 The first line of cellular defense is not the specialized immune cells, but is the individual cell itself. The surprising abilities of individual cells, called cell autonomous immunity, are now becoming clearer when non immune cells also combat microbes.
The first line of cellular defense is not the specialized immune cells, but is the individual cell itself. The surprising abilities of individual cells, called cell autonomous immunity, are now becoming clearer when non immune cells also combat microbes.
In fact, individual cells in the brain have developed completely different personal immune responses to many different bacteria and viruses. Recently, a study showed that neurons in the cortex had completely different mechanisms than the granule cell in the cerebellum to one specific virus. Since each virus and bacteria that are able to penetrate the brain has different approaches to invasion of the neuron, the details become extremely complex and varied, showing great variability in responses. Much of survival turns out to be cellular intelligence versus invader intelligence.
Previous posts have demonstrated that for viral-like elements called jumping genes, moving from place to place in the genome, individual cells have developed their own version of an immune system. The genome fights back against the potential genetic influences of the jumping genes with microRNAs. So, it is not surprising that complex 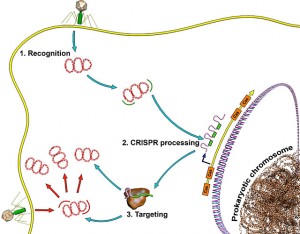 human cells have also developed mechanisms to isolate and fight bacterial and viral invaders, while they wait for the immune system to use adaptive and innate mechanisms.
human cells have also developed mechanisms to isolate and fight bacterial and viral invaders, while they wait for the immune system to use adaptive and innate mechanisms.
Individual bacterial cells have been shown to have quite advanced forms of immune responses to viruses and other bacteria. In fact, individual human cells also have an elaborate ability to defend against invading DNA from viruses. This CRISPR system (clustered regularly interspaced short palindromic repeats) recognizes specific sequences of DNA.
Immune Systems
 Most animals have innate immunity with specialized immune cells, and in larger animals, such as vertebrates, adaptive immunity using antibody production. But, all smaller creatures also have intrinsic self-defense by individual cells. Even the immune cells that are engaged in helping defend other cells utilize these types of defenses by individual cells, not another immune system. There are many examples of these individual cellular mechanisms, one being an enzyme used against bacteria for manufacturing nitric oxide. This mechanism works in insects and a similar pathway exists in humans. While this mechanism is very ancient, many creatures have very rapidly evolved other mechanisms against bacteria.
Most animals have innate immunity with specialized immune cells, and in larger animals, such as vertebrates, adaptive immunity using antibody production. But, all smaller creatures also have intrinsic self-defense by individual cells. Even the immune cells that are engaged in helping defend other cells utilize these types of defenses by individual cells, not another immune system. There are many examples of these individual cellular mechanisms, one being an enzyme used against bacteria for manufacturing nitric oxide. This mechanism works in insects and a similar pathway exists in humans. While this mechanism is very ancient, many creatures have very rapidly evolved other mechanisms against bacteria.
Previous posts have shown that the shape of proteins in the neuron are the language of the mind. In the cell’s quest to defend against invaders, the shape of compartments of the cell and the membranes between them become the most important factors in this defense. These compartments are, of course, determined by the shapes of scaffolding molecules.
Rooms in the Eukaryote Cell Mansion
 Eukaryote cells are very large compared with bacteria and have many different compartments, each surrounded by membranes and complex machinery. Compartments are a mixed blessing, a place for invaders to hide, and a place where they can be localized and killed based on the unique characteristics of that smaller region. Sometimes, by destroying one region, a cell can save itself.
Eukaryote cells are very large compared with bacteria and have many different compartments, each surrounded by membranes and complex machinery. Compartments are a mixed blessing, a place for invaders to hide, and a place where they can be localized and killed based on the unique characteristics of that smaller region. Sometimes, by destroying one region, a cell can save itself.
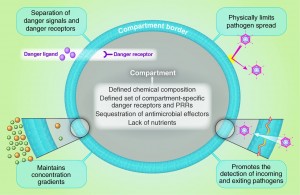
Bacteria must cross multiple different membranes to arrive at their favorite location in the cell. At each membrane they must have special mechanisms to fool the membrane guards. Each membrane has special receptors and other sensory devices to recognize an invasion using PAMP (pathogen associated molecular patterns), DAMPs (dangerous associated molecular patterns), and PPR (pattern recognition receptors.)
Also, on each membrane there are special molecules to stop an invader from multiplying. Another important mechanism measures the density gradients of the material and cells in a particular compartment compared with others. Knowing the gradient of compartments, allows cells to understand which are infected. Also, damage created by the invaders in a compartment contributes to the measurement of abnormal density gradients.
Enveloped viruses such as HIV, Rous sarcoma virus and herpes simplex all enter the cell by fusing with the outer membrane; cells use special antiviral proteins in the membrane to fight the fusion.
Non-enveloped viruses and some bacteria and fungi use the different vehicle of vesicles to enter the cell. Once in the cell these invaders must get out of the vesicle before they are transported to the compartment that is used to kill the contents of vesicles. The microbes have devised multiple ways to get out of the vesicle usually by secreting enzymes to open it. Pattern recognizing receptors then fight back by finding the pathogen inside the cell and they send LAP LC3 assisted phagocytosis. The cell also has special proteins that are stimulated by interferon that stabilize the vesicle membrane to stop it from breaking open.
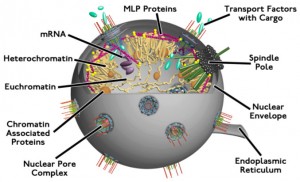 The nuclear envelope is a very elaborate enzyme structure protecting the DNA. Especially complex are the nuclear pores. But, the nuclear envelope is vulnerable during cellular division. Many viruses cannot get through the pores, but only invade the nucleus during cell division. HIV and herpes both hijack factors that usually import material into the nucleus, or specific proteins that make up the pores. The nuclear pore is very small 10 to 20 nanometers, and many viruses are 20 to 100 nanometers.
The nuclear envelope is a very elaborate enzyme structure protecting the DNA. Especially complex are the nuclear pores. But, the nuclear envelope is vulnerable during cellular division. Many viruses cannot get through the pores, but only invade the nucleus during cell division. HIV and herpes both hijack factors that usually import material into the nucleus, or specific proteins that make up the pores. The nuclear pore is very small 10 to 20 nanometers, and many viruses are 20 to 100 nanometers.
Getting out of a compartment is another issue for the virus. Viruses try to create vesicle buds at the membrane and the membrane has factors to defend against this.
Tetherin is a protein anchored in two places on the membrane and grabs and holds the virus by the viral envelope to the membrane as it forms buds in the membrane. Another molecule viperin breaks lipids from the virus used to manipulate the lipid membranes.
Outside the Cell
Cells use a variety of chemicals to deter pathogens from attacking outside of the cell. One is the well-known cleaner hydrogen peroxide that is synthesized specially near the exterior membrane. In the gut, lining cells manufacture a bactericide called RegIIIγ, which keeps clean a region near the lining of the small intestine. Bacteria rapidly colonize animals that don’t have this device.
But, the virus and bacteria do enter cells.
Molecules, such as glycans, normally inside of cellular vesicles called lysosomes and endosomes are released into the general compartment when a vesicle is destroyed.
 Galectins are a molecule that senses that there are loose glycans and something is wrong. This receptor molecule then stimulates an intra cellular mechanism for autophagy (controlled cell death, apoptosis) of bacteria. Plants have a mechanism of stimulating cell death in bacteria with no moving immune cells. Another aspect of these mechanisms includes stimulating a type of inflammation within the cell.
Galectins are a molecule that senses that there are loose glycans and something is wrong. This receptor molecule then stimulates an intra cellular mechanism for autophagy (controlled cell death, apoptosis) of bacteria. Plants have a mechanism of stimulating cell death in bacteria with no moving immune cells. Another aspect of these mechanisms includes stimulating a type of inflammation within the cell.
Another intracellular mechanism occurs if antibodies, which are being triggered outside of the cell by the larger organism, enter the cell. Serum antibodies are attached to the bacteria or non-enveloped virus and then brought into the cell with the pathogen. Once inside the cell and detecting these attached antibodies, the cell tags them with ubiquitin for destruction by a host of different enzymes. This mechanism is a link between the antibody response of the organism and the individual cellular response.
DAMP – Dangerous Associated Molecular Patterns
A stronger defense (DAMPs) doesn’t rely upon the specific structure of the microbe and therefore can be helpful with brand new invaders. When the microbes evade the PAMP defense (pathogen associated molecular patterns) they still must get through the nuclear pore. Those microbes that escape the phagocytosis and need to go through the nuclear pore are susceptible to the DAMP mechanisms.
Compartments in the Cell
 The bacterial internal immune system CRISPR destroys random DNA. This involves the bacteria determining which is the inherent DNA through markers, and then noting the new invading DNA. The new invading DNA is tagged by having an enzyme that adds a methyl group (a well known DNA marker in epigenetics) to the enzyme producing DNA.
The bacterial internal immune system CRISPR destroys random DNA. This involves the bacteria determining which is the inherent DNA through markers, and then noting the new invading DNA. The new invading DNA is tagged by having an enzyme that adds a methyl group (a well known DNA marker in epigenetics) to the enzyme producing DNA.
But, in the eukaryote cells, this process doesn’t occur. Instead, the DNA is very well protected by multiple mechanisms in the nucleus. And, therefore, whenever DNA is detected outside of the nucleus this is noted to be abnormal. Eukaryote cells have many different receptors and sensors to detect this aberrant DNA in places where it should not be. With all of the many defenses at different levels of entry and release, only a very small percent of viruses or other microbes actually succeed.
Certain components of the bacteria elicit strong reactions. One is the type III secretion system that has flagella and rods. Another is capsids (coverings) of viruses because their multiple loops are similar to antibodies. This process involves the loops in a process that makes a lattice out of the loops, undressing the virus prematurely.
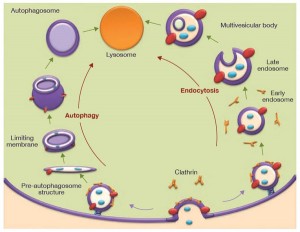
The Autophagosome – A Vesicle of Destruction
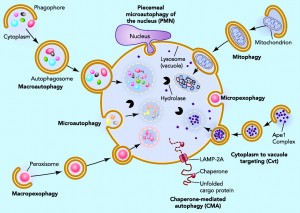 Another process builds a vesicle around the microbe –an autophagosome. This process uses crescent shaped phagophores that accumulate near the microbe building a structure around it. The ordinary autophagy (apoptosis) cleans defective cells and defective organelles, such as mitochondria. This cleaning system uses parts of cells, turning them into a defense by attacking vesicles containing microbes. This process occurs by first tagging the vesicle to be eaten with three different receptors. This mechanism is very effective against tuberculosis and salmonella. Microbes like listeria that naturally are in the cytoplasm, not in vesicles, are not defensed by these mechanisms.
Another process builds a vesicle around the microbe –an autophagosome. This process uses crescent shaped phagophores that accumulate near the microbe building a structure around it. The ordinary autophagy (apoptosis) cleans defective cells and defective organelles, such as mitochondria. This cleaning system uses parts of cells, turning them into a defense by attacking vesicles containing microbes. This process occurs by first tagging the vesicle to be eaten with three different receptors. This mechanism is very effective against tuberculosis and salmonella. Microbes like listeria that naturally are in the cytoplasm, not in vesicles, are not defensed by these mechanisms.
In some situations the bacterial secretion system is tagged, such as a system VII system, that has been described in previous posts as very similar to viruses. By eating some of the secretion system, the microbe’s DNA is released into the cell cytoplasm and then it is tagged and eaten. For viruses, capsids can be tagged and consumed. As some proteins are eaten, bacterial peptides (pieces of the proteins) are released and stimulate more autophagy activity. When cellular molecules are fighting the microbe these cellular components can trigger the autophagy as well.
Cellular Rooms for Virus to Find DNA Materials
Some cells have the components of DNA present inside of a cellular compartment—the smaller molecules that make up the DNA. This would make that compartment of the cell inviting to viruses because their own extra DNA is less likely to be noticed among the particles of DNA components. So, when a virus is present, specific processes eliminate these free-floating components of DNA such as NTPs (deoxynucleotide triphosphates). These NTPs otherwise would be available for the viruses to use to multiply. White blood cells secrete the enzymes that will be used by the cells to eliminate extra NTPs.
 Another tactic the cell uses is to get rid of extra amino acids, used to make proteins, which will also be used by viruses and bacteria reproducing themselves. An example of this mechanism includes specific enzymes that metabolize l-tryptophan, which defends against herpes virus and hepatitis B viruses, as well as bacteria like Chlamydia and Toxoplasmosis gondii, (previously described as stimulating suicidal ideation in humans and self destructive behavior in animals.)
Another tactic the cell uses is to get rid of extra amino acids, used to make proteins, which will also be used by viruses and bacteria reproducing themselves. An example of this mechanism includes specific enzymes that metabolize l-tryptophan, which defends against herpes virus and hepatitis B viruses, as well as bacteria like Chlamydia and Toxoplasmosis gondii, (previously described as stimulating suicidal ideation in humans and self destructive behavior in animals.)
In the phagolysosomes there are multiple mechanisms of microbe destruction that make this compartment particular dangerous and destructive to any microbe. They have pumps that eliminate essential metals from the microbe and create a very low pH (4.5) that kills microbes. Another mechanism is use of specific peptides that damage the microbe’s membrane. Special enzymes from the Golgi degrade other microbe materials. Reactive oxygen and nitrogen destroy membrane fats, DNA and proteins.
Unique Cilia Space
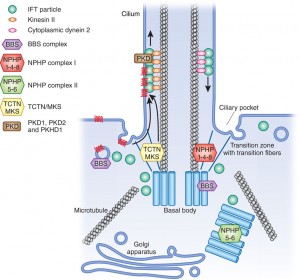 Cilia create a unique compartment with many important functions for the cell. These need to be protected from destruction by invaders. Cilia are critical antennae-like sensory devices for cells as well as motors for movement. They exist in a wide range of human cells and small organisms. They sense urine flow in kidney cells, light wavelengths in eye cells, pressure in cartilage, and blood flow in heart cells.
Cilia create a unique compartment with many important functions for the cell. These need to be protected from destruction by invaders. Cilia are critical antennae-like sensory devices for cells as well as motors for movement. They exist in a wide range of human cells and small organisms. They sense urine flow in kidney cells, light wavelengths in eye cells, pressure in cartilage, and blood flow in heart cells.
There is a wide range of sensory devices in the cilia that track forces and situations outside of the cell. They are critical in understanding and responding to the environment, by measuring mechanical and chemical forces and sending signals to other parts of the cell, or larger organism. The basic structure of a cilium is a tube of specific diameter, which limits how molecules can enter it. (cilia are one 1/10,000th the size of the cell). Recently, it was revealed that most of the proteins in the cell could fit into this tube. But, certain proteins stay in the tube and are anchored there. Like other compartments in the vast cell, the cilia has unique properties.
Multiple Barriers
These multiple defenses, none of them perfect, add up to very good protection for an individual cell, even without the organisms’ adaptive and innate immunity.
It is extremely difficult for a microbe to enter the tough outer membranes, then compartment membranes, then to avoid multiple enzymes and tagging systems, and finally to go through the very well protected nuclear pore. They also have to transport themselves between one compartment and another to arrive at the nuclear pore.
 But, once in the nucleus, there are more difficulties for the microbe. When the microbe is trying to place its DNA into the cell’s DNA, there are factors that will change the amino links in the virus DNA interrupting the process. Also, the processes that reduce the dNTPs needed by the virus are destroyed as in outer compartments. Also, in the nucleus the DNA repair system will attack the new microbe DNA.
But, once in the nucleus, there are more difficulties for the microbe. When the microbe is trying to place its DNA into the cell’s DNA, there are factors that will change the amino links in the virus DNA interrupting the process. Also, the processes that reduce the dNTPs needed by the virus are destroyed as in outer compartments. Also, in the nucleus the DNA repair system will attack the new microbe DNA.
Even if the DNA is placed into the cell’s DNA then there are repressors that will attempt to stop the DNA from making RNAs and proteins. Even after the proteins are made, there are mechanisms to block the assembly of the virus, and to block the membrane budding needed to get out.
Each microbe, therefore, needs a larger number of different unique mechanisms for each compartment of the cell, and adapted to different cells, and different animals.
A Very Intelligent Process In Neurons
In particulars, neurons have many unique mechanisms developed for many different kinds of RNA viruses.
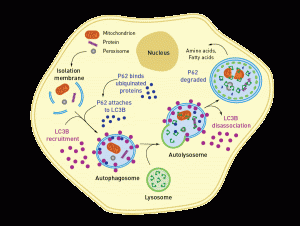 In neurons a specific set of genes direct the autophagy mechanism, and these direct tagging and cleaning of a variety of specific viruses. The Sidbus virus, for example, triggers clearing of this virus’s proteins, protecting against neuron cell death. A factor p62 targets capsid proteins of the virus for autophagosomes.
In neurons a specific set of genes direct the autophagy mechanism, and these direct tagging and cleaning of a variety of specific viruses. The Sidbus virus, for example, triggers clearing of this virus’s proteins, protecting against neuron cell death. A factor p62 targets capsid proteins of the virus for autophagosomes.
Recently, a study showed that different types of neurons, in this case from the cortex and cerebellum granule cells, have different mechanisms to counteract the same viruses, and therefore differential vulnerability to those viruses. Three genes are stimulated by interferon, (a factor produced by immune cells responding to an intrusion of an invader) and this triggers different approaches to fighting the viruses in the two kinds of brain cells. In these two different neurons there were also differences in epigenetic markings and different microRNAs involved in the process of fighting the virus. It appears that each of these neurons evolved different mechanisms to fight the same virus.
Non Immune Cells Also Combat Bacteria
 The eukaryote cell is, in fact, an enormous civilization with many buildings, rooms, pathways, transport systems, free living symbiotic bacteria called mitochondria travelling to where energy is needed, and elaborate machinery of many types with thousands of interlocking proteins. The nucleus is highly guarded at the entrance pores and the DNA is protected by elaborate large molecules that wind the DNA around spools and then cover it, making it very hard for invaders to find. Despite all of this, microbes find the DNA and manipulate it for their own purposes.
The eukaryote cell is, in fact, an enormous civilization with many buildings, rooms, pathways, transport systems, free living symbiotic bacteria called mitochondria travelling to where energy is needed, and elaborate machinery of many types with thousands of interlocking proteins. The nucleus is highly guarded at the entrance pores and the DNA is protected by elaborate large molecules that wind the DNA around spools and then cover it, making it very hard for invaders to find. Despite all of this, microbes find the DNA and manipulate it for their own purposes.
The cell is constantly being assaulted by invaders, and most are eliminated by specific mechanisms the cell has evolved for each different type of virus and bacteria. How can we not say that this enormously complex eukaryote cell defending against countless different microbes and strategies does not exhibit great intelligence?