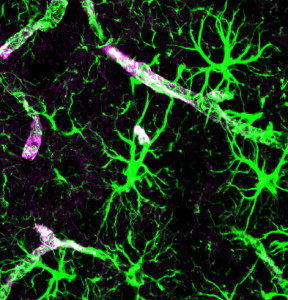
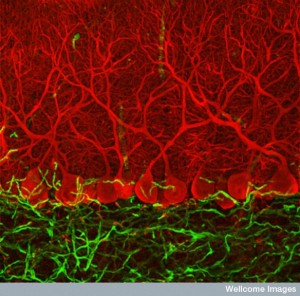
Up to 40% of the brain is made up of astrocytes—many more than neurons—with a network larger than the neuronal connectome. Other posts have described how astrocytes are critical for every aspect of creation, maintenance, and pruning of neuronal synapses. Another post described how complex individual astrocytes can be with specific sub-compartments of different calcium signaling. Now, it has been found that astrocytes are extremely diverse—perhaps as diverse as neurons. Previous posts described that there are at least a thousand different species of neurons. This post describes the search for the immense complexity of astrocyte diversity.
Up to 40% of the brain is made up of astrocytes—many more than neurons—with a network larger than the neuronal connectome. Other posts have described how astrocytes are critical for every aspect of creation, maintenance, and pruning of neuronal synapses. Another post described how complex individual astrocytes can be with specific sub-compartments of different calcium signaling. Now, it has been found that astrocytes are extremely diverse—perhaps as diverse as neurons. Previous posts described that there are at least a thousand different species of neurons. This post describes the search for the immense complexity of astrocyte diversity.
Astrocytes completely tile the brain, with a barrier and regulator near every blood vessel as well as attachments to every neuron and synapse. It was long assumed that astrocytes are similar throughout the brain. But, recent research shows they are quite varied in different regions, neuronal circuits, and small micro-circuits. They take on multiple different shapes and have a vast capacity for unique signaling and modulation of ions. Malfunctioning astrocytes are very important in brain disease. Astrocytes are the connection between neuronal activity and the regional blood flow.

The ratio of astrocytes to neurons varies across brain regions with more astrocytes per neuron in the cortex and more neurons per astrocyte in the cerebellum. There are fewer astrocytes among neuronal cell bodies (grey matter), such as in the layers of hippocampus. Large amounts of astrocytes exist in white matter near axons and dendrites. Astrocytes look differently in grey (protoplasmic astrocytes) and white matter (fibrous astrocytes).
This post will describe the recent research about fantastic astrocyte diversity. First, there is a brief summary of information from other posts.
Summary of Astrocyte Functions
A previous post describes astrocytes creating, maintaining and pruning synapses; and greatly influencing circuits and neuronal connections.
Funtions include:
 Maintaining balance of critical ions, such as taking up potassium during action potentials.
Maintaining balance of critical ions, such as taking up potassium during action potentials.- Picking up neurotransmitters like glutamate from the synaptic cleft
- Storing the only energy in the brain in the form of glycogen, and determining the amount of glucose entering the brain through their connections with synapses and blood vessels.
- Forming a non-overlapping web structure that controls all blood flow in the brain.
- Providing nutrients and digest waste for neurons.
- Regulating synapse formation, maturation, maintenance, pruning and neuroplasticity through secreted signals and direct contact.
- Using both electric gap junctions (see post on electrical synapses) and neurotransmitters.
Other important findings include:

From CopperKet Great complexity has been found in calcium signaling in multiple small compartments of the astrocytes in the many different processes.
- Oligodendrocytes have gap junctions with astrocytes.
- Neurons can have 100,000 synapses, but astrocytes connect thousands of different neurons at the same time in a much greater network.
A previous post described extremely complex local calcium signaling communicating to small regions of axons, dendrites and the neuronal cell bodies. Astrocytes greatly influence whether neurons provide excitatory or inhibitory signals, such as regional inhibition during phases of sleep. They, also, regulate the potassium in the extra cellular space affecting the transmission of the action potential. They regulate every aspect of neuroplasticity changes.
Astrocyte neurotransmitters from vesicles are critical to the function of synchronous brain waves, including local fast gamma communication. They use a variety of neurotransmitters and remove neuronal neurotransmitters in the synapses. They maintain homeostasis as well as altering signals when necessary. Secreted astrocytes molecules have many different effects. Adenosine decreases the neuron’s activity and glutamate increases it.
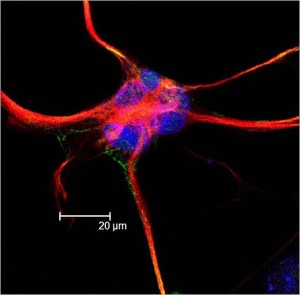
Human astrocytes are vastly more complex than rodents, uniquely, contributing to human level cognition. Human astrocytes are 20 times larger and can coordinate a vast amount of synapses—up to millions.
Astrocytes regulate neuroplasticity and particularly dendrite spines. Actin that forms in these processes is highly mobile and responsive to calcium signaling affecting the complex tasks of regulating the synapse, the local circuit, the distance circuit and the meaning in the neuronal signal
Astrocytes send energy particles and ions back and forth between the neurons and the blood vessels. They can draw nutrients from a great distance to feed a particular neuron. This is especially important during stress, seizures and other crises. Astrocyte calcium signaling is very variable, at times the entire cell relating to thousands of neurons or one local axon. The organelles respond to local synapses.
Measuring Astrocyte Diversity
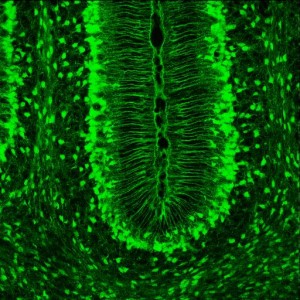 One thing that makes study of astrocytes difficult is that they have stable potassium levels with no conventional electrical current. They, also, have many small branches that stick out creating sub-regions with different calcium properties, making study by conventional means impossible.
One thing that makes study of astrocytes difficult is that they have stable potassium levels with no conventional electrical current. They, also, have many small branches that stick out creating sub-regions with different calcium properties, making study by conventional means impossible.
One marker for astrocyte variability is glial fibrillary acidic protein (GFAP), which shows different levels not only in different regions but, also, in local groups. Hippocampus astrocytes have this protein, whereas thalamus astrocytes do not. In the cortex, middle layers don’t and superficial and deep do. White matter astrocytes show it and gray matter don’t. Also, there are five different varieties of GFAP dispersed through these areas (?, ?, ?, ?, ?). One problem with this marker is that it occurs in other types of cells, as well.
Other markers are glutamate transporters with one more abundant in the hippocampus and another in the cerebellum. A protein related to gap junctions is in gray matter, but not white matter. Potassium channel KIR4.1 is variable in different regions of white and gray, with highest in hippocampus and cerebellum (see post on vast complexity of potassium channels). Also, genetic analysis of a wide range of proteins shows great diversity with different types of stimulation.
In one region, there can be many very diverse astrocytes. In one small area of the cortex, a small group of astrocytes have particular adhesion molecules. These show variable responses in many different astrocytes. In fact, there are a wide variety of different molecules now showing increasing diversity in local astrocytes.
Astrocyte Shape
There are large shape differences in astrocytes that change during particular activities. For example, visible changes of shape occur in the hypothalamus during dehydration, lactation and during birth. Recently, the fine structures that stick out of the astrocyte have been studied and show a wide variation in shape. These are often called leaflets, branchlets, lamellae, sheets, astrocyte processes, perisynaptic processes and fingers. Most of the studies have been in mice, but it is known that human astrocytes are far more complex with much greater variability, but this is difficult to study.
One astrocyte has up to a dozen major arms and thousands of small processes that are near the synapses. There are a large number of small fingers, with 90% of the surface area of an astrocyte in these large branches, small branchlets and smaller leaflets. Astrocytes take up one particular region and have small inter-digitations with the next astrocyte in the tile. The smallest leaflets can only be seen by electron microscopes, not optical.
One or two of the branches are unique and are connected to end-feet that sit on the blood vessels and regulate the blood flow. The other branches with leaflets don’t have this function.
 The number of synapses connected to one astrocyte varies tremendously, as well, as does the way that the leaflets are located near the synapse. These relationships are changed when the brain is injured and in different brain diseases. In the hippocampus one astrocyte is connected to over 100,000 synapses; half have leaflets touching the synapse. In the cerebellum all synapses have attached leaflets, and in the neocortex 30%. In the cerebellum astrocytes are unique and called “radial astrocytes”.
The number of synapses connected to one astrocyte varies tremendously, as well, as does the way that the leaflets are located near the synapse. These relationships are changed when the brain is injured and in different brain diseases. In the hippocampus one astrocyte is connected to over 100,000 synapses; half have leaflets touching the synapse. In the cerebellum all synapses have attached leaflets, and in the neocortex 30%. In the cerebellum astrocytes are unique and called “radial astrocytes”.
The smallest processes, the leaflets, are in the shape of a thin, jagged sheet, filled with ribosomes and glycogen and no mitochondria, microtubules or ER. They cover about a third of a dendrite’s surface and are more prominent in bigger synapses, possibly related to the activity level. When the synapse is active, leaflets are actively moving. They are stimulated by calcium and glutamate. With more neuroplasticity, there is more leaflet movement. The changes of shape of the leaflets appear to be related to the regulation of the synapse activity. The activity, also, is related to gap junction proteins.

The closer the leaflets are to the dendrite and synapse, the more effects they have on the neuronal circuit and the behavior of the animal. Leaflets are critical in the original development of neurons in the fetus and their dendrites.
Radial glial cells are bipolar-shaped cells spanning the entire developing cortex. They are stem cells producing neurons, astrocytes and oligodendrocytes. Radial astrocytes are used as scaffolds for neurons to reach their final destinations. Radial astrocytes show a particular AMPA calcium mechanism that alters the shapes of the leaflets. This glutamate mechanism alters the shapes of the astrocytes by different stimulated by extra cellular calcium signals of different types.
Single Astrocytes
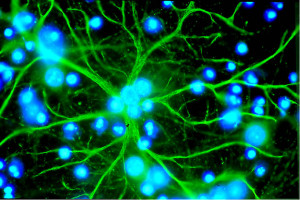
A previous post described internal calcium signaling of astrocytes. Recent research shows that different sub compartments of the astrocyte have different kinds of calcium signals and that these sub-compartments interact with particular neurons in particular ways.
Now, it is shown that specific branches and branchlets have many different types of internal calcium signals.
- Calcium signals unrelated to neuronal firing.
- In the CA1 and dentate of the hippocampus related to the neuron firing.
- A large calcium wave, unrelated to neuronal firing occurs on an entire branch or more.
- A calcium signal in the entire astrocyte, which comes from very active neuronal firing.
- A special type in the end feet.
- Over the entire astrocyte related to the astrocyte movement.
- A very long lasting, up to a minute, and in an extremely small region of a branchlet.
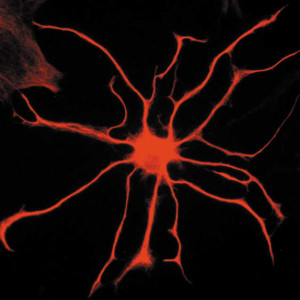
These many different types of calcium signals occur differently in different regions and different types of astrocytes. It is important to remember that these various calcium signals are variable in specific branches, branchlets, and leaflets and that they are not tied to signals in the cell body.
The actions of these various signals are just being studied, but they have a wide range of effects:
- Changing blood vessel size
- Releasing many different signals and trophic factors
- Altering potassium levels
- Regulating the uptake of neurotransmitters in the synapse
- Modulating the neuron
- Movement of the leaflets
- Altering the axon
- Altering the action potential
- Synchronizing neuron activity
- Releasing immune factors
- Altering membrane potential states in neurons
Along Circuits
In the hippocampus, glutamate release triggers glutamate receptors on the astrocyte with responses of CA1 astrocytes. The increase of calcium triggers special gliotransmitters (neurotransmitters from glia) such as ATP regulating neuronal release of neurotransmitters. In the dorsal ganglia there are several types of calcium signals triggered either by a neurotransmitter or neuronal action potential. The astrocytes serve to notice neuronal activity locally and then regulate other neurotransmitter release at that synapse.
In the CA3 the function is different. Astrocytes use large calcium signals throughout an astrocyte during bursts of neuronal activity. They don’t provide the synapse regulating functions, but do help neurons synchronize with others. The difference appears to be the way the leaflets are used, being much closer in the CA1 group.
For neuroplasticity in the hippocampus, a basal level of astrocyte calcium is important, but large spikes are not used for this purpose. Individual astrocytes can have different effects on the functioning of the neuronal circuit.
In the cerebellum the situation is different. There are at least three different kinds of calcium signals. One—a flare—is intermittent without a clear trigger but occurs in widespread regions during the animal’s voluntary movement. The other two are active most of the time—called sparkles in one process and bursts spreading over many astrocytes
Both the hippocampal and cerebellar astrocyte responses may be due to specific animal states such as arousal.
In the thalamus, there are peptides that are similar to the tranquilizer medications benzodiazepines—endozepines. These are present in astrocytes in the thalamus nuclei and astrocytes are necessary for their tranquilizing effects. Astrocytes behave differently in relation to the GABA circuits thought related to the tranquilizing effects of benzodiazepines, where users may experience benzodiazepine withdrawal symptoms while trying to come off this drug.
 In the medulla, a nucleus measures blood H+, CO2 and O2 chemistry related to regulation of breathing. In this mechanism, astrocytes release the critical ATP to regulate the breathing rate. They respond especially to pH with increased calcium signals.
In the medulla, a nucleus measures blood H+, CO2 and O2 chemistry related to regulation of breathing. In this mechanism, astrocytes release the critical ATP to regulate the breathing rate. They respond especially to pH with increased calcium signals.
The spinal cord has many different types of astrocytes in different locations. They have a variety of very specific types of astrocytes that operate in distinct different neuronal circuits.
There are two major types of calcium signals in neuronal circuits–global and local. One consists of wide ranging global effects such as during the animals movement when different regions communicate through specific types of astrocytes (such as between cortex and cerebellum.) These waves operate slowly. The large global waves function with local circuits as well such as when a visual local signal is stimulated with the total movement of the animal.
There are many examples of global astrocyte signaling working with local circuit signaling.
Astrocytes in Disease
When there is damage to the brain, astrocytes change their shape, number and function—called astrogliosis. But, it has been recently found that inherent problems with astrocytes can stimulate these same reactions.
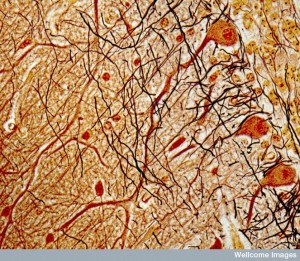 In models of Huntington’s disease, astrocytes have abnormal huntingtin protein. The disease is associated with abnormal astrocytes in the striatum, not other areas. These abnormal states can occur before astrogliosis. One key finding in Huntingtin’s is the abnormal function of spiny neurons that is caused by abnormal astrocytes. In humans, astrocytes show gradual increase in hyperactivity as the disease progresses.
In models of Huntington’s disease, astrocytes have abnormal huntingtin protein. The disease is associated with abnormal astrocytes in the striatum, not other areas. These abnormal states can occur before astrogliosis. One key finding in Huntingtin’s is the abnormal function of spiny neurons that is caused by abnormal astrocytes. In humans, astrocytes show gradual increase in hyperactivity as the disease progresses.
In Alzheimer’s disease mouse models, there is increased basal calcium in astrocytes near the sites of amyoid plaques.
In brain injuries, astrogliosis is varied to respond to the specific insult. Responses to inflammation and ischemic damage are very different. Those related to injury have a gradient of different amounts of astrocytes at different distances from the damage. The astrocytes can dramatically change the genetic networks operating. The cell itself can enlarge, form scar tissue or create entirely new types of network structures. Differences can occur very locally with multiple different types of astrocytes working in different ways to deal with the damage.
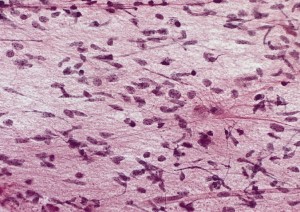 Although there are many responses, two specific astrocyte alterations are prominent—increased size and scar tissue formation. Astrocytes form a border around the damage or inflammation that separates the normal and abnormal brain tissue.
Although there are many responses, two specific astrocyte alterations are prominent—increased size and scar tissue formation. Astrocytes form a border around the damage or inflammation that separates the normal and abnormal brain tissue.
New astrocytes are used for these borders in the cortex, hippocampus, spinal cord and thalamus. These astrocytes are abnormal in that they don’t stay in neatly tiled in specific locations as other astrocytes, but rather encircle the region with many arms that entangle with nearby astrocytes forming a more secure border. These new cells come from the astrocytes surrounding blood vessels and migrating stem cells. Inside the border, debris can be removed and the infection treated. They interact broadly with many immune cells. These new structures may be permanent.
In more generalized brain damage, astrocytes change their shape and structure and stay in their original location. They alter genetic networks to produce many new products and become enlarged. They continue their local processes and connections and function. These changes can be reversible. They alter their modulation of ions, neurotransmitters and transporters.
Fantastic Astrocyte Diversity
 A previous post noted how foolhardy it is to consider that understanding of the brain will occur by only mapping neuronal networks, when astrocytes are not only almost half of the brain, but have vast complexity in their modulation of the neuronal networks. Recent work just adds to the complexity of astrocytes, showing that they are extremely diverse—like the thousand different types of neurons.
A previous post noted how foolhardy it is to consider that understanding of the brain will occur by only mapping neuronal networks, when astrocytes are not only almost half of the brain, but have vast complexity in their modulation of the neuronal networks. Recent work just adds to the complexity of astrocytes, showing that they are extremely diverse—like the thousand different types of neurons.
Where is the direction for this fantastic diversity? Any mental event, including thoughts and feeling, triggers vast changes in many brain cells. The neuroplasticity occurs all over wide circuits at the same instant and with many different mechanisms. There has been no center found in the brain to direct all of these processes. Each cell responds in different unique ways to these mental events. Each genetic network, microtubule, actin scaffold, and organelle responds instantly to these mental events.
How can this be directed? How can mental events trigger all of these vast changes instantly in thousands of places at once? It obviously cannot be directed from genetic networks in one cell, or from a network of neurons.
Isn’t it reasonable to consider mind operating in and interacting with each and every cell, organelle and molecule?