 Neuroplasticty is the way a brain makes lasting alterations of its own circuits when responding to experience. A vast array of mechanisms have been discovered for neuroplastic changes at synapses. In fact, large circuits engage in simultaneous varied mechanisms at synapses across the brain (See Post). There is no current way to explain these global whole brain phenomenon with extremely intricate molecular processes in many places at once. Another unanswered question is how the brain arranges for diverse large and small circuits to organize speeds of signals so that multiple inputs arrive in sync and decisions can be made at synapses. Recent research shows complex variations in myelin patterns and shapes provide large-scale coordination of circuit speeds. This research shows myelin facilitation of whole brain neuroplasticity.
Neuroplasticty is the way a brain makes lasting alterations of its own circuits when responding to experience. A vast array of mechanisms have been discovered for neuroplastic changes at synapses. In fact, large circuits engage in simultaneous varied mechanisms at synapses across the brain (See Post). There is no current way to explain these global whole brain phenomenon with extremely intricate molecular processes in many places at once. Another unanswered question is how the brain arranges for diverse large and small circuits to organize speeds of signals so that multiple inputs arrive in sync and decisions can be made at synapses. Recent research shows complex variations in myelin patterns and shapes provide large-scale coordination of circuit speeds. This research shows myelin facilitation of whole brain neuroplasticity.
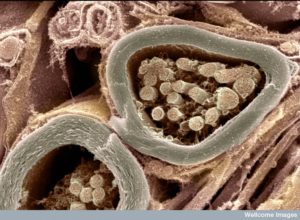
 For decades, myelin has been understood as a simple insulator of axons that increases the speed of neuronal transmission. There was little known about oligodendrocytes—the cells that make myelin. Recently, vast complexity in the process of oligodendrocytes’ wrapping and compressing layers of myelin has been discovered. In fact, this three dimensional process is one of the most elaborate in nature. It involves signaling between the oligodendrocytes and neurons for many different stages of the process—determining where myelin will be built and where first contact occurs between the two cells; wrapping the layers and growing them along the axon; determining how many layers are wrapped and where the nodes will be; and the intricate compaction of the layers. The process, also, keeps lines of communication through the compacted regions and maintains the structure sometimes for decades. Also, stem cells making oligodendrocytes travel throughout the brain determining where the myelin will be built. All of this is connected with the needs of the neuron and astrocyte networks across the brain. It is vital for all learning and cognition.
For decades, myelin has been understood as a simple insulator of axons that increases the speed of neuronal transmission. There was little known about oligodendrocytes—the cells that make myelin. Recently, vast complexity in the process of oligodendrocytes’ wrapping and compressing layers of myelin has been discovered. In fact, this three dimensional process is one of the most elaborate in nature. It involves signaling between the oligodendrocytes and neurons for many different stages of the process—determining where myelin will be built and where first contact occurs between the two cells; wrapping the layers and growing them along the axon; determining how many layers are wrapped and where the nodes will be; and the intricate compaction of the layers. The process, also, keeps lines of communication through the compacted regions and maintains the structure sometimes for decades. Also, stem cells making oligodendrocytes travel throughout the brain determining where the myelin will be built. All of this is connected with the needs of the neuron and astrocyte networks across the brain. It is vital for all learning and cognition.
Laying down myelin is far more complex than synapses and neuro muscular junctions. The immensely elaborate process of modeling, remodeling, and maintenance of myelin occurs all over the brain simultaneously. Neuroplasticity has been assumed to be a vast array of mechanisms related to synapses. Now, new research shows an entirely new dimension, where myelin is a vital part of neuroplasticity for all memory, not just motor memory. Myelin neuroplasticity is directed by electrical activity of the neuron and by elaborate communication with oligodendrocytes and many other cells. The stem cells that produce oligodendrocytes are, also, directed by conversations with epithelial cells in blood vessels.
Neuroplasticity Related to Speed of Conduction
 This new global view of neuroplasticity comes from data that correlates greatly increased complexity in myelin related to the timing and conduction of the signals along the axons. With so many circuits connecting at one moment, the speed of the current has to be adjusted to reach the synapses at the correct moment. These speed alterations are controlled and modified as needed by the different patterns of myelin.
This new global view of neuroplasticity comes from data that correlates greatly increased complexity in myelin related to the timing and conduction of the signals along the axons. With so many circuits connecting at one moment, the speed of the current has to be adjusted to reach the synapses at the correct moment. These speed alterations are controlled and modified as needed by the different patterns of myelin.
Myelin was considered to be relatively inert—a fatty insulator making signals faster. This picture was complicated recently by finding new patterns of myelin in layers of the cortex and elsewhere (see the previous post Complexity in the Myelin Code). Other research finds that some neurons are un myelinated in specific regions so that local communication can occur sideways off of the axon to local immune cells (see post on immune signaling from axons). Now, the entire picture is greatly complicated by finding myelin can optimize the timing of transmission of signals with different structures including the number of wraps and the placing of nodes. Along with this are findings about elaborate communication between neurons and oligodendrotyces.
Brain imaging shows that with learning there are new neurons in the hippocampus and diverse alterations in synapses. The new research finds alterations in the white matter (axons) related to myelin and the neuronal electrical activity.
Myelin Production Influenced by Neuron Electrical Activity
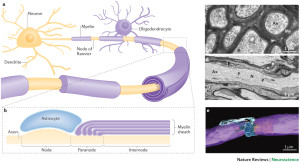
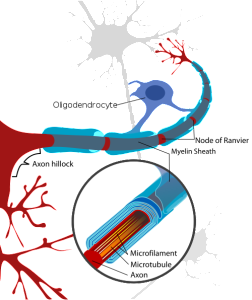 Oligodendrocytes are multi polar cells that produce fatty myelin sheaths and wrap them around the axons many times. This sheath greatly enhances the speed of the electrical signal along the axon. Without it, the electrical wave depends on movement from each small sodium and potassium pore to the next all along the membrane. With myelin, the electrical signal rapidly jumps between nodes with long stretches of insulated myelin.
Oligodendrocytes are multi polar cells that produce fatty myelin sheaths and wrap them around the axons many times. This sheath greatly enhances the speed of the electrical signal along the axon. Without it, the electrical wave depends on movement from each small sodium and potassium pore to the next all along the membrane. With myelin, the electrical signal rapidly jumps between nodes with long stretches of insulated myelin.
Before synapses could be visualized a hundred years ago, it was assumed that myelin was related to the specific functioning of the neuron. This view went out of favor and was ignored until recently. As regions of the brain form myelin and the connections begin to operate, specific abilities coincide with the development. The gradual growing of myelin down the long motor neurons from the brain produce a stepwise increase in function in babies—first, stiffening neck, then arm movement, sitting up, standing up and walking. Later, speaking and cognitive abilities appear with myelinated brain circuits. It is only as young adults that the frontal lobes myelinate with the dawning of judgment and adult decision making.
Now it has been learned that throughout life the process of myelination is influenced by the neuron firing its action potential electrical signal. This produces signals from the membrane of the axon that influences the cells that make myelin.
Coordination of Signal Timing in Circuits for Global Neuroplasticity
 An important question is how neuronal networks can handle an extreme amount of information through regulation of the rate of speed of electrical signals between parts of a circuit. This is necessary for multiple inputs to have complex interactions. Regulating this timing needs a global perspective of the entire system. Memory and learning requires various circuits to be timed and coordinated. One simple example is conditioned learning experiments, where there has to be both a signal and a conditioned response at the same time.
An important question is how neuronal networks can handle an extreme amount of information through regulation of the rate of speed of electrical signals between parts of a circuit. This is necessary for multiple inputs to have complex interactions. Regulating this timing needs a global perspective of the entire system. Memory and learning requires various circuits to be timed and coordinated. One simple example is conditioned learning experiments, where there has to be both a signal and a conditioned response at the same time.
Signals must arrive within milliseconds to be effective in complex circuits. Plasticity for synapses depends on simultaneous arrivals so that decisions can be appropriately made for the next axon firing in the postsynaptic neuron. The next signal is weakened if one contributing circuit arrives late and strengthened if early within a very narrow time scale. Myelin creates rapid speeds, but it must produce the exact level of speed needed for the neuroplasticity. In fact, speeds of myelinated axons vary widely in a scale of a thousand—a tenth of a yard per second to 200 yards per second.
 As well as the number of wraps the the spacing of nodes, another consideration is different types of electrical signals. Some sensory signals are simply a singular spike of a certain intensity. In much of the brain however, there are circuits that provide timed pulsed signals at different frequencies for particular background tasks.
As well as the number of wraps the the spacing of nodes, another consideration is different types of electrical signals. Some sensory signals are simply a singular spike of a certain intensity. In much of the brain however, there are circuits that provide timed pulsed signals at different frequencies for particular background tasks.
Timed pulses occur in circuits of the default mode network and, also, with attention, sleep, top down regulation of sensory input and perception, to name just a few. These need regulation of the velocities of signals at various rates. For example, brain wave oscillations, such as gamma waves can be altered by delays in the speed of the conduction by even a very small amount. When conduction is altered it can lead to brain disease related to white matter changes.
Neuroplasticity in White Matter
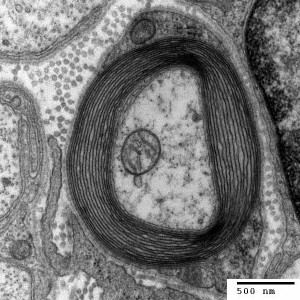
Changes in white matter (axon bundles) have been noticed with neuroplasticity related to specific learning, such as reading and playing musical instruments. Imaging currently can barely make out these nano changes in axon bundles. Even though changes cannot be well visualized in imaging studies, it is now known that alterations in white matter (axon bundles) occur related to the amount of electrical use of a nerve.
Oligodendrite stem cells are critical to these alterations in white matter. Increasing stimulation for learning (enriched environments) increases stem cells for oligodendrotyes (OPCs or oligodendrocyte progenitor cells). Exercise increases OPCs in the cortex and the corpus callosum. When new OPCs were blocked, mice couldn’t learn as well in motor learning tasks.
Surprisingly, myelin is produced within five days of training in brain regions for learning, along with neuroplasticity changes in neurons and astrocytes. Increased myelin is correlated with the amount of learning. Some changes occur very rapidly after only two hours of learning in humans and animals. The large white matter coming out of the hippocampus memory center is called the fornix, which was altered with 2 hours of learning.
In car racing video game studies, the amount of white matter changed is based on increased skills. Recent studies show that when action potentials occur, the protein that makes up myelin can be rapidly produced. It also produces a complex that further connects the axon and the glia cell.
White matter changes from learning are correlated with alterations in myelin in several studies. In fact, it is correlated with the exact amount of learning. Other studies show alterations in the structure of white matter with motor skills that are just learned. This includes more myelin, but only electron microscopic studies will be able to tell if this involves greater thickness of the myelin.
Initial Axon Segment Similar to Node of Ranvier
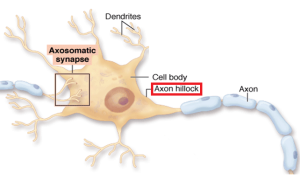 The first part of the axon (initial axon segment or AIS) near the cell body is unique in that only half of it is myelinated and therefore part of it is similar to half a node of Ranvier. The AIS is where the electrical spike is started with very complex ion channels that regulate the entire process. The AIS determines not just when the electrical current will start but how big it will be and its electrical “shape”. This determines exactly how much neurotransmitter will be discharged at the synapse, as well as the exact neurotransmitter to be sent.
The first part of the axon (initial axon segment or AIS) near the cell body is unique in that only half of it is myelinated and therefore part of it is similar to half a node of Ranvier. The AIS is where the electrical spike is started with very complex ion channels that regulate the entire process. The AIS determines not just when the electrical current will start but how big it will be and its electrical “shape”. This determines exactly how much neurotransmitter will be discharged at the synapse, as well as the exact neurotransmitter to be sent.
A very significant finding is that the AIS actually alters is size and position to regulate the complex information that is sent. Since the AIS is similar to a node of Ranvier, it is thought that similar alterations occur at each node with local stimulation. There is evidence of changes in the sizes of nodes and the length between them during neuroplasticity in the ear.
Oligodendrocyte Communication with Neurons
 An important question is how oligodendrocytes know that neuroplasticity and learning is occurring. This is accomplished through a wide range of signals with neurons and other cells, such as astrocytes and blood vessel epithelium. Another question is how oligodendrocytes know what electrical activity is occurring in the nearby neutron and how many wraps of myelin should be produced or modified. There is great variation in the sheath thicknesses and nodes can, also, be altered.
An important question is how oligodendrocytes know that neuroplasticity and learning is occurring. This is accomplished through a wide range of signals with neurons and other cells, such as astrocytes and blood vessel epithelium. Another question is how oligodendrocytes know what electrical activity is occurring in the nearby neutron and how many wraps of myelin should be produced or modified. There is great variation in the sheath thicknesses and nodes can, also, be altered.
Oligodendrocytes produce many different neurotransmitter and growth factor receptors to communicate with neurons and other cells. They, also receive genetic messages in vesicles (exosomes – see post on exosome communication between cells).
 In addition, ion channels all along the oligodendrocyte’s membrane are used for communication. Oligodendrocytes do not themselves conduct electrical action potentials as axons do, so the ion channels can help the oligodendrocyte understand the electrical activity of the neuron that is nearby. Another special electrical communication between cells (ephaptic coupling – see post on electricity in the brain) uses ion channels for direct communication between two cells.)
In addition, ion channels all along the oligodendrocyte’s membrane are used for communication. Oligodendrocytes do not themselves conduct electrical action potentials as axons do, so the ion channels can help the oligodendrocyte understand the electrical activity of the neuron that is nearby. Another special electrical communication between cells (ephaptic coupling – see post on electricity in the brain) uses ion channels for direct communication between two cells.)
Until recently it was thought that signaling from neurons only occurs at synapses. In fact, signals between neurons and oligodendrocytes are sent far from the synapse along the axon. These signals are secreted either in vesicles or as free molecules. ATP, glutamate, adenosine and GABA are among the signals that are secreted and they stimulate more oligodendrocytes and Shwann cells. This transmission in un myelinated axons not near a synapse is similar to the way neurons communicate from axons directly with immune cells in sections without myelin.
Vesicles Directly from the Axon
 Along the axon, vesicles are, also, released from the axon without the complex setups seen at the synapse. Along the axon there are bulges, called axonal varicosities that produce vesicles for transmission from the middle of the axons in the autonomic system, particularly in the gut. This same process occurs for acetylcholine transmission in the cortex. The process is generally called vesicle fusion.
Along the axon, vesicles are, also, released from the axon without the complex setups seen at the synapse. Along the axon there are bulges, called axonal varicosities that produce vesicles for transmission from the middle of the axons in the autonomic system, particularly in the gut. This same process occurs for acetylcholine transmission in the cortex. The process is generally called vesicle fusion.
These vesicles stimulate the oligodendrocyte for more myelin production. This same type of signaling was previously found between astrocytes and neurons related to calcium in the nerves of the eye.
Neurotransmitters Without Vesicles
 Another way that signals are sent from the axon is through channels in the membrane without vesicles. ATP is sent from these channels related to the neuronal electrical activity levels. Special locations on the axon produce the ATP release, which is related to the production of myelin at different stages. Myelin has receptors for these signals including ATP, AMPA, NMDA and kainite.
Another way that signals are sent from the axon is through channels in the membrane without vesicles. ATP is sent from these channels related to the neuronal electrical activity levels. Special locations on the axon produce the ATP release, which is related to the production of myelin at different stages. Myelin has receptors for these signals including ATP, AMPA, NMDA and kainite.
A specific neurotransmitter can have many different effects in different ways. Adenosine from neurons slows the production of oligodendrocytes, but ATP doesn’t. ATP slows Schwann cells, but not oligodendrocytes. There are many other factors that stimulate, inhibit and regulate this very complex process. Glutamate can stimulate more stem cells or trigger cell death. Many other types of cells release ATP and other related purine molecules and this, also, modulates the myelin process. Many factors such as neureglin and BDNF are also involved. Very elaborate neurotransmitter release between oligodendrocytes and neuron axons, not from synapses, regulate the myelin process in neuroplasticity.
There are other factors in this communication. Under extreme conditions of stress, such as lack of oxygen and energy, normal mechanisms for re uptake of neurotransmitters reverse and start to work in the other direction sending signals to the nearby glia. Nitric oxide and other growth factors are sent from neurons to oligodendrocytes. This signaling can activate astrocytes and blood vessels to help stimulate oligodendrocytes to produce more myelin.
Stem Cells Throughout the Brain Produce Oligodendrocytes
 Stem cells making oligodendrocytes (OPCs) produce the largest amount of new cells in the brain at any given time. These stem cells have many different receptors related to this continual process of remodeling myelin structures with learning. While some neuroplasticity occurs with learning in particular regions such as the hippocampus, the neuroplastic remodeling of myelin occurs all over the brain simultaneously.
Stem cells making oligodendrocytes (OPCs) produce the largest amount of new cells in the brain at any given time. These stem cells have many different receptors related to this continual process of remodeling myelin structures with learning. While some neuroplasticity occurs with learning in particular regions such as the hippocampus, the neuroplastic remodeling of myelin occurs all over the brain simultaneously.
These OPCs engage in synaptic coupling with neurons using glutamate and GABA. Once new oligodendrocytes produce myelin, the coupling fades as they mature. Signals from neurons stimulate OPCs to make more oligodendrocytes, which then builds myelin. For cognition, the synapse has very rapid exact neurotransmission. But, to gradually build myelin over the course of hours or days, the signals can be less intense.
Myelin Wrapping Process is Very Complex
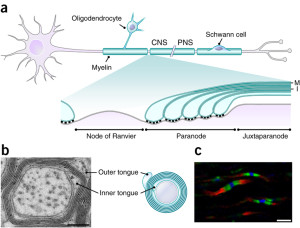 Triggering oligodendrocytes to make myelin in special ways involves a very elaborate multi step process. Myelin must be produced by triggering elaborate timed genetic networks including correct placement of the first wrap of the sheath around the membrane of the axon. It cannot just be left there. The sheath has to, then, be serviced for a very long time, often decades.
Triggering oligodendrocytes to make myelin in special ways involves a very elaborate multi step process. Myelin must be produced by triggering elaborate timed genetic networks including correct placement of the first wrap of the sheath around the membrane of the axon. It cannot just be left there. The sheath has to, then, be serviced for a very long time, often decades.
The oligodendrocyte sends out a hand like process to touch the axon membrane where a very unique junction region is created. It is similar in some ways to the extremely complex way that neurons make contact with muscles and form the neuro muscular junction (see post on Complexity in Neuro Muscular Junctions describing this back and forth interactive process of the two cells). Oligodendrocyte to neuron junctions were called a “spot weld” fifty years ago when it was first observed by electron microscope.
The special junction is triggered, at least, by glutamate signaling from vesicles. Glutamate triggers NMDA and mGluR receptors on the oligodendrocyte processes stimulating an increase of calcium. Immunoglobulin on the neuron is necessary to trigger the oligodendrocyte’s activity. Very elaborate cascades produce a multi unit protein factory built out of scaffolding tubules, as well as receptors, to direct the unfolding of the complex myelin producing process. Signaling regulates the proteins needed to make myelin and the way they are placed in the membrane to hold it in place. Very complex cascades of molecules include kinase enzymes, integrin and molecules based on the lipid cholesterol. The original immunoglobulin is stimulated by particular frequencies from the neuron and is only essential for the first layer of the wrapping of the sheath.
 Wrapping of the myelin will not occur without a special measurement of the size of the axon circumference including special curvature sensors. The axon has to be above a particular circumference for the process to start. The cellular process makes a spiral sheet in 3D. It is an amazing process where a length of 100 micrometers (10-6) with 10 spirals takes 6400 square micrometers of membrane. The wrapping process is very hard to understand and there are several competing models to grasp it, currently.
Wrapping of the myelin will not occur without a special measurement of the size of the axon circumference including special curvature sensors. The axon has to be above a particular circumference for the process to start. The cellular process makes a spiral sheet in 3D. It is an amazing process where a length of 100 micrometers (10-6) with 10 spirals takes 6400 square micrometers of membrane. The wrapping process is very hard to understand and there are several competing models to grasp it, currently.
A leading tongue crawls around the axon leaving a sheet of myelin. Additional wraps and lengthening of the total myelin happens at the same time. A process from oligodendrocytes connects to the myelin and more wraps occur there. The inner layers are smaller than the later layers. The tongues are pushed by actin. Later the inner layers grow and catch up to the covering layers. As the second wrap occurs, the bottom layer is growing bigger. Each layer attaches permanently in a particular placement near where the node of Ranvier will be forming small paranode attachment areas.
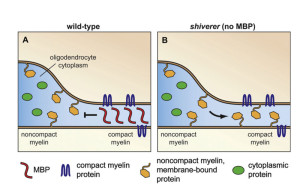 The myelin wrapping grows in two ways at once—more layers and the entire sheath gets longer at each level. When three layers are formed, a complex process of compaction starts at the outside layers. During compaction, special channels are created to allow transport of materials through the compacted areas and the growth zone. Compaction occurs both along the length of the axon and around the circular wrap at the same time. When compaction has eliminated most of the cytoplasm by pushing it out, a special protein binds the two membranes together as if in a tight junction between cells. All proteins with more than 30 amino acids are removed. This process is similar to a zipper.
The myelin wrapping grows in two ways at once—more layers and the entire sheath gets longer at each level. When three layers are formed, a complex process of compaction starts at the outside layers. During compaction, special channels are created to allow transport of materials through the compacted areas and the growth zone. Compaction occurs both along the length of the axon and around the circular wrap at the same time. When compaction has eliminated most of the cytoplasm by pushing it out, a special protein binds the two membranes together as if in a tight junction between cells. All proteins with more than 30 amino acids are removed. This process is similar to a zipper.
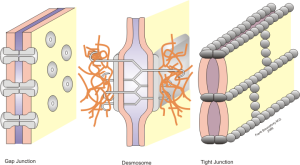
Adhesions and compaction also occur between the extracellular membranes of two layers of wrap. Compaction is not fully understood, but it involves eliminating electrostatic repulsion and other forces. Many receptors and signals such as neureglin regulate the number of wraps, which determines the properties of the electrical signal for neuroplasticity.
Just as there are many different kinds of neurons discovered recently (see post How Many Different Kinds of Neurons Are There), at least six distinct types of oligodendrocytes are now known based on gene networks. Stem cells arise from particular regions and produce unique oligodendrocytes for that region.
Mechanisms at the node of Ranvier are just being discovered. There are more vesicles and channels at the nodes than other regions and it is in close contact with astrocytes, the same that are surrounding the synapse. The node has altered axon transport as well with intense signaling to the oligodendrocytes and astrocytes.
Myelin Neuroplasticity is Very Hard to study
 What makes myelin so hard to study is that it encompasses a very large area of the brain and yet it relies on many complex nano mechanisms. The effects of myelin are global considerations of the speed of large circuits throughout the brain. Therefore, myelin must be studied at many scales of magnitude at the same time from electron microscopes to axons that are a yard long. On the other hand, other forms of neuroplasticity in synapses are studied at nano chemical scale.
What makes myelin so hard to study is that it encompasses a very large area of the brain and yet it relies on many complex nano mechanisms. The effects of myelin are global considerations of the speed of large circuits throughout the brain. Therefore, myelin must be studied at many scales of magnitude at the same time from electron microscopes to axons that are a yard long. On the other hand, other forms of neuroplasticity in synapses are studied at nano chemical scale.
Another complication is that oligodendrocytes are in close communication with blood vessels. The brain has more types of cells than any organ in nature and they must travel to exact places over long distances. Oligodendrocytes migrate the most of all cells throughout life. It was recently found that stem cells to make oligodendrocytes travel along blood vessels. Elaborate signaling between the oligodendrocytes and the blood vessels define their routes.
Surprisingly it is the blood vessels that signal constantly for the cells to remain as general stem cells, until they arrive at their proper destination and switch to unique types of stem cells making particular types of oligodendrocytes for the region. Like other endothelial cells (see posts on Intelligent Intestinal Epithelial Cell and Intelligent Skin Epithelial Cell), blood vessel endothelium have vital intelligent functions through signaling. Blood vessels perform different functions in different brain regions and are different shapes. The stem cells also alter the vessels in some ways.
 Other recent studies show that myelin has critical functions in the grey matter of the cortex. It is involved in determining where axons will sprout and where synapses will form. A previous post discussed how myelin has elaborate patterns in different cortex segments (see post on New Complexity of Myelin Code). The AIS is longer in Cortex layers III and IV.
Other recent studies show that myelin has critical functions in the grey matter of the cortex. It is involved in determining where axons will sprout and where synapses will form. A previous post discussed how myelin has elaborate patterns in different cortex segments (see post on New Complexity of Myelin Code). The AIS is longer in Cortex layers III and IV.
Another problem is that other animals don’t have the same myelin complexity as humans and are quite variable from species to species. (Mice have only 10% white matter where humans have 50%).
Neuroplasticity research has largely focused on the alterations of synapses. This is extremely complex by itself (see Fanntastic Array of Neuroplasticity Mechanisms). But, studying myelin all over the brain is much more complicated and the way that oligodendrocytes give feedback over time is not clear despite communication by exosomes, electricity and neurotransmitters.
Myelin Facilitation of Whole Brain Neuroplasticity
In order to have coherent circuits functioning at large distances across multiple regions of the brain, the timing of individual neuronal electrical currents have to be very accurately in sync. The regulation of the timing and the strength and speed of currents in specific neuronal loops are vital factors in neuroplasticity mechanisms. Until recently, it has not been clear how this could be accomplished.
Previously, myelin was considered a simple process, but now research shows that it is a very complex and vital for this coordination and timing. Myelin, and with it the speed and timing of circuits, is altered during normal learning and neuroplasticity. Like other aspects of neuroplasticity, such as regulating synapses, there is a vast array of mechanisms involved in myelin production. Studies of the ways white matter changes during neuroplasticity are just beginning.
Many different kinds of learning produce complex alterations in myelin amount, shape, size, pattern and distribution. These changes are part of neuroplasticity for many kinds of learning, not just the habit motor type. It is surprising how rapidly these changes can occur. It was previously thought that the only changes in myelin in the adult were damaging or responding to damage. Now, it has been shown that there is dynamic active modeling and remodeling of myelin in white matter in children and adults with learning. This includes re consolidation of memory. The alterations in myelin occur through oligodendrocyte stem cells that wander throughout the brain along blood vessels and then stop to differentiate into particular types of oligodendrotyes to make specific types of myelin.
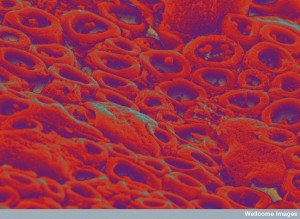 The junction of a neuron and oligodendrocyte that forms myelin is vastly complex—much more complex even that synapses and neuro muscular junctions. There are many stages that have to precisely timed and executed. It is necessary to coordinate huge amounts of material and then build precise shapes. It has to be spaced exactly with all the correct placement of ion channels. The enormous structure has to be maintained sometimes for a lifetime.
The junction of a neuron and oligodendrocyte that forms myelin is vastly complex—much more complex even that synapses and neuro muscular junctions. There are many stages that have to precisely timed and executed. It is necessary to coordinate huge amounts of material and then build precise shapes. It has to be spaced exactly with all the correct placement of ion channels. The enormous structure has to be maintained sometimes for a lifetime.
Once again, we must ask where the direction lies for stem cells wandering all over the brain constantly building and updating myelin of particular types to keep speeds in sync for neuroplasticity. No one can consider this a random process. This is another example of intelligent communication between a vast number of cells simultaneously all over the brain. All of this is correlated with mental activity.
It seems reasonable that only a mind interacting with all these cells and molecules in the brain at the same time could coordinate such global complex events.