 A small RNA strand able to make ten proteins has become the scourge of much of the world in the form of Dengue fever. This remarkable tiny virus is able to thwart the complex human immune reactions that fight viruses. Using some unique features, it is rapidly spreading through the world and infects millions of people per year—over 100 countries with 3.6 billion people at risk of very serious infection and death. Thankfully, most of the illnesses are mild, but a small percentage of the illness (unfortunately, still as very large number of people) is serious.
A small RNA strand able to make ten proteins has become the scourge of much of the world in the form of Dengue fever. This remarkable tiny virus is able to thwart the complex human immune reactions that fight viruses. Using some unique features, it is rapidly spreading through the world and infects millions of people per year—over 100 countries with 3.6 billion people at risk of very serious infection and death. Thankfully, most of the illnesses are mild, but a small percentage of the illness (unfortunately, still as very large number of people) is serious.
Dengue virus shows unique intelligence and behavior in entering cells with multiple techniques, evading immune responses and, very surprisingly, actively working to stop the effectiveness of vaccines by constantly altering protein shapes.
Other Intelligent Viruses
Previous posts have described the remarkable behavior of several viruses that have very few genes and proteins. They use constantly evolving very complex maneuvers to block the immune receptors, cellular signals and metabolic cascades that are used to fight virus infection.
Ebola virus has only 7 genes and proteins, but is able to build a very large complex vessel to travel between cells (see post). It uses many different attachment devices to enter each type of human cells. Ebola is able to fool cells into helping it enter. It is able to evade complex immune mechanisms with equally complex counter measures, while relying on this small number of proteins. Ebola is able to create a decoy to confuse the immune system and it commandeers complex protein rafts that are floating in the cell membrane to help it build the new viruses.
HIV was described in another post with only 9 genes and proteins. It travels with special proteins in its cover to evade immune reactions and enter cells. It attacks the most intelligent and complex T cells and immobilizes them with their few molecules. Inside the cell it evades immune reactions in very complex ways and then manipulates the nuclear pore and the genetic machinery.
 Herpes has more genes—70—but has a very complex lifestyle (see post). It travels up and down neurons and in and out of skin cells. It changes its behavior in the neuron and becomes quiet where it can sit for many years. Then it self activates and travels again.
Herpes has more genes—70—but has a very complex lifestyle (see post). It travels up and down neurons and in and out of skin cells. It changes its behavior in the neuron and becomes quiet where it can sit for many years. Then it self activates and travels again.
It is quite remarkable what these viruses can do with so few genes and proteins. It is quite fantastic that each protein can have multiple complex enzymatic funtions. It is not at all clear how they can do so much with so little. Now, Dengue virus is spreading and attacking with only 10 genes. It has been uniquely difficult to stop because of unusual reactions to antibodies.
Life Cycle
 Dengue is a single strand RNA virus that lives either in humans or in two specific mosquitoes, Aedes albopictus and aegypti, but is now the major virus spread through insects in the world. There are four sub types, all of which can cause severe illness. The current species developed in the past hundred years.
Dengue is a single strand RNA virus that lives either in humans or in two specific mosquitoes, Aedes albopictus and aegypti, but is now the major virus spread through insects in the world. There are four sub types, all of which can cause severe illness. The current species developed in the past hundred years.
Dengue virus has many different ways to attach to various cells. It uses a variety of human molecules to enter cells, such as multiple glycosaminoglycans, lactins, mannose receptors, and special immune proteins (immune modulatory proteins TIM and TAM receptors.). Using this many different ways of entry makes study very complex and the most common method is not yet known or fully understood. It is quite remarkable that it can have all of these methods with so few genes and proteins.
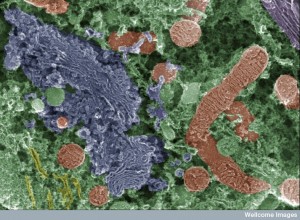
Dengue is able to enter critical immune cells such as mast cells (which secrete factors to help open blood vessels for travel) macrophages (which are supposed to eat them) and monocytes and dendritic cells (which are supposed to bring virus particles to T cells to make antibodies and stimulate attack cells). Dengue, also, enters liver cells and endothelial cells (see post on intelligent endothelial cells). They hijack all of these and change their behavior to protect the virus.
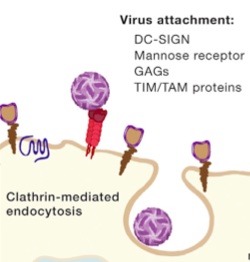 When the virus envelope (a vesicle sac in the form on an endosome) attaches to the membrane of the cell, it hijacks the complex clathrin mechanism to fuse with the cell’s membrane. This alters the sac properties through acidity, which induces the shape of the envelope (E) protein to change in order to aid the fusion of the two membranes.
When the virus envelope (a vesicle sac in the form on an endosome) attaches to the membrane of the cell, it hijacks the complex clathrin mechanism to fuse with the cell’s membrane. This alters the sac properties through acidity, which induces the shape of the envelope (E) protein to change in order to aid the fusion of the two membranes.
When the virus-protein particle is released into the cell, the RNA strand almost immediately makes one long protein consisting of all ten proteins. This large molecule is then cut into three structural proteins (C, E and M proteins) and seven non structural proteins (NS1, NS2A, NS2B, NS3, NS4A, NS4B and NS5) produced by the virus. The exact cutting process is not understood, but, it appears that it is somehow done by a human protease enzyme. This involves great cooperation of the human cell.
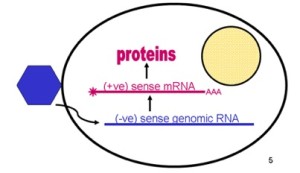 From the positive RNA strand (like a messenger RNA that can make proteins immediately) it makes a negative strand RNA and in turn from this negative strand manufactures many positive strands for the future viruses. This production occurs near cellular membranes, which creates an active working network.
From the positive RNA strand (like a messenger RNA that can make proteins immediately) it makes a negative strand RNA and in turn from this negative strand manufactures many positive strands for the future viruses. This production occurs near cellular membranes, which creates an active working network.
New positive RNAs are surrounded by a version of the E protein and pre membrane M protein to create an immature virus particle. Later, the E protein again changes its shape to form the more permanent and stable mature virus that leaves the cell. These changes occur with alterations in the three separate sub domain regions of the E protein. The immature virus particle has E proteins that take on a shape with two of them attached to prM (pre membrane protein). This complex of two Es and one prM forms a lattice with spikes sticking out all over the surface of the developing virus structure.
In the Golgi, special human proteases (furin proteases) are involved in the maturation of this structure. The maturation occurs when the E protein structures are altered so that they lie flat instead of the immature spike forms sticking out of the sphere. The pre membrane M protein is eliminated in this process. The smooth final surface resembles icosahedral or herringbone patterns and will leave the cell in membrane sacs by hijacking the exocytosis mechanism.
The changing shape of E proteins on the surface are a major problem for destroying the Dengue virus with antibodies, both in the immune reactions and the trials of vaccines. The smooth arrangement consists of antiparallel molecules of two E proteins in a herringbone pattern (a fish bone pattern).
There is much that is not known about Dengue virus’ complex behavior. Cutting of the prM protein is necessary for maturation of the virus to become infectious. But, many of these proteins are not cut exactly, which complicates antibody attack on the virus. These partially cut versions can, also, be infectious, but less so. As mentioned, partially effective antibodies start a process that actually increases the danger of the virus with strong secondary infections.
 When Dengue enters humans, the higher temperature allows even more variations and more antibody uncertainty. Some of these variations include rotated E proteins that allow the inner membrane to be exposed. This changes the effects of antibodies in unpredictable ways. The E proteins are quite flexible and variable in shape, which adds even greater complexity to its function and eradication. These changes alter the effects of different shaped antibodies in ways that are not really understood.
When Dengue enters humans, the higher temperature allows even more variations and more antibody uncertainty. Some of these variations include rotated E proteins that allow the inner membrane to be exposed. This changes the effects of antibodies in unpredictable ways. The E proteins are quite flexible and variable in shape, which adds even greater complexity to its function and eradication. These changes alter the effects of different shaped antibodies in ways that are not really understood.
Production of effective antibodies is a critical piece in conquering the viral illness. The three parts of the E protein make three different kinds of environments for the antibodies. Some of the E proteins will respond to some, but not to others.
 Dengue virus is able to trick the body’s factors that allow it to enter cells. It is taken in by endocytosis with its sac fused to the cell’s membrane. This is a complex process that is aided by the virus E protein (envelop protein). The viroid particle then travels to the endoplasmic reticulum where the RNA is copied and the various proteins are made in ribosomes. The virus is able to interact with the complex membranes of the ER for these processes. Antibodies attach to virus structures of one or two of the E proteins in several of the positions of the subunits. When the RNA is copied, it makes small virus particles that are surrounded by ER membrane buds.
Dengue virus is able to trick the body’s factors that allow it to enter cells. It is taken in by endocytosis with its sac fused to the cell’s membrane. This is a complex process that is aided by the virus E protein (envelop protein). The viroid particle then travels to the endoplasmic reticulum where the RNA is copied and the various proteins are made in ribosomes. The virus is able to interact with the complex membranes of the ER for these processes. Antibodies attach to virus structures of one or two of the E proteins in several of the positions of the subunits. When the RNA is copied, it makes small virus particles that are surrounded by ER membrane buds.
Cells have recognition receptors (TLRs) that notice and latch onto loose RNA particles. RNA, also, triggers special sensors that are used to detect abnormal genetic material in the cell—however, this pathway is not understood. One of these pathways uses a molecule sensor, which the Dengue virus protein NS2B/3 cuts and the NS5 destroys. This stops the immune fight against the virus.
Dengue Virus Structure
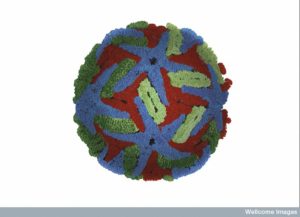
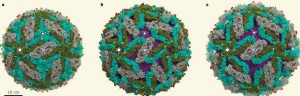
 The virus is spherical approximately 50nm is size. Several years ago, it was noted that the shape is altered by temperatures outside and inside of the human host. Three proteins make the structure—C (capsid protein), pre-membrane and membrane protein (prM/M proteins) and the envelope protein (E protein). An envelope sac made of lipids surrounds the protein structure. The strand of positive RNA is 10 kilo-bases long and is capped.
The virus is spherical approximately 50nm is size. Several years ago, it was noted that the shape is altered by temperatures outside and inside of the human host. Three proteins make the structure—C (capsid protein), pre-membrane and membrane protein (prM/M proteins) and the envelope protein (E protein). An envelope sac made of lipids surrounds the protein structure. The strand of positive RNA is 10 kilo-bases long and is capped.
Dengue is a positive sense virus meaning that it is equivalent to a messenger RNA, which can immediately make proteins with the host machinery. Negative sense RNA first must be copied into a messenger RNA, which makes the initial process more complex. But, to make more RNA strands for the final virus, a negative strand is necessary as the template of the positive strands.
E protein is made of three subunits in a row. It is critical for many steps in copying the RNA to make more viruses. E protein is involved in the attachment to cells and entry into the cell. It is critical for fusion of the virus envelope and the cell membrane. It, also, helps assembles the new virus.
The prM (pre membrane) protein helps the formation and maturation of the viral particle; its structure is seven antiparallel β-strands stabilized by three disulfide bonds
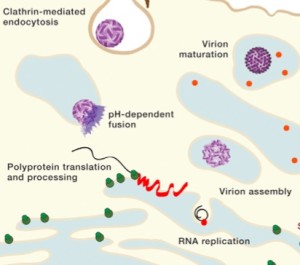 The virus shell is made of glycoproteins with 180 complexes in a pattern made of E and M proteins. First, it forms an immature structure out of E and prM (pre membrane protein), with 90 complexes in a herringbone pattern that makes spikes around the spherical shape. This immature version buds from a piece of the endoplasmic reticulum membrane and travels to the Golgi along the secretory pathway (see Complexity of Cellular Membrane post). In the Golgi, low pH changes the shape of the E protein that dissociates from the prM proteins making a flat pattern of just E proteins around the surface of the spherical final virus.
The virus shell is made of glycoproteins with 180 complexes in a pattern made of E and M proteins. First, it forms an immature structure out of E and prM (pre membrane protein), with 90 complexes in a herringbone pattern that makes spikes around the spherical shape. This immature version buds from a piece of the endoplasmic reticulum membrane and travels to the Golgi along the secretory pathway (see Complexity of Cellular Membrane post). In the Golgi, low pH changes the shape of the E protein that dissociates from the prM proteins making a flat pattern of just E proteins around the surface of the spherical final virus.
The pre membrane portion is cut from the M protein by the host furin protease enzyme. However, even the cut pr portion hangs on until the virus is released from the cell acting as a cap covering the fusion loop used for entry. The M protein sits under the E protein as part of the cover and membrane in the final product. It is remarkable the the pre membrane protein has so many completely different uses.
Non-Structural Proteins
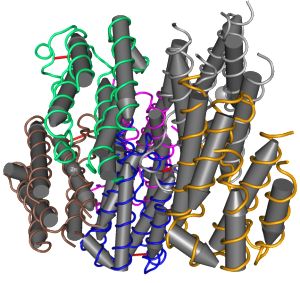
NS3 protein is an enzyme that cuts at the serine amino acid. It is, at the same time, an enzyme that remodels RNA (helicase) and provides multiple other enzymatic functions in the RNA transcription process. It is a very clever protein and has a complex structure to provide all of these functions with six β-strands arranged into two β-barrels formed by residues 1–180 of the protein. The most important section of the protein that catalyzes all of these reactions is between the two β-barrels.
 NS3’s function, also, needs NS2B, which serves as a co factor. NS2B wraps around the catalytic section and is active in the various critical enzyme functions. There are three other sections from amino acid 180 to 618 that are active as well. These are made up of different combinations of six-stranded parallel β-sheets, 2 antiparallel β-strands and α-helices.
NS3’s function, also, needs NS2B, which serves as a co factor. NS2B wraps around the catalytic section and is active in the various critical enzyme functions. There are three other sections from amino acid 180 to 618 that are active as well. These are made up of different combinations of six-stranded parallel β-sheets, 2 antiparallel β-strands and α-helices.
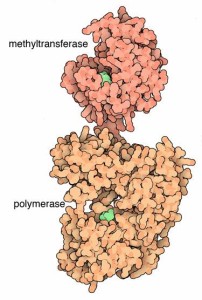
The NS5 protein has 900 peptide residues and functions as a methyltransferase on one end and a RNA-dependent RNA polymerase at the other. The methyltransferase active site includes a sandwich structure of sheets of α/β/β along with other sub units. It has special sections that are able to work with nucleotides.
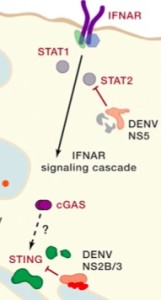
Nonstructural proteins allow the inhibition of various mediators of the innate immune system response. These proteins act on two levels. First, they inhibit inferferon. NS4B sits in the ER and stops phosphorylation of the critical immune factor STAT 1 from the interferon cascade.
NS5 inactivates STAT2, another critical part of the interferon cascade of anti viral activity. The complex process NS5 is cut with NS4B by a virus protease NS2B3. This damages STAT2. After the cut, E gets in the act and a section of E, E3, targets STAT2
NS2B3 has a special protease complex with a core of the last 40 amino acids of NS2B and the first 180 amino acids of NS3. Cutting the NS2B3 precursor triggers this protease complex, which inhibits production of type I interferon by triggering genetic networks and stopping the interferon promoter.
Evasion of Innate Immunity
A major immune reaction to viruses is triggered by interferon type 1 (IFN). Several cellular receptors trigger this reaction for Dengue after sensing RNA in a place where it shouldn’t be. The signaling cascade that occurs has many different molecules and triggers a large genetic network of hundreds of genes. These produce molecules that block the Dengue processes.
Somehow, Dengue, with its few proteins, is able to counteract these measures. Dengue is able to decrease type I IFN manufacture as a whole and stop particular signals that trigger important immune myeloid cells. Even more surprising is that the virus can alter specific ways that the immune cells present the antibodies (such as dendritic cells) so that the problems already mentioned with creating antibodies are increased. Dengue is able to specifically take advantage of the confusion relating to effects of antibodies.
The inhibition of the type I IFN is accomplished by a molecular structure consisting of two of their non-structural proteins NS2B and NS3. This creates a large enzyme that attacks specific proteins, one of which is the STING molecule that is critical in the cascade of interferon effects. It breaks down STING so that many other cascading molecules can’t work. This breakdown only occurs in human STING, not mouse. Stopping the effect of the cascade has multiple effects that are positive for Dengue.
 However another, entirely different mechanism occurs in this cascade by NS4B. It blocks another signaling molecule stopping phosphorylation. And yet another attack on the IFN cascade occurs by NS5.
However another, entirely different mechanism occurs in this cascade by NS4B. It blocks another signaling molecule stopping phosphorylation. And yet another attack on the IFN cascade occurs by NS5.
Therefore, three entirely different mechanisms fight this critical antiviral pathway with three of the seven non structural proteins.
Immune reactions to Dengue are not totally understood. When there are a large amount of antibodies produced, they attach to the virus and neutralize them. However, if there is a lesser concentration, they can do the opposite and actually help the virus to enter cells by triggering a specific receptor. (FcγR). Antibodies are generated against specific proteins made by the virus and these then increase a secondary infection with a more severe disease. In research, adding antibodies increased disease in animals. This same effect can occur from mother’s giving birth with more severe disease in the children.
Vaccines
There has been a great effort to develop a vaccine for this terrible virus, but Dengue presents unique challenges for this. Any vaccine of one of the sub types increases the danger of the others. The variable nature of the E proteins, also, adds to this danger. Current strategies in development are using four yellow fever vaccine viruses with four sub types of Dengue. Surprisingly it only helped people previously exposed. Another complex version had a 50% response. Another version is more promising with one study at 90%.
There are other possible treatments that attack mechanisms in the Dengue life cycle. But, limiting mosquitoes is probably most important currently.
 Infection produces antibodies for one particular sub type. But, as soon as the other subtypes activate, the first antibody effect increases the illness and possible hemorrhagic shock. The virus copying increases and is able to overcome macrophages trying to eat them. This is especially difficult when mothers who are infected give birth and pass on their particular insufficient antibodies. This increases the chance of severe shock in the babies.
Infection produces antibodies for one particular sub type. But, as soon as the other subtypes activate, the first antibody effect increases the illness and possible hemorrhagic shock. The virus copying increases and is able to overcome macrophages trying to eat them. This is especially difficult when mothers who are infected give birth and pass on their particular insufficient antibodies. This increases the chance of severe shock in the babies.
Dengue is able to alter innate immunity reactions by stopping specific receptors on white blood cells including mast cells. It increases IL-10 and alters the normal T cell reactions. It causes immune cells to secrete substances that alter the blood vessels and make them leaky. Another type of T cell attempts to continue to fight the infection.
Spread of the Intelligent Dengue Virus
 How can a small strand of RNA produce proteins to accomplish so many different complex maneuvers? Where is the direction for all of this?
How can a small strand of RNA produce proteins to accomplish so many different complex maneuvers? Where is the direction for all of this?
How do these proteins each perform many different complex actions? How do these proteins add new complex functions as the virus develops over time and fights the extremely elaborate innate immune system?
Dengue virus hijacks multiple different receptors for cell entry and it uses the host cell’s complex clathrin mechanism. Commandeering the machinery to make its proteins involves another dimension. How does the virus arrange several critical steps using enzymes from the host. How does it know to use its proteins to cut parts of the IFN cascade?
The extremely successful and complex function of a strand of RNA has to raise questions of where the guidance originates. Is this mind interacting with the molecules?