 As technology increases our capacity to observe tiny biological systems, communication among cells has been observed with ramifications everywhere in biology. Up until recently, it has been very difficult to observe the behavior of individual cells everywhere in the body, but especially in the vast brain. Observing molecular signals often sent a great distance, such as immune cytokines, are much harder. This data is only now appearing. For many years the brain was considered to have very little immune activity under normal circumstances. But, posts have described how vital T cells travel in the brain and are to normal brain function, even sending signals to stimulate normal cognition or to alter cognition for the “sick feeling”.
As technology increases our capacity to observe tiny biological systems, communication among cells has been observed with ramifications everywhere in biology. Up until recently, it has been very difficult to observe the behavior of individual cells everywhere in the body, but especially in the vast brain. Observing molecular signals often sent a great distance, such as immune cytokines, are much harder. This data is only now appearing. For many years the brain was considered to have very little immune activity under normal circumstances. But, posts have described how vital T cells travel in the brain and are to normal brain function, even sending signals to stimulate normal cognition or to alter cognition for the “sick feeling”.
 Just as neurotransmitters are known to be one vocabulary of the brain, immune cytokine networks are now found to be another. Immune cytokine signals are part of the normal brain conversations among all neurons and glia, and they are very important in infections and abnormal brain states. These signals can be sent either in larger synapses that include brain cells as well as immune cells, or can be secreted into tissue and travel to an immune cell.
Just as neurotransmitters are known to be one vocabulary of the brain, immune cytokine networks are now found to be another. Immune cytokine signals are part of the normal brain conversations among all neurons and glia, and they are very important in infections and abnormal brain states. These signals can be sent either in larger synapses that include brain cells as well as immune cells, or can be secreted into tissue and travel to an immune cell.
With a much smaller number of immune cells in the brain than anywhere else, cytokines can have outsized importance in abnormal states. It is the brain cells themselves that produce most of the cytokines causing inflammation in neurodegenerative diseases involving protein clumps (e.g., Alzheimer’s and Parkinson’s). Immune cells, often coming from outside of the brain, are mostly involved in other types of inflammations such as autoimmune disease multiple sclerosis and brain nfections called encephalitides.
There are now more than 300 cytokine signals identified and all have the property of producing different effects in different cells and situations. Some of these are also called growth factors, interferons (IFN), interleukins (IL), chemokines (travel signals), and lyphokines. One cytokine can cause a cell to grow or do the opposite. Making their effects more complex, various cytokines operate in the context of other signals at the same time and these co stimulators alter their effects, even having one cytokine produce many different effects, even opposite effects. Immune signals in the brain can have varied effects based on circumstances such as the cell producing it, other stimulated cofactors, and the stages of inflammation.
 One example is a factor that triggers a T cell regulating inflammation. But, if combined with the cytokine IL-6, they then produce a cytokine that does the opposite (IL-17). In reality, many cytokines operate at the same time and this makes it very hard to understand what is occurring. Also, each organ responds differently to the same signals in different circumstances.
One example is a factor that triggers a T cell regulating inflammation. But, if combined with the cytokine IL-6, they then produce a cytokine that does the opposite (IL-17). In reality, many cytokines operate at the same time and this makes it very hard to understand what is occurring. Also, each organ responds differently to the same signals in different circumstances.
The brain is most unique because of the relatively small amount of immune cells present as well as the unique supportive glia brain cells that also serve as immune cells. Each region of the brain can respond differently to a cytokine. Inflammation of degenerative illness comes from glia signals, whereas other infections be triggered by signals from immune cells that enter the brain from the blood. Many other effects occur in the special environments produced by cancer cells with communication among all the local supportive cells.
Signals in the Developing Brain
 Signals coming from all cells and even extra cellular matrix direct brain development in the fetus. Cytokines help by supporting brain cells from very early in fetal life. Several families of signals, such as TGFβ, (in a family including BMP or bone morphogenic proteins), and IL-6 (includes LIF leukemia inhibitory factor and CNTF or ciliary neurotrophic factor) each have many variations in protein super families. Both are highly involved in determining the outcomes of stem cells producing neurons, astrocytes, and other glia in the brain. These signals are a vast subject in themselves.
Signals coming from all cells and even extra cellular matrix direct brain development in the fetus. Cytokines help by supporting brain cells from very early in fetal life. Several families of signals, such as TGFβ, (in a family including BMP or bone morphogenic proteins), and IL-6 (includes LIF leukemia inhibitory factor and CNTF or ciliary neurotrophic factor) each have many variations in protein super families. Both are highly involved in determining the outcomes of stem cells producing neurons, astrocytes, and other glia in the brain. These signals are a vast subject in themselves.
The cytokine IL-34 from cortex, olfactory regions, and hippocampus neurons is vital for microglia in the adult, but its effect is still questioned in development. Similar but different signals regulate macrophages in other tissues, which are relatives of microglia. This cytokine may be involved in Alzheimer’s and affects the blood brain barrier tight junctions. This cytokine is vital for normal function but might be vital in disease also.
Neuro Inflammation and Degeneration
 Previous posts have described how neurons can produce all of the symptoms of inflammation not just pain, as a form of neuroplasticity. But, the word neuroinflammation is used also for inflammation in the brain generally, which can be quite different. This can be from strokes and trauma where astrocytes and microglia produce a large number of different cytokines. In diseases like multiple sclerosis (MS), immune cells come from outside of the brain and produce cytokines and autoimmune damage. Cytokines can increase damage and stop inflammation. When networks are altered, either can happen.
Previous posts have described how neurons can produce all of the symptoms of inflammation not just pain, as a form of neuroplasticity. But, the word neuroinflammation is used also for inflammation in the brain generally, which can be quite different. This can be from strokes and trauma where astrocytes and microglia produce a large number of different cytokines. In diseases like multiple sclerosis (MS), immune cells come from outside of the brain and produce cytokines and autoimmune damage. Cytokines can increase damage and stop inflammation. When networks are altered, either can happen.
Damage to Brain Tissues
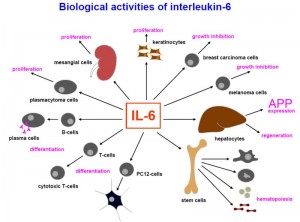 Alarmins are signal molecules that alert glial cells to damage in the brain. With damage from amyloid-β peptide (Aβ) and tau, a wide range of receptors on microglia respond (SRA1, 2, CD36, RAGE) and the more well-known Toll like receptors (TLRs). TLRs bind to the amyloid clumps themselves. When blood cells produce inflammation, an array of cytokines are produced and these also respond to tissue damage—IL-2, IL-23, IL-6, IL-1β.
Alarmins are signal molecules that alert glial cells to damage in the brain. With damage from amyloid-β peptide (Aβ) and tau, a wide range of receptors on microglia respond (SRA1, 2, CD36, RAGE) and the more well-known Toll like receptors (TLRs). TLRs bind to the amyloid clumps themselves. When blood cells produce inflammation, an array of cytokines are produced and these also respond to tissue damage—IL-2, IL-23, IL-6, IL-1β.
IL-33 and 34 are triggered by tissue damage and neurodegeneration. These are inside oligodendrocytes while they are making myelin and then released with damage. Astrocytes and microglia respond to this and produce many other cytokines that increase repair and decrease damage. But, they can also promote damage.
In chronic situations, like degenerative diseases that develop over many years, cells can become exhausted and alter their responses. Many opposite findings have emerged where the same signals hurt rather than help the situation.
Too much of the signal IL-6 increased abnormal amyloid. But, it also increased microglia efforts to eat it. TNF is increased with Alzheimer’s. Too much IL-1β reduced some amyloid and increased microglia
But, other research showed the exact opposite. That is, getting rid of these cytokines helped reduce amyloid and tau and helped the brain function.
It is true that many cytokines are produced when accumulations of abnormal proteins form. The signals come from glia and they increase inflammation—IL-1, 6, 12, 23, and TNF. These seem to help at first and then when they become chronic, they hurt the tissues. At first, they clear some of the abnormal protein, then they increase it. They hurt by producing as yet unknown other factors.
Inflammation Disease in the Brain
In dementia, there are few outside immune cells in the brain. The activity is from the glia functioning as immune cells. When outside immune blood cells invade, the situation is different. Some responses are the same from glia when leukocytes enter the brain—IL-1, IL-6, and TNF. But, the leukocyte cytokines take prominence. They stimulate glia to alter their production of signals and they call for more leukocytes to enter the brain.
Experimentally produced inflammation of the brain in mice is called EAE (for experimental autoimmune encephalomyelitis). In some ways it is similar to MS. It can be produced from antigens to myelin and other parts of the brain. The disease is caused by Th cells (T helper cells) that are uniquely produced in this situation. These Th cells that respond can be injected into others and produce the same disease. These Th cells produce specific cytokines.
There are cytokines that stimulate the production of this specific type of T cells and there are special cytokines that are produced by the T cells.
 IL-23 comes from both glia and APCs (antigen presenting cells). We are just learning, that like T cells, there are many different types of APCs with different actions. The APC superfamily T-12 super family is made of sub units p35 and p40. IL-23 is made up of subtypes p19 and p40. These APCs are the cells that present antigens to T cells to activate them. These occur in lymph tissues outside of the brain and with the interaction they alter the T cells. IL-23 helps produce MS and EAE and stimulates the change to Th cells before they enter the brain. It is not known what the small amount of IL-23 produced by microglia does.
IL-23 comes from both glia and APCs (antigen presenting cells). We are just learning, that like T cells, there are many different types of APCs with different actions. The APC superfamily T-12 super family is made of sub units p35 and p40. IL-23 is made up of subtypes p19 and p40. These APCs are the cells that present antigens to T cells to activate them. These occur in lymph tissues outside of the brain and with the interaction they alter the T cells. IL-23 helps produce MS and EAE and stimulates the change to Th cells before they enter the brain. It is not known what the small amount of IL-23 produced by microglia does.
As well, IL-1 and 6 outside of the brain are both necessary to make Th cells and maintain them. IL-6 is necessary to stimulate EAE. In the brain these cytokines keep the Th cells in the state that causes damage. Their natural tendency without these cytokines is to help not hurt the brain inflammation.
 T cells are very dynamic and can alter their appearance and function continually. They need stimulation to continue a particular activity. The environment with these many signals can allow T cells to totally change their types. Also, T cells can produce many different cytokines at once producing variable behaviors and effects.
T cells are very dynamic and can alter their appearance and function continually. They need stimulation to continue a particular activity. The environment with these many signals can allow T cells to totally change their types. Also, T cells can produce many different cytokines at once producing variable behaviors and effects.
In the brain, T cells can change to produce IL-17a and F, and IL-22 as well as others. The cells that pick up these signals are connective tissue cells, astrocytes, and lining cells. They produce holes in the barriers of the blood brain barrier, which allows more blood cells and T cells to come into the brain. Astrocytes then produce damaging signals and calls for more blood cells into the brain
Cytokines that Increase Inflammation – IFN and TNF
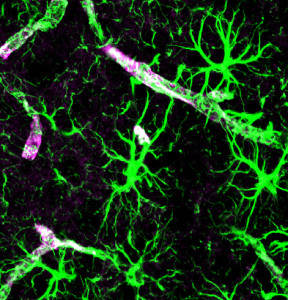 Although some cytokines that have been associated with MS for example appear to increase inflammation, this is not certain since there are so many factors and each cytokine as multiple opposite effects. Both injecting and blocking a factor increased inflammation. Results vary with the timing of injections—early application increased inflammation and later did the opposite. IFNγ can stop Th and IL-17. Without this cytokine there are more Th cells. With it, astrocytes produce cytokines and it also increases tight junctions in the blood brain barrier (BBB). Surprisingly, the same IFNγ has different effects in the cortex and spinal cord with opposite astrocyte signaling. In experiments, inflammation was down in brain, up in spinal cord. This could be different effects on the BBB versus blood CSF barriers.
Although some cytokines that have been associated with MS for example appear to increase inflammation, this is not certain since there are so many factors and each cytokine as multiple opposite effects. Both injecting and blocking a factor increased inflammation. Results vary with the timing of injections—early application increased inflammation and later did the opposite. IFNγ can stop Th and IL-17. Without this cytokine there are more Th cells. With it, astrocytes produce cytokines and it also increases tight junctions in the blood brain barrier (BBB). Surprisingly, the same IFNγ has different effects in the cortex and spinal cord with opposite astrocyte signaling. In experiments, inflammation was down in brain, up in spinal cord. This could be different effects on the BBB versus blood CSF barriers.
One signal is produced only by lymphocytes and therefore doesn’t cause brain protein clumping. But, T cells with it can decrease abnormal amyloid. Meanwhile, other research showed increased amyloid plaques.
TNF is another signal that goes both ways and can be sent from many kinds of cells. It is soluble, found on membranes, and triggers many different receptors. It can kill oligodendrocytes possibly from less astrocyte uptake of glutamate. It helps T cells cross the BBB with many more adhesion molecules for the transfer with signals from lining cells including astrocytes.
Without TNF, MS relapsed more. This was through medications blocking TNF. TNF effects involve multiple receptors.
Conversations of T Helper Cells (Th) with Other Immune Cells
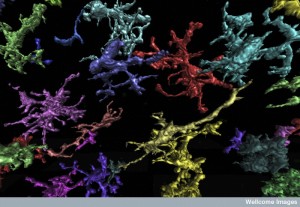 Although it has been shown that leukocytes have affects in the brain, the effects of helper T cells are less clear as they cause damage related to MS. These T cells seem to be the organizers of the other cells including those that eat debris—phagocytes. These eating phagocytes can also cause damage and cause damaging reactive oxygen species (ROS). Microglia are closely related to the phagocytes that come from outside the brain. Neutrophils also help cause MS. Helper T cells can signal to all of these increasing damage. Multiple different vital cytokines and chemokines regulate the actions of all of these cells.
Although it has been shown that leukocytes have affects in the brain, the effects of helper T cells are less clear as they cause damage related to MS. These T cells seem to be the organizers of the other cells including those that eat debris—phagocytes. These eating phagocytes can also cause damage and cause damaging reactive oxygen species (ROS). Microglia are closely related to the phagocytes that come from outside the brain. Neutrophils also help cause MS. Helper T cells can signal to all of these increasing damage. Multiple different vital cytokines and chemokines regulate the actions of all of these cells.
Another gets into the act when monocytes are attracted from the blood and become phagocytes in the brain. They can eat myelin.
Immune Signals in the Brain
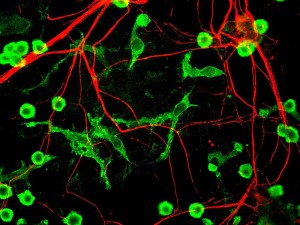 Cytokines have been known to contribute to MS for some time. But, now these same cytokine pathways are shown to be involved in all degenerative brain diseases. MS has leukocytes producing cytokines causing more inflammation and destruction. Glia cells are now known to be involved. Now conversations are found using many different new cytokines with many different immune cells in the brain, notably helper T cells. These T cells talk with all other cells.
Cytokines have been known to contribute to MS for some time. But, now these same cytokine pathways are shown to be involved in all degenerative brain diseases. MS has leukocytes producing cytokines causing more inflammation and destruction. Glia cells are now known to be involved. Now conversations are found using many different new cytokines with many different immune cells in the brain, notably helper T cells. These T cells talk with all other cells.
The question is not whether cytokines are influencing disease, but what are the exact conversations. The same signals from varied cells will cause different effects. Multiple overlapping conversations also influence the outcome. Finding the exact signals will determine future treatments for many different brain diseases.