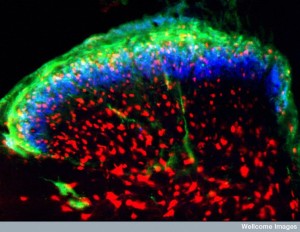
 Increasingly, physiological events are correlated with complex back and forth communication between a wide variety of local and distant cells. Previous posts have described a new concept of the neuro-immune interface, also called the neuro-immune synapse. Instead of the well known synapse of a pre and postsynaptic neuron (with a guiding astrocyte) the neuroimmune synapse includes signaling between a large number of different kinds of cells, such as neurons, astrocytes, microglia, T cells, endothelial (vessel lining) cells, macrophages and many other immune cells, that all participate in elaborate cross talk at one time. Not surprisingly, recent research shows that chronic pain is a result of conversation by immune and brain cells.
Increasingly, physiological events are correlated with complex back and forth communication between a wide variety of local and distant cells. Previous posts have described a new concept of the neuro-immune interface, also called the neuro-immune synapse. Instead of the well known synapse of a pre and postsynaptic neuron (with a guiding astrocyte) the neuroimmune synapse includes signaling between a large number of different kinds of cells, such as neurons, astrocytes, microglia, T cells, endothelial (vessel lining) cells, macrophages and many other immune cells, that all participate in elaborate cross talk at one time. Not surprisingly, recent research shows that chronic pain is a result of conversation by immune and brain cells.
Neuro Immune Signaling
 A recent post described the complex role of neurons in all aspects of inflammation, not just pain. Neurons can stimulate movement of immune cells, microglia, astrocytes, and alter the blood vessel cells through very complex communication. They can grade the stimulus to produce either full blown inflammation or a neuroplasticity of the circuit.
A recent post described the complex role of neurons in all aspects of inflammation, not just pain. Neurons can stimulate movement of immune cells, microglia, astrocytes, and alter the blood vessel cells through very complex communication. They can grade the stimulus to produce either full blown inflammation or a neuroplasticity of the circuit.
Another post described the elaborate signaling that occurs between the choroid plexus cells that line the cerebral spinal fluid, T cells that are allowed to enter the CSF and neurons. T cells, while traveling in the CSF, send wireless signals to the neurons to increase cognition. When they sense microbes and start an inflammatory response, T cells alter their signals to neurons in order to stimulate a change in cognition called “the sick feeling.” The choroid plexus cells can determine what types of inflammation are occurring throughout the brain (at long distance) and modify immune responses. All of this is done through back and forth communication with wireless signals.
 Many mechanisms produce pain. At first pain is useful, then it can becomes pathological. If a nerve is damaged the blood brain barrier can be altered and allow inflammation, immune cells, large molecules and fluids into the tissue. Neuron stimulated inflammation causes increases blood flow, extensive cytokine wireless signals and immune cell migration and can, also, cause more pain. The inflammation can spread to a larger region. Glial cells may not be able to keep up with buffering of glutamate released from dying cells, which causes more problems and more pain. This situation can become worse with a spread to generalized pain, spontaneous pain, radiating pain and chronic pain.
Many mechanisms produce pain. At first pain is useful, then it can becomes pathological. If a nerve is damaged the blood brain barrier can be altered and allow inflammation, immune cells, large molecules and fluids into the tissue. Neuron stimulated inflammation causes increases blood flow, extensive cytokine wireless signals and immune cell migration and can, also, cause more pain. The inflammation can spread to a larger region. Glial cells may not be able to keep up with buffering of glutamate released from dying cells, which causes more problems and more pain. This situation can become worse with a spread to generalized pain, spontaneous pain, radiating pain and chronic pain.
This post will discuss the signaling and cross talk at the brain higher levels that can cause chronic pain.
Various Causes of Different Kinds of Pain
Elaborate signaling between immune cells and brain cells is critical in development of chronic pain. Neurons can be excessively stimulated by well-known neurotransmitters from neurons, but, also, by lesser-known cytokines from microglia, astrocytes, and T cells.
 Acute pain has been part of the description of inflammation for a century, but only recently has it been demonstrated that the brain plays a part in all aspects of inflammation. In fact, the neuron uses the pathways of inflammation in neural stimulated inflammation as a form of neuroplasticity. But, also, chronic pain can come from this vast communication between neurons and immune cells, rather than local events in tissues.
Acute pain has been part of the description of inflammation for a century, but only recently has it been demonstrated that the brain plays a part in all aspects of inflammation. In fact, the neuron uses the pathways of inflammation in neural stimulated inflammation as a form of neuroplasticity. But, also, chronic pain can come from this vast communication between neurons and immune cells, rather than local events in tissues.
Hyperalgesia, an increased sensitivity to pain, along with the sick feeling, have been associated with immune response but the mechanisms were not known. Recently, the important cytokine interleukin 1B (IL-1b) was found to stimulate both hyperalgesia  caused by illness or without specific cause. This was the first demonstration that cytokines from the brain affect pain and behavior. Also, peripheral nerves, such as the vagus was shown to locally communicate with immune cells sideways from unmyelinated neurons.
caused by illness or without specific cause. This was the first demonstration that cytokines from the brain affect pain and behavior. Also, peripheral nerves, such as the vagus was shown to locally communicate with immune cells sideways from unmyelinated neurons.
Astrocytes and microglia in the spine were the first central cells found to signal with cytokines during nerve injuries. Signaling from these spinal glial cells cause Allodynia, that is, pain from a stimulus that usually doesn’t cause pain. Most recently, it was discovered that an inhibitor of another interleukin increased all types of experimental pain.
Brain Relays To Higher Centers
The two types of pain, nociceptive and inflammatory, are warnings to provide protection to an injury. Nociceptive receptors pick up stimuli at a higher harmful intensity. Inflammation causes pain through alterations in the tissues including toxins and excess fluids and cells.
 When sensory pain fibers are stimulated by increased pressure, temperature or chemicals, a neuronal signal is sent to higher brain regions. This is, then, relayed by a variety of neurotransmitters (glutamate, substance P, calcitonin gene related peptide or CGRP, AMPA, Kainate, GABA) to a second level of brain centers and to the spinal dorsal horn.
When sensory pain fibers are stimulated by increased pressure, temperature or chemicals, a neuronal signal is sent to higher brain regions. This is, then, relayed by a variety of neurotransmitters (glutamate, substance P, calcitonin gene related peptide or CGRP, AMPA, Kainate, GABA) to a second level of brain centers and to the spinal dorsal horn.
A third level of relay of the signals is to the higher cortex where the complex experience of pain is perceived. Top down signals from these higher signals using neurotransmitters serotonin and norepinephrine can modify the response, perception and experience of pain.
Some pain lasts longer than is useful to heal an injury—weeks, months and years. A substantial percent of people appear to have versions of this pathological pain.
One mechanism involves the first sensory neurons repeatedly firing, creating longer lasting non-useful pain. The center in the spine for this type of loop is the dorsal horn and a pathological firing there is called central sensitization.
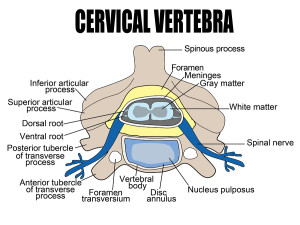 A form of neuroplasticity occurs at the dorsal horn involving glutamate NMDA receptors to facilitate this long lasting pain. Because of this, small sensory signals can produce a much larger pain that would be ordinary. Immune cell signals and activity from specific sensory nerves further amplify the sensitization.
A form of neuroplasticity occurs at the dorsal horn involving glutamate NMDA receptors to facilitate this long lasting pain. Because of this, small sensory signals can produce a much larger pain that would be ordinary. Immune cell signals and activity from specific sensory nerves further amplify the sensitization.
Up until very recently, central sensitization in the spinal cord was considered to be the cause of chronic or pathological pain. Now, it appears that all of the pain cannot be explained by this local spinal sensitization. Another cause is the cross talk at the neuro-immune synapses including the higher brain regions and local peripheral regions.
Back and Forth Signaling of Neurons and Immune Cells
 Previous posts have described extremely elaborate signaling that occurs between microglia, astrocytes, oligodendrocytes, endothelial cells, T cells in the CSF and other immune perivascular macrophages. These immune cells are not at all passive, but rather signal to and regulate neurons.
Previous posts have described extremely elaborate signaling that occurs between microglia, astrocytes, oligodendrocytes, endothelial cells, T cells in the CSF and other immune perivascular macrophages. These immune cells are not at all passive, but rather signal to and regulate neurons.
A post described the vital work of astrocytes in controlling all aspects of synapses and repair, including elaborate cytokine communication. Microglia have been described as the active immune cells patrolling and controlling the neuronal networks. They can alter their shape and function and numbers to produce a range of immune structures and signals. These are the most abundant cells in the brain. Some of these alterations are directly related to pain signaling. In particular they multiply in a process called gliosis, which leads to pain.
 Another post described the incredibly intelligent endothelial cells that signal with a wide variety of cytokines to microbes, neurons and immune cells. These cells in the choroid plexus are critical to stimulate T cells travel in the CSF. They allow immune cells to enter the brain when needed and modify neuronal signaling.
Another post described the incredibly intelligent endothelial cells that signal with a wide variety of cytokines to microbes, neurons and immune cells. These cells in the choroid plexus are critical to stimulate T cells travel in the CSF. They allow immune cells to enter the brain when needed and modify neuronal signaling.
The specific injury determines the types of cells that are involved in this complex neuronal-immune synapse. Different types of cells and signals are triggered in spinal cord injury and peripheral nerve injury. Neurons not only use the traditional neurotransmitters, but also, can signal with a large number of different cytokines. They can signal locally, sideways from un-myelinated neurons, or can signal from the brain at a distance.
Brain Signals to Immune in Pathological Pain
 When an injury occurs, microglia are the first to respond, while sending many complex signals. They alter their entire metabolism to a state where they rapidly multiply, called gliosis. Signaling occurs with immune cells that enter the damaged tissue locally (see post Complex Migration of Leukocytes). Astrocytes and T cells then respond with increases in numbers and another large set of signals. These many different signals are then secreted from the dorsal horn, the center of the local pain response.
When an injury occurs, microglia are the first to respond, while sending many complex signals. They alter their entire metabolism to a state where they rapidly multiply, called gliosis. Signaling occurs with immune cells that enter the damaged tissue locally (see post Complex Migration of Leukocytes). Astrocytes and T cells then respond with increases in numbers and another large set of signals. These many different signals are then secreted from the dorsal horn, the center of the local pain response.
Neurons take up the cause and, also, secrete more cytokines, and chemokines (that attract traveling immune cells). Injured neurons secrete signals that attract local microglia and T cells. These neuronal signals increase the production of the inflammatory microglia. This increase in the number of microglia is correlated with a stimuli producing pain that normally doesn’t (allodynia,) and with neuropathic pain.
Damaged neurons also release extracellular matrix molecules that increase sensitivity to pain and neuropathic pain. These ECM molecules then trigger other neurons as if they were microbes. In one particular example in the trigeminal nerve, a series of chemicals are secreted from neurons and immune cells that alter the sensitivity of the nerve causing continuing pain.
 This vast number of different signals causes an equally wide variety of signaling cascades inside of cells. These cascades produce factors such as NF-kb (nuclear factor-kb) that triggers large genetic networks inside cells promoting inflammation. Many of these signaling cascades increase the production of the mass of microglia that are involved in producing pain.
This vast number of different signals causes an equally wide variety of signaling cascades inside of cells. These cascades produce factors such as NF-kb (nuclear factor-kb) that triggers large genetic networks inside cells promoting inflammation. Many of these signaling cascades increase the production of the mass of microglia that are involved in producing pain.
Chronic Pain
There are many ways that cytokines produce pain.
- It was mentioned above that immune cells and microglia multiply while producing a large number of cytokine signals, produce pain. This type of signaling can continue on for months producing chronic pain.
- Another mechanism was found recently where neurons are directly stimulated by a microbe cytokine to produce lasting pain.
 Signals from immune cells and glia trigger receptors on both pre and post synaptic neurons in the dorsal horn causing changes in the sensitivity to pain. Unlike traditional neurotransmitters, these affects can occur with a small amount of cytokines.
Signals from immune cells and glia trigger receptors on both pre and post synaptic neurons in the dorsal horn causing changes in the sensitivity to pain. Unlike traditional neurotransmitters, these affects can occur with a small amount of cytokines.- Another example involves multiple immune cytokines directly influencing glutamate excitatory signals. This type of neuronal influence on increased pain involves gliosis of astrocytes, not microglia. These glutamate signals are increased by astrocytes affecting the molecules that take up glutamate after secretion. This occurs after peripheral nerve injury and causes pain hypersensitivity.
Very Complex Language of Chronic Pain
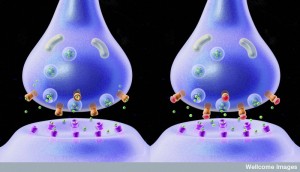 There are too many factors to list here. And many different factors are used in different brain regions. To give a flavor of the complexity, the following is a list of cytokines and receptors a neuro immune synapse consisting of a presynaptic neuron, post synaptic neuron, T cell, microglia, inhibitory neuron, and astrocyte.
There are too many factors to list here. And many different factors are used in different brain regions. To give a flavor of the complexity, the following is a list of cytokines and receptors a neuro immune synapse consisting of a presynaptic neuron, post synaptic neuron, T cell, microglia, inhibitory neuron, and astrocyte.
- Presynaptic Neuron: glutamate, CCL2, IFNg, IL-1B, ROS, TNF, receptors TRPV1, TRPA1, IL-1R1, NMDAR
- Astrocyte: IL-1b, TNF, EEAT1, EAAT2, CXCL1, IL-6,
- Inhibitory Neuron: IL-1B, IL-6, ROS, TNF, GABA, Glycine,
- T Cell and Microglia: BDNF, CCL2, IFNg, IL-1B, IL-6, IL-17, PGE2, ROS, TNF
- Post synaptic neuron: TRKB, BDNF, KCC2, IL-1R1, GRK2, PGE2, PKA, ERK, CREB, EP2, GABAR, GlyR3, IL-1B, NR2A, NR2B, NR1, NMDAR, IL-17, ROS, CCL2, AMPAR, TNFR1, CXCL1, CXCR2, CCR2
Multiple Layers of Regulation
There are, also, many inhibitory systems that regulate these pain conversations. It is strange that when there is more spinal cord inhibition, chronic pain can occur through sensitization. In the list above for the neuro immune synapse of the six cells, many are different types of inhibitory pathways causing dis-inhibition. When this dis inhibition occurs then even sensory input from innocent sources cause pain.
 When a nerve is damaged, a huge regulatory network attempts to control the central signaling of the neuron-immune synapse. Special immune cells are triggered that can fight against inflammation. Anti-inflammatory factors are secreted into the synapse. When this network is not operating correctly, chronic pain can result.
When a nerve is damaged, a huge regulatory network attempts to control the central signaling of the neuron-immune synapse. Special immune cells are triggered that can fight against inflammation. Anti-inflammatory factors are secreted into the synapse. When this network is not operating correctly, chronic pain can result.
To fight inflammation, both macrophages and microglia, change their shape into the M2 form. T helper cells and T regulatory cells are activated to fight hypersensitivity after a nerve injury. Many of these cells secrete the powerful anti-inflammatory cytokines IL-1RRA, IL-4 and IL-10. These cells can diffuse the gliosis of microglia that cause chronic pain. Another set of signals comes from enzymes that alter the extra cellular matrix. All of these different mechanisms fight against the central sensitization causing pain.
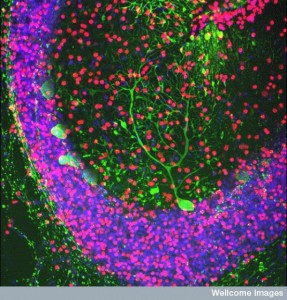 While these mechanisms fight inflammation, it is necessary to have a rapid and effective immune response to fix the damage, eliminate microbes, clear the gliosis and the debris. TNF seems to be critical in this process that fixes nerves in the brain, while TNF and IL1B help in the peripheral nerves. Many other factors are, also, involved.
While these mechanisms fight inflammation, it is necessary to have a rapid and effective immune response to fix the damage, eliminate microbes, clear the gliosis and the debris. TNF seems to be critical in this process that fixes nerves in the brain, while TNF and IL1B help in the peripheral nerves. Many other factors are, also, involved.
When the damage is fixed, microglia are critical in cleaning up debris and eliminating microbial molecules that can trigger further inflammation. They also secrete factors or place them on their membranes that stop other immune cells from continuing to come to the region. This process also can be altered and contribute to chronic pain.
Medications Targeting the Neuro Immune Synapse
This research implies that medications for chronic pain could target these many mechanisms including the factors increasing inflammation from local factors and distant cytokine signals. Research has demonstrated (in animals) specific cytokines targets, such as TNF and IL-1Ra. But, unfortunately, these have wide ranging effects that could cause other problems if used as medications. Right now, the interplay between all of the factors is just too complex for new medications.
 Some already approved medicines have effects in the complex neuro-immune synapse: minocycline, methotrexate. But, the results have been inconsistent probably because of the thousands of different mechanisms in the conversation between neurons and immune cells. There is a wide range of experimental agents being studied, but many have multiple effects, not a single helpful effect. One class includes molecules that effect lipids in the extra cellular matrix and help cleanup including resolvins, protectins, and lipoxins.
Some already approved medicines have effects in the complex neuro-immune synapse: minocycline, methotrexate. But, the results have been inconsistent probably because of the thousands of different mechanisms in the conversation between neurons and immune cells. There is a wide range of experimental agents being studied, but many have multiple effects, not a single helpful effect. One class includes molecules that effect lipids in the extra cellular matrix and help cleanup including resolvins, protectins, and lipoxins.
New research uses DNA to produce cytokines against inflammation. Gene therapies in animal research have involved IL-4 and IL-10. However, it has not been proven that animal models are identical to humans.
Current research has proven that complex cytokine factors are critical in some specific pain conditions. Astrocytes in the spinal cord have stimulated chronic pain with HIV. A regional pain syndrome involves gliosis of astrocytes and microglia. Specific mechanisms of neuropathic pain have been demonstrated in ALS and multiple sclerosis. Also, depression, drug abuse and anxiety all have specific central mechanisms of chronic pain from the central system of neural immune synapse.
Opiods
 Opiod medications are used for pain of all types, but in chronic pain it leads to addiction and a paradoxical symptom of increased pain called hyperalgesia. The central conversation of neurons and immune cells now has been shown to explain some of these properties. Morphine signals at the central neural immune interface, especially with the signal TLR4—Toll like receptor 4. This factor in communication with immune cells seems to be involved in reward, craving, withdrawal and induction of the paradoxical increased central pain. Other studies show opiod receptors on microglia including BDNF contribute to hyperalgesia.
Opiod medications are used for pain of all types, but in chronic pain it leads to addiction and a paradoxical symptom of increased pain called hyperalgesia. The central conversation of neurons and immune cells now has been shown to explain some of these properties. Morphine signals at the central neural immune interface, especially with the signal TLR4—Toll like receptor 4. This factor in communication with immune cells seems to be involved in reward, craving, withdrawal and induction of the paradoxical increased central pain. Other studies show opiod receptors on microglia including BDNF contribute to hyperalgesia.
Chronic Pain is A Result of Conversation By Immune and Brain Cells
 The conversation between neurons of all types and at all levels, with the many immune and vessel lining cells is called the neural-immune synapse. Chronic pain of many types arises from this complex conversation in the molecular language of factors, cytokines and neurotransmitters. Along with neurons, key players include microglia, astroctytes, oligodendrocytes, T Cells, macrophages and extra cellular matrix.
The conversation between neurons of all types and at all levels, with the many immune and vessel lining cells is called the neural-immune synapse. Chronic pain of many types arises from this complex conversation in the molecular language of factors, cytokines and neurotransmitters. Along with neurons, key players include microglia, astroctytes, oligodendrocytes, T Cells, macrophages and extra cellular matrix.
Individual cells and communities of cells engage in elaborate back and forth cross talk with wireless cytokine signals, determining brain and immune function.