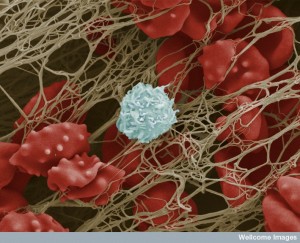
 Platelets have been mostly thought of as a fragment of a cell that stops bleeding and not much else. They can, also, produce damage because clotting can block arteries causing strokes and heart attacks. So, it is a surprise that recent research finds platelets have many other roles including being critical players in defense against microbes. In fact, they are able to find microbes and rapidly receive and secrete messages that stimulate immune defense. They have been found to be vital in calling for leukocytes and work with the immune cells to fight infections. They are, even, able to aid the very intelligent T cell when it communicates with B cells to enhance antigen responses. They are able to send complex signals and respond to signals from other cells. How can this little piece of cytoplasm, not even a full cell and without a nucleus, perform all of these functions? Does this research show platelet intelligence?
Platelets have been mostly thought of as a fragment of a cell that stops bleeding and not much else. They can, also, produce damage because clotting can block arteries causing strokes and heart attacks. So, it is a surprise that recent research finds platelets have many other roles including being critical players in defense against microbes. In fact, they are able to find microbes and rapidly receive and secrete messages that stimulate immune defense. They have been found to be vital in calling for leukocytes and work with the immune cells to fight infections. They are, even, able to aid the very intelligent T cell when it communicates with B cells to enhance antigen responses. They are able to send complex signals and respond to signals from other cells. How can this little piece of cytoplasm, not even a full cell and without a nucleus, perform all of these functions? Does this research show platelet intelligence?
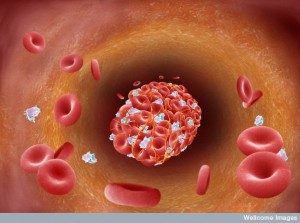 The well-known, absolutely critical, function of the platelet is to stop blood loss that would otherwise kill the organism. This hemostatic function is complex and involves at least three competing forces. The organism must continue the blood flow to every organ at all costs and stopping bleeding can help or hurt this cause. Organs are injured if too much flow is stopped, or if the blood becomes hypercoaguable—a state where there is too much blood clotting. Also, the cells lining blood vessels, endothelial cells, must keep the vessels open and in good order.
The well-known, absolutely critical, function of the platelet is to stop blood loss that would otherwise kill the organism. This hemostatic function is complex and involves at least three competing forces. The organism must continue the blood flow to every organ at all costs and stopping bleeding can help or hurt this cause. Organs are injured if too much flow is stopped, or if the blood becomes hypercoaguable—a state where there is too much blood clotting. Also, the cells lining blood vessels, endothelial cells, must keep the vessels open and in good order.
Platelets Do Much More
 The easiest way for a microbe to enter the body is to follow the path of a trauma or foreign body. Following the opened area, microbes can slip into the tissue or the bloodstream. Therefore, as first responders to injury, it is natural that platelets could have both a central role in maintenance of blood flow and defense against invaders.
The easiest way for a microbe to enter the body is to follow the path of a trauma or foreign body. Following the opened area, microbes can slip into the tissue or the bloodstream. Therefore, as first responders to injury, it is natural that platelets could have both a central role in maintenance of blood flow and defense against invaders.
Injury immediately stimulates a blood clotting reaction that produces thrombin, which rapidly induces platelets to alter their shape and stop the flow of blood locally. Thrombin stimulates receptors on the platelets that attract connective tissue fibers (collagen, laminin, fibronectin, fibrinogen and other molecules). These fibers work with the platelets to plug damage with a scar.
 In creatures much earlier in the evolutionary tree, such as very early vertebrates and invertebrates, there are specialized cells (hemocytes) that provide both blood flow and immune functions. In mammals, cells became more specialized into leukocytes, lymphocytes and platelets.
In creatures much earlier in the evolutionary tree, such as very early vertebrates and invertebrates, there are specialized cells (hemocytes) that provide both blood flow and immune functions. In mammals, cells became more specialized into leukocytes, lymphocytes and platelets.
Mammals are the only creatures with platelets. It was thought that platelets dealt with only regulation of blood flow, leukocytes with inflammation and lymphocytes with regulation of immune function. Recently, it was noticed that platelets still have kept the capacity to help regulate immune responses. But, how can a cell without a nucleus do so much?
Basic Platelets
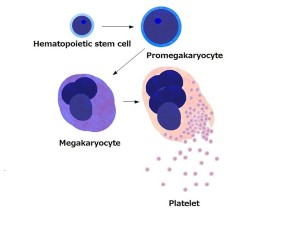
Platelets are small–2 to 4 μm (micron is 1/1000 of a millimeter)– and live for a week. Platelets are convex on both sides–shaped like a lens. They are manufactured by the large megakaryote blood cells in the bone marrow, which become 20 times larger when making platelets.
A hormone from the liver and kidneys, thombopoietin, is the signal to make more platelets. Each megakaryote makes 1 to 3 thousand platelets and a trillion platelets are manufactured daily. From the bone marrow, they flow into the blood stream. Some are stored in the spleen for emergency situations and released by signals from the sympathetic nervous system (fight and flight).
Platelets are born without a nucleus and their own DNA, but have many messenger RNAs that allow them to make proteins by themselves. The only DNA is in mitochondria.
Activation
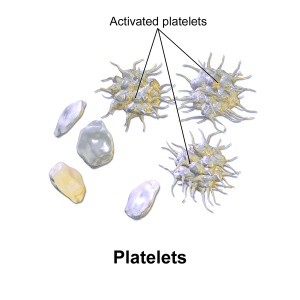
When first formed, platelets look like little wrinkled brains. They have a large amount of extra membrane that is folded into channels, so that the platelet can change shape quickly. When activated by receptors, they go through dramatic shape change.
First, platelets attach to the broken vessels and then they become activated. When activated they produce many special receptors (using the ribosomes they already have, not DNA in a nucleus). Calcium increase triggers microtubules and actin to dramatically alter the scaffolding and the platelet produces many long arms that make it look like a small astrocyte or dendritic cell.
The shape change has been described in three stages. The first produces the multiple arms and legs. The second spreads out the body of the platelet and the third makes a thicker central body.
The changes occur through an interaction of the microtubules and a change in the membrane with its invaginations. The motor of these changes is just below the membranes, which creates a dramatic increase in membrane surface and then produces the arms and legs. It is a remarkable process that occurs without stretching or adding lipids to the membrane.
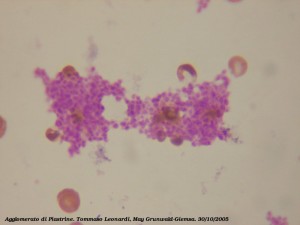
The next phase is when the platelets join their arms and form aggregates.
Releasing Granules
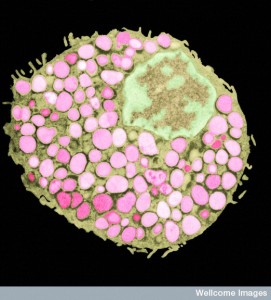 One group of white blood cells is called granulocytes because they contain many vesicles filled with chemicals that can be secreted. These include neutrophils, basophils and eosinophils. Like larger granulocytes, platelets, also, have three types of granules.
One group of white blood cells is called granulocytes because they contain many vesicles filled with chemicals that can be secreted. These include neutrophils, basophils and eosinophils. Like larger granulocytes, platelets, also, have three types of granules.
- δ- includes molecules that regulate blood flow—containing nucleotides such as ADP, amines such as histamine and serotonin, and ions such as Ca2 and PO3–.
- α- has many different proteins and enzymes that attach to and kill microbes. They, also, have factors that increase clotting.
- λ-, lysosomal vesicles are filled with many enzymes that will alter the shape of the clot so that healing can occur.
To use these remarkable capacities, platelets change from the circular type that is well known, to amoeboid cells with pseudopods (arms) that can interact with microbes and even secrete reactive molecules that can kill microbes. The platelet uses a very large amount of specific molecules for various different microbes.
Platelets Have Many Sensory Receptors
 There are more platelets than other blood cells and they have many pattern recognition receptors that often serve as the first responders to injury and invaders. When they recognize trouble, platelets deliberately move to the center of the trauma or infection and become the most numerous cells there. They specifically accumulate where there are microbe generated proteins and complement proteins. When the platelet receptor picks up specific microbe peptides, it rapidly changes its shape by altering the tubule scaffolding, stimulates calcium and releases some of its vesicles.
There are more platelets than other blood cells and they have many pattern recognition receptors that often serve as the first responders to injury and invaders. When they recognize trouble, platelets deliberately move to the center of the trauma or infection and become the most numerous cells there. They specifically accumulate where there are microbe generated proteins and complement proteins. When the platelet receptor picks up specific microbe peptides, it rapidly changes its shape by altering the tubule scaffolding, stimulates calcium and releases some of its vesicles.
Platelets have chemokine receptors for all four of the major types (C, CC, CXC, CX3C), so they can be drawn from a great distance. Other mechanisms draw the platelets there—they respond to chemical signals related to collagen that, also, attract neutrophils. Importantly, platelets at the site release a class of molecule called kinocidins, that draw many other immune cells.
Platelets, also, have receptors for specific immune molecules such as IgG and IgA and IgE, CRP and other cytokines. Platelets receptors detect pathogen-associated molecular patterns (PAMPs) by using the well-known Toll-like receptors (TLR) and various types of cytokines in the IL-1 family. These responses form a cascade inside the cell even without a nucleus, including TNF and other major signals.
Platelets Use Complex Signaling
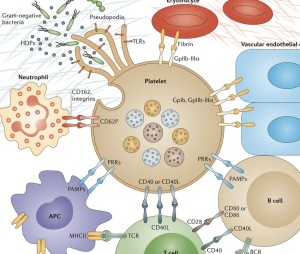 Like other intelligent larger cells, platelets engage in very complex signaling with many important Toll Like receptors (TLR), such as TLR1, TLR2, TLR4, TLR6, and TLR9. In different situations they use IL-6 and COX-2 in cross talk with other cells. It is too complex to relate here, but in many different situations, a vast number of different signals are used by this cell that has no nucleus. They respond differently to various subtypes of microbes and, then, transmit very specific cytokines from their large vocabulary. These cytokines cause other particular immune cells to activate in very precise ways.
Like other intelligent larger cells, platelets engage in very complex signaling with many important Toll Like receptors (TLR), such as TLR1, TLR2, TLR4, TLR6, and TLR9. In different situations they use IL-6 and COX-2 in cross talk with other cells. It is too complex to relate here, but in many different situations, a vast number of different signals are used by this cell that has no nucleus. They respond differently to various subtypes of microbes and, then, transmit very specific cytokines from their large vocabulary. These cytokines cause other particular immune cells to activate in very precise ways.
As with all mechanisms, sometimes they go awry. Platelets can mistakenly stimulate increased inappropriate reactions that create clots in the wrong places.
Amplification of Important Signals
The platelet has a unique amplification system for important signals with many different mechanisms. One response to staph aureus includes release of δ granules with ADP and ATP and then a succession of stimulation by neighboring platelets releasing α granules with specific proteins and kinocidins. The microbe, then, attempts to stop the granule secretion and to break down the specific factors that are released.
Direct Attack on Microbes
Like the larger granulocyte cells such as neutrophils, platelet granules have many different proteins and peptides. Platelets have recently been found to release at least four distinct categories of anti microbe molecules in their granules.
- Kinocidins including CSCL4, CSCL7 and CCL5 – these can function as chemokines, that is attracting specific immune cells or directly killing microbes.
- Defensins including BD2 and Tb4
- Proteins made from breaking up other molecules
- Thrombocidins, which are made by enzymes cutting CXCL7
Kinocidins Are Very Complex Platelet Secreted Molecular Signals
 Kinocidins are a complex large class of molecules. They include CSCL4, CSCL7 and CCL5, which can function as chemokines, attracting specific immune cells, as well as, directly killing microbes. Kinocidins are either full sized proteins, or pieces of proteins that are created by enzymes cutting the secreted proteins. They are similar to molecules housed in monocytes and macrophages. Their concentration is sensed by special thrombin receptors in the platelets that can increase the messenger RNA to make more. This increases the messenger RNA product by 30 to 100 times.
Kinocidins are a complex large class of molecules. They include CSCL4, CSCL7 and CCL5, which can function as chemokines, attracting specific immune cells, as well as, directly killing microbes. Kinocidins are either full sized proteins, or pieces of proteins that are created by enzymes cutting the secreted proteins. They are similar to molecules housed in monocytes and macrophages. Their concentration is sensed by special thrombin receptors in the platelets that can increase the messenger RNA to make more. This increases the messenger RNA product by 30 to 100 times.
The major kinocidin is the peptide CXCL4, also known as platelet factor 4. It has a complex structure with different regions and effects—some to kill microbes, some to communicate with other cells. It has an interesting module that allows it to rapidly evolve to keep up with corresponding microbe evolution. These multiple parts are stabilized into a very specific structure.
Platelet kinocidins have unusual properties. They are released into the blood when triggered by microbe invasion. Those from the much larger immune cells are only used locally. Platelet kinocidins, also, serve as immune signals to recruit other cells, as well as stimulating a larger response. The kinocidins of larger immune cells don’t have these elaborate functions.
 Another unusual property is that platelet kinocidins can be cut into smaller pieces that are, also, powerful poisons to microbes. Thrombin, which is the basic initiator of clots, functions as an enzyme to cut platelet kinocidins making many different small powerful molecules that attack microbes. These small pieces, called thrombocidins, have different capacities to attack different microbes.
Another unusual property is that platelet kinocidins can be cut into smaller pieces that are, also, powerful poisons to microbes. Thrombin, which is the basic initiator of clots, functions as an enzyme to cut platelet kinocidins making many different small powerful molecules that attack microbes. These small pieces, called thrombocidins, have different capacities to attack different microbes.
To make matters even more complex, the kinocidins attract specific phagocytes to fight inflammation, which produce enzymes that further break down these pieces into smaller pieces that are also able to attack microbes. In fact, microbes inadvertently produce enzymes that help produce these smaller molecules, some which are inactivated, but some ultimately attack the microbe itself.
The kinocidins are able to distinguish membranes of the microbes and the immune cells, often by the ability to bind to specific lipids that are in membranes. They are able to understand which to use at different levels of pH. They are able to choose among many small particles for many different microbes.
Platelet Kinocidins Can Attack Many Different Microbes

The platelet kinocidins are very effective against viruses, bacteria, fungus and protozoa. They have recently been shown to be critical in defense against HIV-1. s Other platelet factors have been shown to work against S aureus, strep, and others. They appear to limit strep heart infections.
Plasmodium falciparum, the cause of malaria, is greatly affected by platelet factors. It appears that CXCL4 is critical to human defense against malaria. The platelet factor can uniquely get into infected red blood cells. This is the major way that malaria can be killed in red blood cells. A larger number of platelets represents a greater fight with infection, whereas low platelet counts are caused by monocytes eating platelets.
Platelets have, also, been shown to be effective against many fungi—Candida, Aspergillus, Toxoplasma , Leishmania, and Schistosoma.
Other Platelet Mechanisms Against Microbes
Platelets make other chemicals against microbes. They can make hydrogen peroxide and other reactive oxygen particles. These are critical to combat worms.
Platelets don’t really eat microbes, but they do cover them so that others can eat them. They also form a surface for other complexes, such as the large thrombin protein complex that kills bacteria.
Platelets Call for Help
 One of the most important capacities of platelets is recruiting and helping other larger immune cells. Unique platelet properties include surveillance of the surface of other immune cells, such as macrophages and liver cells. Platelets seem to be critical to finding infected macrophages.
One of the most important capacities of platelets is recruiting and helping other larger immune cells. Unique platelet properties include surveillance of the surface of other immune cells, such as macrophages and liver cells. Platelets seem to be critical to finding infected macrophages.
Platelets are critical in helping neutrophils by actively stimulating receptors that bind to leukocytes and monocytes as well as binding to microbes, thereby connecting the two. This increases the ability of neutrophils to eat bacteria. These complexes built of platelets and leukocytes activate both cells and recruit more. They build larger complexes, called neutrophil extracellular traps, NETs, consisting of DNA strands from the neutrophil, histones and other fibers which attach to all types of proteins and help them to kill bacteria. This is a mechanism of killing bacteria without hurting the tissue.
Also, platelet kinocidins directly increase phagocytosis by neutrophils, which kill microbes inside the cell.
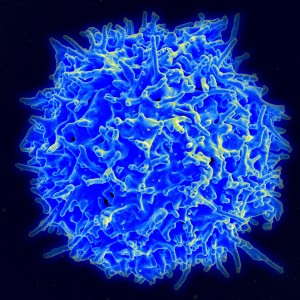 Platelets stimulate immune dendritic cells, which are the key cells that present antigens to T cells and B cells. Platelets rapidly connect with the microbe and aid the dendritic cells in getting antigens from them. When dendritic cells meet the platelets, they signal with a wide range of cytokines. These events greatly increase the specificity of the response against a particular microbe.
Platelets stimulate immune dendritic cells, which are the key cells that present antigens to T cells and B cells. Platelets rapidly connect with the microbe and aid the dendritic cells in getting antigens from them. When dendritic cells meet the platelets, they signal with a wide range of cytokines. These events greatly increase the specificity of the response against a particular microbe.
Platelets directly aid T cells by secreting very specific cytokines and kinocidins, which activate T cells. When viruses have invaded platelets, they signal to enhance the T cells that are required against that virus. This signaling helps the T cells to be ready to attack other infected cells as well. Of the many types of T cells, that both help and hurt different situations, the platelet is able to signal for the one that will enhance the specific response for a specific microbe. Signals from other cells including T cells stimulate many new receptors on the platelets, which brings neutrophils into the defense.
Platelets are able to help B cells build up an army to attack viruses. Without the platelet signals they are unable to perform. The platelets appear to be a necessary communication intermediary between T cells and B cells to help each other.
Microbes Strike Back
 Some microbes are able to hijack platelet mechanisms and use activated platelets to seek out and attack other immune cells. They are, also, able to decrease the clotting effects of platelets and use the platelet itself for increased strength of attack. In one case, the microbe forms a matrix of fibrin and platelets to form an infected clot on a heart valve.
Some microbes are able to hijack platelet mechanisms and use activated platelets to seek out and attack other immune cells. They are, also, able to decrease the clotting effects of platelets and use the platelet itself for increased strength of attack. In one case, the microbe forms a matrix of fibrin and platelets to form an infected clot on a heart valve.
Platelet Intelligence
 Platelets are critical to control blood loss, but also for many immune functions. They have a smaller, but very effective, repertoire of immune receptors and cytokines than the professional larger white blood cells. The fact that they are much smaller and more numerous is an advantage in traveling into difficult terrain. Recently, it was learned that this little cell, with no nucleus or DNA, but messenger RNA and mitochondria, is able to make large numbers of very complex molecules to fight a vast number of different microbes, infections, and injuries.
Platelets are critical to control blood loss, but also for many immune functions. They have a smaller, but very effective, repertoire of immune receptors and cytokines than the professional larger white blood cells. The fact that they are much smaller and more numerous is an advantage in traveling into difficult terrain. Recently, it was learned that this little cell, with no nucleus or DNA, but messenger RNA and mitochondria, is able to make large numbers of very complex molecules to fight a vast number of different microbes, infections, and injuries.
In fact, platelets are at the center of complex communication with a very large number of different cells. They are critical for the function of the leukocyte, dendritic cell, T cell, B cell and neutrophil and stay in constant communication with the vascular endothelial cell and red blood cells. They are able to make a very complex kinocidin protein, which can then be broken down into multiple very powerful specific small particles to fight many different microbes. Kinocidin, also, has a region that evolves along with the microbes to stay effective.
How can the small platelet, without a nucleus, be so intelligent?