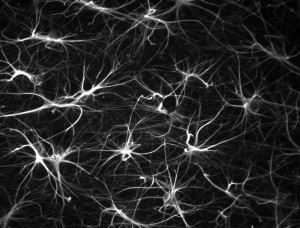
Astrocytes are the neuron’s critical partners—helping to establish synapses, as well as, maintaining and pruning them. Astrocytes—five to ten times as numerous as neurons and making up half of the brain—create a huge scaffold. The astrocyte network signals with calcium fluctuations, while the neuron’s action potential is based on sodium and potassium channels. Astrocytes have many large arm-like processes that both touch and connect neuronal synapses and have feet wrapping around and controlling blood vessel flow in the brain—providing the amount of oxygen needed for a region of neurons (the MRI measure of blood flow). A major question has been how astrocyte calcium signaling links to neuronal signaling. Now, new research shows that the astrocyte calcium fluctuations and oscillations that perform many different critical functions are vastly more complex that previously thought. In fact, along each astrocyte process there are many different compartments with diverse types of communicating oscillations forming a new very complex mechanism of information transfer with a large number of neurons. In this process astrocyte calcium signaling leads to more brain complexity and is much more significant and sophisticated than ever imagined.
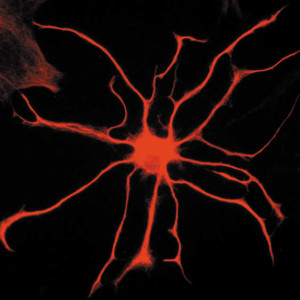
Twenty years ago it was found that in response to synaptic activity, astrocytes increase internal calcium levels. Intracellular calcium levels increased with sensory stimuli, motor activity, and sympathetic and parasympathetic activity. Research showed that astrocytes have complex calcium fluctuations when different neurotransmitters were secreted as well as other brain factors. It was thought that neurotransmitters in the synapse trigger receptors on astrocytes and signaling cascades, which start in the cell body (soma) from calcium released by the endoplasmic reticulum. This production of calcium was thought to produce a wide variety of different signals in the astrocyte. Just recently, it was found that many compartments along the astrocyte processes appear to have their own signaling completely independent of the soma, making astrocyte signaling orders of magnitude more complex. It is possible that these compartments coordinate in some way with the soma signaling.
Recently, also, many types of astrocyte calcium signaling have been discovered. Calcium by itself can trigger receptors in some brain regions—cortex with  NDMA receptors and cerebellum with AMPA receptors. Calcium spikes can also be stimulated by interactions with sodium and calcium channels in the neuron through exchanges of sodium and calcium. Astrocytes, also, show calcium spikes unrelated to neuronal synapse activity, especially in the fetus. In addition, there are other channels that can independently produce calcium fluctuations.
NDMA receptors and cerebellum with AMPA receptors. Calcium spikes can also be stimulated by interactions with sodium and calcium channels in the neuron through exchanges of sodium and calcium. Astrocytes, also, show calcium spikes unrelated to neuronal synapse activity, especially in the fetus. In addition, there are other channels that can independently produce calcium fluctuations.
The old simple mechanism has been found to be inaccurate. Instead, many different types of oscillations in different compartments and many different mechanisms are used by astrocytes to respond to both neuronal neurotransmitters and their own astrocyte gliotransmitters.
Astrocytes Are Half of the Brain
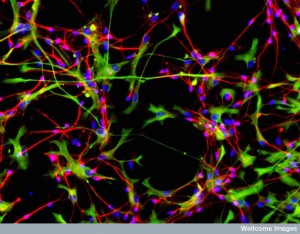 Astrocytes, called radial glia, produce stem cells that produce neurons. As well as providing support, astrocytes take up potassium released during neuron action potentials to maintain ionic balance and pick up neurotransmitters like glutamate and GABA that are secreted at synapses. They store the only energy in the brain in the form of glycogen, and determine the amount of glucose entering the brain through their connections with synapses and blood vessels.
Astrocytes, called radial glia, produce stem cells that produce neurons. As well as providing support, astrocytes take up potassium released during neuron action potentials to maintain ionic balance and pick up neurotransmitters like glutamate and GABA that are secreted at synapses. They store the only energy in the brain in the form of glycogen, and determine the amount of glucose entering the brain through their connections with synapses and blood vessels.
In each region astrocytes form a web like structure, which doesn’t overlap others. Each astrocyte communicates with a large number of neurons, while each neuron can have as many as 100,000 synapses. They control every aspect of synapses including the ionic environment of the extracellular space. Astrocytes provide neurons with nutrients and digest old parts. Recently, dramatic findings show that astrocytes are critical for the entire life cycle of synapses – their formation, neuroplasticity, normal function, and their pruning. (see previous post for details on Astrocyte Control of Synapses).
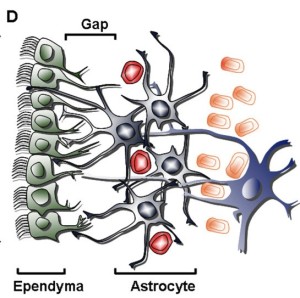 The two ways astrocytes control synapses are through secreted signals and direct contact. The signaling is very complex with many back and forth signals. During migration of new neurons, astrocytes create a pathway of tunnels for the neuron to travel. Astrocytes have receptors in their membrane and secrete many gliotransmitters—signaling molecules from a glia cell. During fetal brain development, neurons cannot receive a synapse without contact first by an astrocyte that “teaches” the neuron how to have a synapse. Adhesion molecules, like neurexin, are critical in this synapse creation.
The two ways astrocytes control synapses are through secreted signals and direct contact. The signaling is very complex with many back and forth signals. During migration of new neurons, astrocytes create a pathway of tunnels for the neuron to travel. Astrocytes have receptors in their membrane and secrete many gliotransmitters—signaling molecules from a glia cell. During fetal brain development, neurons cannot receive a synapse without contact first by an astrocyte that “teaches” the neuron how to have a synapse. Adhesion molecules, like neurexin, are critical in this synapse creation.
Astrocytes send their own calcium signal spikes over wide regions of the brain, just as neurons send action potentials along axons in wide regions. Astrocytes modify neuronal signals by secreting neurotransmitters and their own special gliotransmitters, including glutamate. The details of this vast astrocyte communication is just being discovered.
New Class of Transmitters
 New types of signaling molecules secreted by astrocytes are called gliotransmitters. These signals can participate in the large neuro-immune synapse described in previous posts (this signaling interface consists of neurons, astrocytes, microglia, oligodendrocytes, vascular cells, T cells, macrophages and other immune cells). Gliotransmitters are triggered in different time sequences from milliseconds to seconds and can affect microglia, oligodendrocytes, neurons and vascular cells. Some gliotransmitters are the same as neurotransmitters—glutamate, d-serine—while others are different–ATP (adenosine triphosphate rapidly metabolized into adenosine). These gliotransmitters can have effects similar to neuronal neurotransmitters with simulation, inhibition and neuroplastic effects including long term potentiation LTP and depression LTD.
New types of signaling molecules secreted by astrocytes are called gliotransmitters. These signals can participate in the large neuro-immune synapse described in previous posts (this signaling interface consists of neurons, astrocytes, microglia, oligodendrocytes, vascular cells, T cells, macrophages and other immune cells). Gliotransmitters are triggered in different time sequences from milliseconds to seconds and can affect microglia, oligodendrocytes, neurons and vascular cells. Some gliotransmitters are the same as neurotransmitters—glutamate, d-serine—while others are different–ATP (adenosine triphosphate rapidly metabolized into adenosine). These gliotransmitters can have effects similar to neuronal neurotransmitters with simulation, inhibition and neuroplastic effects including long term potentiation LTP and depression LTD.
Many Different Types of Astrocytes
 The best current research shows a very complex picture that is beyond our current understanding. Conflicting findings in different regions points to a much more sophisticated type of signaling in many different regions and with diverse mechanisms. Increasingly, studies are finding a variety of different types of astrocytes, much as we have found a large number of very different types of neurons. The calcium fluctuations and their effects appear to be quite different in different regions, and most surprisingly, in different compartments of the same astrocyte—even the same arm of one astrocyte.
The best current research shows a very complex picture that is beyond our current understanding. Conflicting findings in different regions points to a much more sophisticated type of signaling in many different regions and with diverse mechanisms. Increasingly, studies are finding a variety of different types of astrocytes, much as we have found a large number of very different types of neurons. The calcium fluctuations and their effects appear to be quite different in different regions, and most surprisingly, in different compartments of the same astrocyte—even the same arm of one astrocyte.
In the hippocampus region of CA3-CA1, one study showed increase of calcium only under certain circumstances. Calcium increases, lasting a long time in one situation, did not lead to modification of synapses or LTP, but using a different research technique they did. 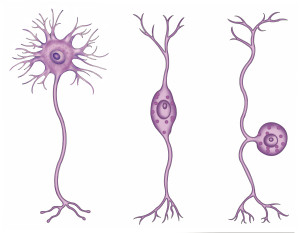 Different research techniques show varied responses in diverse parts of the cell bodies and different regions of the astrocyte network.
Different research techniques show varied responses in diverse parts of the cell bodies and different regions of the astrocyte network.
When looking at the enormous complexity of neurons and their vastly different functions, it is not unreasonable for us to find a similar complexity in the critical network of astrocytes. The problem is that this research is just at the beginning—where neuronal research was a generation ago.
Astrocyte Processes
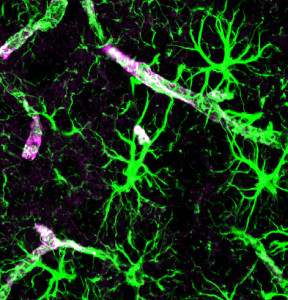 Astrocytes look like stars because of their many arms, called processes, which stick out to perform many different functions. Astrocytes use a unique protein called glial fibrillary acidic protein (GFAP) for building the shape of the processes. Processes are used to guide neurons when they migrate from the ventricle to the cortex. The processes of many astrocytes form a large scaffold structure throughout the entire brain. Astrocyte processes mingle with all elements of the extracellular space including neurons, synapses, axons, dendrites, and myelin. They form a thick layer, which protects the entire cover of the brain and, also, covers the blood vessels with feet that open and close the vessels to determine the blood flow in each region. The astrocyte processes stimulate the formation of tight junctions during fetal growth in the blood brain barrier and form a second barrier to entry of intruders after the blood brain barrier.
Astrocytes look like stars because of their many arms, called processes, which stick out to perform many different functions. Astrocytes use a unique protein called glial fibrillary acidic protein (GFAP) for building the shape of the processes. Processes are used to guide neurons when they migrate from the ventricle to the cortex. The processes of many astrocytes form a large scaffold structure throughout the entire brain. Astrocyte processes mingle with all elements of the extracellular space including neurons, synapses, axons, dendrites, and myelin. They form a thick layer, which protects the entire cover of the brain and, also, covers the blood vessels with feet that open and close the vessels to determine the blood flow in each region. The astrocyte processes stimulate the formation of tight junctions during fetal growth in the blood brain barrier and form a second barrier to entry of intruders after the blood brain barrier.
Astrocyte processes wrap around dendritic spines. They move in and out and change rapidly like amoeba, until they connect exactly with the new dendritic spines, critical for the connection to the postsynaptic neuron.
The current research finds that astrocytes and their processes have a great diversity in the size as well as the frequencies and the cell locations of calcium oscillations.
Astrocyte Processes Show Great Complexity
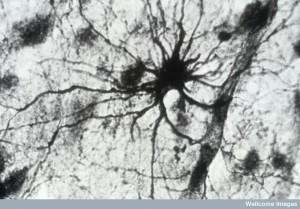 Calcium fluctuations were first studied in the soma (cell body) because it was easiest. Recent research in the cerebellum shows independent calcium activity in small regions of the arms of astrocytes. This activity is not connected to the cell body activity at all. Although it is much more difficult to study these small micro regions, increasingly, unique calcium oscillations are noted in various compartments along the processes.
Calcium fluctuations were first studied in the soma (cell body) because it was easiest. Recent research in the cerebellum shows independent calcium activity in small regions of the arms of astrocytes. This activity is not connected to the cell body activity at all. Although it is much more difficult to study these small micro regions, increasingly, unique calcium oscillations are noted in various compartments along the processes.
The early research in the soma showed that astrocytes only respond when there is strong neuronal stimulation at the synapse. The newer, subtler, research of the processes shows that the small regions in the arms respond in different ways to much smaller synaptic stimulations. In fact, the astrocyte is responding in many different ways at many different levels of activity in the synapse.
 Complexity arises when considering how astrocytes can integrate calcium signals that reflect neuronal activity in different places, at different time scales, and associated with different gliotransmitters.
Complexity arises when considering how astrocytes can integrate calcium signals that reflect neuronal activity in different places, at different time scales, and associated with different gliotransmitters.
Recent research in the hippocampus shows the processes are very active and independent of the calcium activity in the cell body. This signaling activity in the arms, or processes, occurs in multiple different places and is not synchronous with the cell body’s calcium activity. Even a single process has different events occurring at the same time in different places.
Calcium events have been named local or extended—extended being larger communication with other regions of the astrocyte.
The local activity is very rapid and exists only in a small local region. It is not connected with neuronal firing, but may be connected with neurotransmitters that are picked up from the extracellular space. These seem to be part of coordination among a whole group of different regions.
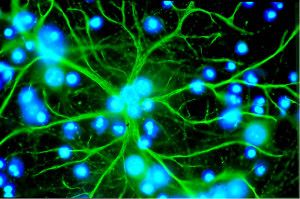
Another extended type of event responds to axons that are nearby. These are large and longer lasting effects and extend over a wider range of the astrocyte regions. The larger events can occur in the same regions as the small local events, with multiple different parts of the expanded event. Integration of many of the smaller events stimulates a larger more powerful effect, such as stimulating more gliotransmitters.
The signal from the astrocyte cell body (soma) is much slower and does not occur as often. It is possible that this is triggered from the combination of the local and expanded events. These soma events appeared to trigger longer opening of the blood vessels (the BOLD signal that is read in MRIs).
A big problem in this research is that different types of experimental techniques appear to trigger different aspects of these complex signals—such as light stimulation, drug blockade and special genetic animal breeds—and therefore cannot, yet, be compared.
Specific Frequencies of Calcium Oscillations
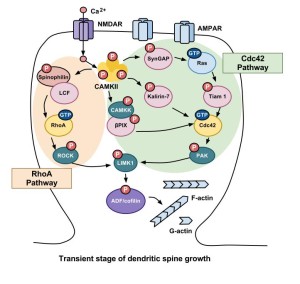 Another complication is that specific frequencies of calcium oscillations trigger different responses (like neuronal oscillations). Smaller fluctuations can stimulate glutamate neurons powerfully. In the cortex, neuroplastic changes of long-term depression correlate with higher frequencies of calcium oscillations.
Another complication is that specific frequencies of calcium oscillations trigger different responses (like neuronal oscillations). Smaller fluctuations can stimulate glutamate neurons powerfully. In the cortex, neuroplastic changes of long-term depression correlate with higher frequencies of calcium oscillations.
Unexpected findings show that there are different types of LTP neuroplasticity stimulated by very different calcium circuits and mechanisms. Specific neurons are involved in different astrocyte responses. In one example, the astrocyte must stimulate cholinergic nerves and activate specific muscarinic receptors to cause the long term potentiation. In another, the LTP did not depend on the cholinergic fibers, but rather, needed d-serine—a neurotransmitter that is added to glutamate to stimulate the special NMDA receptor, which requires two neurotransmitters at the same time. This serine was dependent on specific astrocyte production of extracellular calcium.
Also, astrocytes can receive information from thousands of synapses in large neuronal circuits. They can bridge different unconnected neuronal circuits either in a small local manner or in larger and slower ways.
Different Responses in Different Animals and Ages
Another complication is that completely different astrocyte mechanisms produce LTP neuroplasticity in various types of research animals and ages. Most studies of astrocyte calcium have been in animals in the weeks after birth. In the young brain there is more activity independent of neurons. Later, some activity is coordinated in the soma and then in the compartments of the processes.
Astrocyte Calcium Signaling Leads to More Brain Complexity
 It is impossible to consider understanding or “mapping” the brain without half of the active important cells. Astrocytes not only are critical to create, maintain and prune synapses, but, have their own elaborate communication network that we don’t really understand.
It is impossible to consider understanding or “mapping” the brain without half of the active important cells. Astrocytes not only are critical to create, maintain and prune synapses, but, have their own elaborate communication network that we don’t really understand.
Each area of brain that is studied provides much more complexity than previously thought. A previous post described the new complexity in myelin codes. Now, the very complex ways that calcium oscillations operate in different compartments of the astrocyte is being discovered.
Complications in efforts to “map” the brain already include the fact that neurons send synchronous oscillations wirelessly between sections of the brain, in addition to the wired synapses (See post Limits of Current Neuroscience). Study of neuroplasticity shows that wide circuits of neurons alter synapses simultaneously using many different mechanisms. There are, also, many different types of neurons and circuits, which are constantly changing. Recently, myelin was shown to have its own complex code where  communication can occur laterally and locally from the un myelinated axon in a variety of different ways. (see post New Myelin Code).
communication can occur laterally and locally from the un myelinated axon in a variety of different ways. (see post New Myelin Code).
More than half the brain consists of astrocyte networks and scaffolding. Astrocytes have their own communication through elaborate calcium signaling. Now, great complexity and sophistication has been found in the calcium signals that occur in multiple diverse small compartments of the numerous long processes of astrocytes and communicate with large numbers of connected neurons.