CSF were described 200 years ago, and then forgotten.
There is now a new fluid cleaning system called the glymphatic system whereby fluid flows between the cerebrospinal fluid CSF and the interstitial fluid ISF between neurons. This takes misfolded molecules and debris from the brain tissue into the CSF.
Only the unique anatomy helps us understand how immune and neuro function together. The CSF has an input and output to the lymph channels. The brain tissue does not. It does not allow microglia, the resident immune cells, to leave and go to lymph nodes to alert T cells. The only drainage is from very small antigen molecules. The other problem with research is that mice brains are not the same as humans.
Some Barriers Allow Cells – Others Don’t
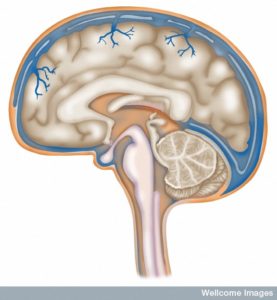 There are three layers of protection with three meningeal layers surrounding the brain tissue from CSF fluid. The one on the outside is dura mater (1mm thick with very thick matrix of fibers. The blood vessels and lymphatics that run through the dura do not connect with the brain tissue. These capillaries do have a blood brain barrier and continue to have holes for material to flow through (fenestration).
There are three layers of protection with three meningeal layers surrounding the brain tissue from CSF fluid. The one on the outside is dura mater (1mm thick with very thick matrix of fibers. The blood vessels and lymphatics that run through the dura do not connect with the brain tissue. These capillaries do have a blood brain barrier and continue to have holes for material to flow through (fenestration).
The next is arachnoid is 200 mm thick and it doesn’t allow fluids to pass through. It has tight junctions that form a barrier to the dura. This forms a version of the blood brain barrier with pumps. The sub arachnoid space (SAS) allows fluid from the CSF. The SAS is a barrier between the blood and the CSF.
Pia mater is one cell thick all over the brain tissue and allows molecules and immune cells through, but not red blood cells. It covers all blood vessels in the SAS and separates SAS from tissue and perivascular regions. The brain tissue is also coated under the pia with a firm barrier made of foot processes from astrocytes and a basement membrane. (glia limitans). Astrocytes allow small molecules through but T cells need special permission and signals.
Arteries go from the SAS through pia, sub pia, and go along the surface of brain tissue, but they are surrounded by a layer of pia. The three layers of the cells lining the blood vessel, the pia, and the glia barrier all combine into one strong barrier with no spaces. There are spaces around the blood vessels only in the basal ganglia and in the regions where axons run (white matter). Veins have less protection without pia, but a layer of cells cover them in the SAS.
Blood Brain Barrier
 BBB is between blood and brain tissue. Special capillary cells have strong tight junctions and do not allow as much material to go through with a variant of the vesicle process (pinocytosis). For T cells to enter they go through venules after the capillaries and then the astrocytes. They need signals from both to enter.
BBB is between blood and brain tissue. Special capillary cells have strong tight junctions and do not allow as much material to go through with a variant of the vesicle process (pinocytosis). For T cells to enter they go through venules after the capillaries and then the astrocytes. They need signals from both to enter.
Another entry point from blood to brain tissue that is different is in the choroid plexus (see post). Choroid cells are a barrier between blood and CSF (BCSFB – blood CSF barrier) and decide what material and cells enter into the CSF. From veins in the SAS immune cells enter the CSF.
The CSF fluid in the ventricles and the SAS are not as immune privileged as the tissues.
Afferent Paths to Lymph
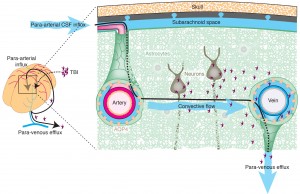 Lymphatics in these brain compartments are not the same as in other organs for antigens and cells as they travel to lymph nodes from the organs. In the brain there are two fluids that eventually send antigens to lymph nodes for evaluation and response. These are the fluid between neurons and glia, the interstitial fluid (IF) and the CSF. Both drain with different routes to lymph nodes in the neck and spine.
Lymphatics in these brain compartments are not the same as in other organs for antigens and cells as they travel to lymph nodes from the organs. In the brain there are two fluids that eventually send antigens to lymph nodes for evaluation and response. These are the fluid between neurons and glia, the interstitial fluid (IF) and the CSF. Both drain with different routes to lymph nodes in the neck and spine.
CSF has lymph vessels through dura and cribiform plate bone in the nose for both cells and molecules. ISF fluid and molecules from brain tissue go along a small space next to a basement membrane in capillaries and arteries to lymph vessels and nodes in the neck and spine. A small amount (15%) of ISF goes to and mixes with CSF. This ISF route does not allow cells. The flow is in the space next to the basement membranes of capillaries and then muscle cells. It goes along arteries in SAS through skull base to neck lymph nodes. This is a rapid route. The size of molecules are limited in this pathway. Larger complexes can block the route. This is also impaired when blood vessels stiffen. Amyloid clumps accumulate along the basement membranes.
Another route is from the white matter near the ventricles to CSF when there are problems. This brings some antigens to immune cells.
CSF is made by choroid cells and travels in ventricles to SAS of cranial and spinal nerves. From the SAS fluid from CSF goes to venous sinuses in arachnoid villi (large in humans, small in mice). Also, it drains through special lymph vessels in the nose and dura. In the nose CSF goes through crifiform plate (ethmoid bone) to nose membranes then to neck lymph nodes (deep cervical). This route is good for T cells and other immune cells. There is another route through lymph vessels in dura and skull base. Having these cells go to lymph allows for rapid response of immune problems in the CSF as opposed to brain tissue.
CSF and IS
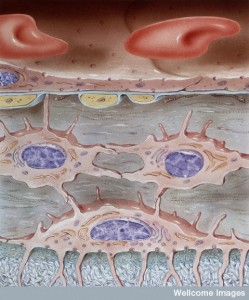 Even though they have very different connections, the fluid in the tissue and ventricles interact at the glia barrier on the brain surface. This process involves a particular channel in astrocyte end feet called aquaporin 4. A flow occurs from CSF into the brain tissue and then to lymph nodes. This has been called the glymphatic system (glia lymphatic). The fluid that drains is a mixture of CSF and IS. This flow can go back into the CSF of SAS or in the spaces along the blood vessels (veins). This system has been found to be involved in clearing debris and is increased during sleep when neurons shrink in half to allow greater cleaning flow.
Even though they have very different connections, the fluid in the tissue and ventricles interact at the glia barrier on the brain surface. This process involves a particular channel in astrocyte end feet called aquaporin 4. A flow occurs from CSF into the brain tissue and then to lymph nodes. This has been called the glymphatic system (glia lymphatic). The fluid that drains is a mixture of CSF and IS. This flow can go back into the CSF of SAS or in the spaces along the blood vessels (veins). This system has been found to be involved in clearing debris and is increased during sleep when neurons shrink in half to allow greater cleaning flow.
But, this whole concept is very complex, not fully understood, and there is some contradictory research coming out because of the immune privilege of the brain tissues.
Immune Response in the Brain
 The unique microglia immune cells and special types of dendritic cells in the brain make the immune response different from any other organ. There are macrophages in SAS from the embryo period, just like the transit of microglia at that time. Neither of these go the lymph nodes as in all other organs.
The unique microglia immune cells and special types of dendritic cells in the brain make the immune response different from any other organ. There are macrophages in SAS from the embryo period, just like the transit of microglia at that time. Neither of these go the lymph nodes as in all other organs.
Each environment and tissue has different ways that dendritic cells and T cells relate and travel. Very specific unique sets of signals in different tissues allow for special travel requirements. Signals include cytokines, vitamins (D and A) and molecules that allow the cells to stick to vessels and travel in different directions. The unique patterns in the brain are not yet understood.
But, somehow T cells in the neck lymph nodes are involved in regulating inflammation in brain tissues. The T cells that drain from the brain have very specific travel programs. There is recent research suggesting that T cells that are allowed into the brain, first must be first permitted through a conversation in the gut or lung involving signals from microbes as well. The exact way that T cells have permission to enter the brain is not clear yet.
Entering the Brain
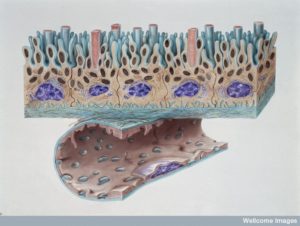 There are many immune cells in CSF but much less than other bodily fluids and the types are unique. There are more cells that are usually called adaptive (effector or memory cells) rather than innate. There are more central memory T cells. Without infection most are T (CD4+ and CD8+) cells.
There are many immune cells in CSF but much less than other bodily fluids and the types are unique. There are more cells that are usually called adaptive (effector or memory cells) rather than innate. There are more central memory T cells. Without infection most are T (CD4+ and CD8+) cells.
Cells related to inflammation use separate pathways. They travel in venules of the pia to sub pia fluid and sub arachnoid veins to SAS. They can go perivascular in the spinal or begin arachnoid and pia from choroid.
When T cells (CD4+) are activated they are given permission to cross the BBB. Those T cells that cross into CSF but are not activated with a dendritic cell (a cell that presents particles and helps activate the T cell – called an APC cell) they aren’t permitted to pass through the glia barrier. They either travel out of the CNS through CSF or they kill themselves. Meeting their antigen producing cell with the particular antigen is enough to activate them in any of the spaces.
Cytokines change the ability to move through the BBB. They also signal for other types of cells to come to the BBB. Signals include cytokines TNF stimulating special enzymes that allow passage through the astrocyte barrier (glia limitans) and into brain tissue. This can produce encephalitis or inflammation of brain tissue with devastating effects.
Choroid cells produce special signals and molecules to allow entry of immune cells from blood into CSF. It is not yet known how choroid cells allow cell into brain tissue through signaling but they appear to do this as well.
Mouse Versus Human Research
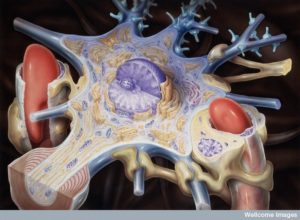 The study of these small spaces is extremely difficult. Currently, there is no way to visualize basement membranes, pia or arachnoid mater, and the spaces. It is therefore hard to observe cells moving into these spaces and barriers.
The study of these small spaces is extremely difficult. Currently, there is no way to visualize basement membranes, pia or arachnoid mater, and the spaces. It is therefore hard to observe cells moving into these spaces and barriers.
Most research has been in mice which are different than humans in important ways. The dura and arachnoid are much thicker in humans. The spaces between the layers and spinal cord are much bigger. The CSF spaces are much bigger. So travel distances of cells are much greater. While immune cells might pass through the choroid plexus and travel to the cortex surface in mice, this may not be possible in humans.
The blood vessels appear to be similar. Arteries start in SAS and go to pia then perpendicular into the cortex with many loops looking like a honey comb. Then it goes into venules after the capillaries and then perpendicular out of cortex. Pia veins feed veins in SAS.
Capillaries can be better visualized in humans in the spinal cord surface and the architechture of the pial veins are different. In humans, blood supply in dura is different than CNS. There are faster ways for immune cells into the brain and out. It is not clear about the other two layers. With small differences, these are the same in humans and mice. Measurement of BBB characteristics are useful in determine different human diseases. Most of the variations are not viewable and in the tissues.
Many Barriers and Compartments Between the Brain and Immune System
 Brain tissue has less adaptive and innate responses. But, many immune cells get into and out of the brain in totally different ways than other tissues. Each pathway is different in various unique compartments determined from the characteristics of fluid, lining cells of the organs, astrocytes, choroid cells, and the varied lining of blood vessels.
Brain tissue has less adaptive and innate responses. But, many immune cells get into and out of the brain in totally different ways than other tissues. Each pathway is different in various unique compartments determined from the characteristics of fluid, lining cells of the organs, astrocytes, choroid cells, and the varied lining of blood vessels.
There is a lot of immune activity in the CSF and SAS and then the glial barrier shuts it down where microglia live and signal. Microglia and other mmune cells respond to signals and are aware of issues in the brain tissue and T and B cells can enter with special permission from BBB cells.
The fluid that comes out of the brain tissues along the basement membranes of the blood vessels does not accept cells. It is different from the flow from CSF to the lymph nodes in the neck. The newly discovered drainage from the lymph in the dura also is separated by Blood CSF barrier and the glia barrier.
The problem with all this research is that only specific materials can be visualized at a time if we already know what they are and therefore there is a lot that is not yet visible. Therefore, understanding how immune cells travel and signal in the brain is still emerging. But, all of it is tied to signaling among cells.