Are you with me or against me?
To determine an “other” we must know who we are. Recent research shows that the two systems that determine a sense of self, that is neurons and immune cells, work much more closely together than previously thought. Neurons use a rapid wired system of cells, whereas the immune system uses a wireless type of communication.
Self and Other
The sense of “I” is critical for every day functioning and appears to be related to a particular series of brain circuits, called the default mode network. As has been discussed i n previous posts (extraordinary experiences – body consciousness) the self is highly connected with bodily sensations, changing body maps, as well as narrative concerning family, professions, and strongly felt ideas. It is also closely associated with autobiographical memory including emotional ties to family, friends, and tribe.
n previous posts (extraordinary experiences – body consciousness) the self is highly connected with bodily sensations, changing body maps, as well as narrative concerning family, professions, and strongly felt ideas. It is also closely associated with autobiographical memory including emotional ties to family, friends, and tribe.
The sense of “other” is also closely associated to the body sense, with a culturally defined social space, often a foot or two around the body, inside which is considered personal space and outside of which is some “other” person. (post on body consciousness).
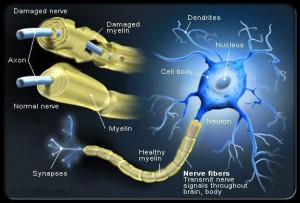
As well as the brain, the immune system is the other major system that has a clear sense of self and other. This sense of self is based upon familiar or unfamiliar molecules on the cells’ outer membranes. These notions of other can go awry evidenced by the immune system attacks on neurons in multiple sclerosis where neurons are believed to be aliens and are attacked.
A Colony of Colonies
Recently, the number of microbes in the human body was found to be 10 times  greater than the number of human cells. The amount of DNA making working factors is 100 times greater for microbe DNA in the human than human cells’ DNA. Other posts have described how critical microbe colonies are to the survival of the human being. A large percentage of the microbes, many trillions, are in the gut, but are also in all other areas.
greater than the number of human cells. The amount of DNA making working factors is 100 times greater for microbe DNA in the human than human cells’ DNA. Other posts have described how critical microbe colonies are to the survival of the human being. A large percentage of the microbes, many trillions, are in the gut, but are also in all other areas.
In the case of the trillions of microbes in humans, it is not so clear which are part of “self” and which are “other.” In fact, the immune system has an important love-hate relationship with the trillions of symbiotic microbes living in us. It has been shown recently that the immune system, while vigilant in the gut for destructive microbes, goes easy on most of the microbes that live in harmony and are vital to our life processes.
With ubiquitous microbes, it is difficult for the immune system to determine where self starts and stops.
Immune System and Nervous System
The immune system is designed to determine intruders and defend against them. But, it is not always certain who is an “other”. In the Golgi apparatus, for example, one type of future cancer cell that has polyploidy is destroyed. Other cells are not considered abnormal and are not eliminated even though they should be.
The two major immune systems, the quick innate immune response and the slow adaptive immune response, are both wireless systems. They are able to respond rapidly with cell-to-cell communications often through secreted signals.
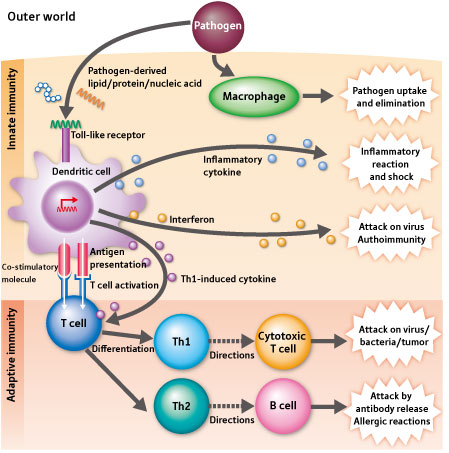 The nervous system is wired with more rapid communication. Both the immune and neuronal systems, in fact, work closely together. The high neuronal speed of communication allows rapid local and total system responses to danger, inflammation, and damage. The close connection of the neuronal and immune systems is another double-edged sword with dysfunctional immune responses damaging nerves in autoimmune and allergic diseases.
The nervous system is wired with more rapid communication. Both the immune and neuronal systems, in fact, work closely together. The high neuronal speed of communication allows rapid local and total system responses to danger, inflammation, and damage. The close connection of the neuronal and immune systems is another double-edged sword with dysfunctional immune responses damaging nerves in autoimmune and allergic diseases.
Nervous System Helping Immune
Historically, four responses to inflammation have been identified: pain, heat, redness, and swelling. But, until recently, only pain has been considered a nervous system response. Now it has been shown that the entire immune process is modulated by the nervous system.
The innate response initiates rapid local immune responses. It is the first line of defense. Local cells, or cells that can be rapidly mobilized to an infected region, provide a generic response to invaders. Unlike the adaptive immune response, these responses do confer a lasting effect. The innate response recruits cells by producing cytokine signals of infection. The elaborate “complement cascade” helps identify bacteria, activate immune cells and helps clearance of dead cells. It stimulates bringing necessary cells to the region, neutrophils, eosinophils, macrophages and T cells .
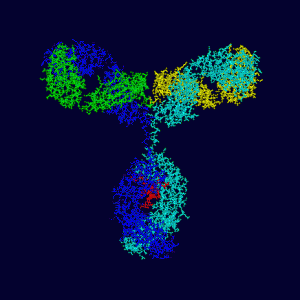
The adaptive immune system responds to specific chemicals usually on the surface of the bacteria, called antigens, and then creates an antibody to neutralize it, creating a longer lasting defense. The almost infinite number of antibodies that are created come from a form of DNA self editing. (see post DNA Proofreading)
Neurons Very Active in Immune Response
Neurons are now known to be extremely active in these processes. Neurons are able to induce mast and dendritic cells which fight the infections. Neuropeptides can actually function as an antibiotic.
 Specific signals from pain fibers influence many of the stages of immune response. Pain fibers have been shown to directly signal to white blood cells, change blood movement, and alter the general immune response. These sensory nerves behave in an unusual manner and send a signal along the nerve in the opposite direction than they usually do.
Specific signals from pain fibers influence many of the stages of immune response. Pain fibers have been shown to directly signal to white blood cells, change blood movement, and alter the general immune response. These sensory nerves behave in an unusual manner and send a signal along the nerve in the opposite direction than they usually do.
Avoidance of danger involves stimulation in the brain to move the body to safety, and remove contact with dangerous substances. In the skin, lungs, urinary and digestive tracts the nerves in the lining are the first responding to danger or invasion.
It is now known that trauma, toxins, microbes, and inflammation trigger many neuron receptors, including a wide variety of cytokine receptors and TRP channels. Calcitonin gene related peptide (CGRP) and substance P (as well as more than ten other other recently discovered neurotransmitters) act on the blood vessels, contributing to heat, swelling and redness. These neurotransmitters attract innate and adaptive immune cells locally. This effect can also be seen in arthritis where cutting the nerve will lower inflammation.
The signals from trauma, painful stimuli, and invading microbes also activate peripheral nerves.
A very unusual new finding is that lymphocytes secrete dopamine in a complex communication loop with neurons.
Immune Surveillance of the CNS
The relationship goes both ways. The immune system also directly influences the brain.
The brain must avoid immune responses that could damage the brain. So, immune surveillance of the brain is different from other regions. Microglia are brain cells and immune cells at the same time. Ordinary immune cells do monitor the brain from the blood vessels including macrophages, dendritic cells, and T cells. It used to be thought that brain does not have lymphatics, but recently a cleaning glymphatic system was discovered (glymphatic for glia plus lymphatic).
 The brain is sequestered by a large number of astrocytes forming a barrier, as well as the better known barrier between the blood and the cerebral spinal fluid. Microglia in the tissue and immune cells in the blood watch for any intruder. Microglia send signals to the neurons and larger glia cells, which determine a response. The intrusion of immune cells in the brain is very rare compared to other tissues, but a response can be vigorous as seen by meningitis.
The brain is sequestered by a large number of astrocytes forming a barrier, as well as the better known barrier between the blood and the cerebral spinal fluid. Microglia in the tissue and immune cells in the blood watch for any intruder. Microglia send signals to the neurons and larger glia cells, which determine a response. The intrusion of immune cells in the brain is very rare compared to other tissues, but a response can be vigorous as seen by meningitis.
Microglia are very important for maintaining synapses as well as the creation and pruning of synapses. They secrete many factors including the important factor BDNF, which stimulates new brain cells (discussed in prevous posts). The microglia lie close together and have processes that constantly move and evaluate every part of the extracellular space every several hours. A previous post showed how they tap each synapse each night to determine whether it should be pruned.
Normally the brain is very low in immune activity. But microglia can stimulate a rapid immune response and can also present antigens to the immune cells. They have machinery to identify all microbes and toxins. There are, however, some microbes that are able to establish residency in the brain such as herpes, and many others in immune compromised patients HIV. (see post on viruses).
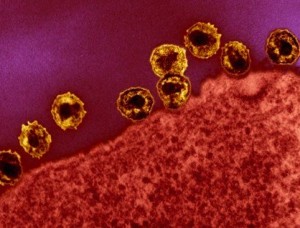 JC polyomavirus, and herpes are widespread in the brain, West Nile and HIV are occasional. But, these are all held in check by the microglia and by adaptive (antigen based) immune responses. There are also a small amount of T cells always in the brain. They only become active when the immune system is suppressed by other illness
JC polyomavirus, and herpes are widespread in the brain, West Nile and HIV are occasional. But, these are all held in check by the microglia and by adaptive (antigen based) immune responses. There are also a small amount of T cells always in the brain. They only become active when the immune system is suppressed by other illness
Another important relationship is related to rebuilding axons. Trauma in the periphery incites immune response that helps rebuild the axons. In the brain there is less reaction and inability to rebuild the axon. A recent study showed that modulating immune response to injury, by altering the activity of macrophage cells, can increase regeneration of damaged and severed axons.
Immune System Pruning Synapses
 One of the most vital of all brain functions is the creation and pruning of synapses. This process of new brain cells and new synapses is closely connected with our subjective experience, and all learning. It has been recently found that the intimate connection between the immune and nervous systems are critical to this synaptic process.
One of the most vital of all brain functions is the creation and pruning of synapses. This process of new brain cells and new synapses is closely connected with our subjective experience, and all learning. It has been recently found that the intimate connection between the immune and nervous systems are critical to this synaptic process.
Complement cascade, mentioned earlier as important in innate immune reactions, are also necessary for pruning synapses. In the immune system they work with antibodies to destroy invading microbes; in the brain they work to eliminate unnecessary synapses. The furious elimination of 900 billion neurons in the fetus is accompanied by a very high amount of complement proteins. After birth this complement declines, but increases again with Alzheimer’s.
 One major way the immune system defines self and other are through proteins called the Major Histocompatibility Complex, MHC. Some of these same proteins from the MHC are also produced in neurons. These molecules are part of the way neurons know where to attach to other neurons. It has become increasingly clear that the way neurons find their proper place in the developing brain, and the way axons find their appropriate place for a synapse involve surface molecules that communicate or adhere to nearby neurons called adhesion molecules. In fact, one important adhesion molecule in the developing brain is similar to the antibody immunoglobulin.
One major way the immune system defines self and other are through proteins called the Major Histocompatibility Complex, MHC. Some of these same proteins from the MHC are also produced in neurons. These molecules are part of the way neurons know where to attach to other neurons. It has become increasingly clear that the way neurons find their proper place in the developing brain, and the way axons find their appropriate place for a synapse involve surface molecules that communicate or adhere to nearby neurons called adhesion molecules. In fact, one important adhesion molecule in the developing brain is similar to the antibody immunoglobulin.
These important proteins on the surface of the cell bind with other MHC molecules on neurons. MHCs are also vital for forming synapses in the fetus. These are also necessary for learning later in life. Cytokines regulate MHC production in the brain and in immune cells, which is how cytokines can regulate memory and learning
Sickness Behavior
Immune system signals trigger many behaviors in human beings. One example is Il-6 (interleukin 6, an important immune cytokine signal) affecting the sense of hunger and fullness, and the ability to burn fat, causing weight loss.
 Importantly, a specific response from the immune system triggers a brain state called “sickness behavior.” This behavior includes sleepiness, fever, lack of interest in food, and increased sense of pain. This is triggered by viruses or bacteria causing lymphocytes to mobilize signaling with cytokines such as IL-1, IL-6. They in turn, activate the vagus nerve to send an electrical signal backwards to the brain (again in the opposite direction than usual). This reverse signal triggers specific neurons in the brain stimulating microglia, which send other signals causing the sickness feeling in the brain.
Importantly, a specific response from the immune system triggers a brain state called “sickness behavior.” This behavior includes sleepiness, fever, lack of interest in food, and increased sense of pain. This is triggered by viruses or bacteria causing lymphocytes to mobilize signaling with cytokines such as IL-1, IL-6. They in turn, activate the vagus nerve to send an electrical signal backwards to the brain (again in the opposite direction than usual). This reverse signal triggers specific neurons in the brain stimulating microglia, which send other signals causing the sickness feeling in the brain.
During health the same signals used in illness are used in learning and memory including immune memories for future reference. For example, recently it was found that after a first baby, adaptive immune responses maintain the knowledge that the next baby is also self, not other, so it won’t be attacked by the immune system.
The sick feeling is also closely related to depression, which increases IL-6 and other cytokines that help create the depressed feeling. Another system, the hypothalamic, pituitary, adrenal system, is involved in fight or flight responses and stress related responses through adrenaline and cortisol. These signals are also closely related to stress, which affects both neurons, and immune cells. Cortisol is known to suppress the immune system, and cortisol receptors in the hippocampus are closely related to the symptoms of depression.
In stress the brain signals through the peripheral sympathetic system (part of the autonomic nervous system discussed in previous post) into immune organs – to bone marrow thymus, spleen and lymph. Signals used by the immune system and on immune cells have the very same receptors as the neurons in the sympathetic system.
The Self and Other
As the constant interactions between the immune and nervous system are characterized, the details are becoming vastly more complex. In fact, it is very difficult to separate these two systems. The nervous system signals in all aspects of immunity and the immune functions are integral to all synapses. They work together constantly to define and defend the integrity of what is considered “self” among the vast  array of cells that make up the human being and the symbiotic microbial communities.
array of cells that make up the human being and the symbiotic microbial communities.
The immune system and the nervous system evolved together. One is wireless and slow and the other is wired and rapid. Crosstalk is essential for both especially during coordinated efforts in danger. The nervous system can help the immune reaction by signaling more rapidly. The immune system closely monitors the health of the brain.
Previous posts have observed how mind stimulates very complex molecular cascades in neurons. It is just now dawning on science how intimately these cascades are related to the immune system as well. Both the nervous and immune systems will be necessary to determine how we decide which is “self” and which is “other.”
