 Natural cannabinoids in the brain, related to those in the marijuana plant, have been described in previous posts as critical for many important brain functions, both in the fetus and the adult. Recent research now shows that they have a great importance in regulating brain waves and circuit hyperactivity that can produce seizures. In fact, several factors make them uniquely able to respond in distinct places and times to control aberrant electrical activity. The manufacture and elimination of cannabinoids and their receptors is very tightly controlled in specific places in the brain and in very specific time scales.
Natural cannabinoids in the brain, related to those in the marijuana plant, have been described in previous posts as critical for many important brain functions, both in the fetus and the adult. Recent research now shows that they have a great importance in regulating brain waves and circuit hyperactivity that can produce seizures. In fact, several factors make them uniquely able to respond in distinct places and times to control aberrant electrical activity. The manufacture and elimination of cannabinoids and their receptors is very tightly controlled in specific places in the brain and in very specific time scales.
Most other neurotransmitters are manufactured and placed in vesicles in particular circuits. Cannabinoids and receptors are triggered as needed in many places in the brain, responding to signals from glutamate, GABA, epinephrine, and dopamine. This production of cannabinoids can occur in an as needed basis, which makes them ideal for controlling hyperactivity in the brain, such as seizures.
Cannabinoids are manufactured in the post-synaptic neuron and travel backwards to the pre -synaptic neuron, unlike most neurotransmitters. There they trigger the CB receptors to stop the release of other neurotransmitters. In this way they regulate many aspects of brain function. They have been found to have critical regulatory functions. One of the important emerging functions is to regulate groups of neurons with synchronous oscillations known as brain waves. Because of this, they have become relevant to the discussion of seizure treatment.
This post will first summarize two previous posts on cannabinoids—Endocannabinoids Critical for Brain Function and Cannabinoids in Inflammation and the Aging Brain. Please refer to these for much more detailed descriptions of the many cannabinoid brain functions. This post will discuss the new vital information related to cannabinoids and seizures.
Previous Descriptions of Cannabinoid Functions
This is a summary of salient facts from the previous post about the cannabinoid system in the developing fetal brain and in adults from the two previous posts.
 In the Fetus
In the Fetus
Cannabinoids are vital for every aspect of the creation of the structure of the fetal brain. Cannabinoids:
- direct neuron migration
- stimulate axons and dendrite development.
- determine new neurons and glia from stem cells
- determine neuron polarity for migration and axon production
- control the formation of the synapses in the cortex.
- build the pattern of neuronal connections.
- build the basic sensory long tracts between the cortex and the thalamus.
- critical for embryo implantation and continued survival of the fetus.
- After stem cells make more neurons, widespread cannabinoid signaling throughout the region stimulates production of many glia cells.
- Another critical fetal structure built by cannabinoids is the basic sensory long tracts between the cortex and the thalamus.
- Cannabinoid lipid signaling is critical for making new astrocytes and oligodendrocytes.
 Cannabinoids in Adults.
Cannabinoids in Adults.
- Cannabinoid signals are important for pain sensation, metabolic systems, appetite, memory, motor learning, neuroplasticity and mood.
- They regulate hunger and interact with the amount of leptin in the blood.
- They bring into balance storage and transport of nutrients and energy storage in fat, intestinal, muscle and pancreas cells.
- Cannabinoids are involved in the complex regulation of insulin, the cortisol stress response and the hypothalamus-pituitary- adrenal system.
- In the immune system, cannabinoids modulate both neurons and immune cells
- Cannabinoids are involved in modulating pain sensation.
- Cannabinoids have different functions in the adult brain and therefore affects of ingested plant cannabinoids are very different in the established adult brain.
- Cannabinoids stimulate the creation of new neurons in the adult brain, in the hippocampus, the sub-ventricular and olfactory zones.
- Cannabinoid stimulation of new neurons by stem cells occurs more powerfully in the elderly brain.
- 2-AG is the main cannabinoid stimulating new neurons in adults. Blocking it decreases new neurons production.
- Cannabinoids maintain neuroplasticity and keep stem cells ready to make new neurons.
- The system responds rapidly to injury, degenerative brain disease, inflammatory brain disease and depression.
- Cannabinoids provide stimulus for new neurons and directions for their travel in the brain.
- In animals, cannabinoids have stopped cancers of glia cells through stimulation of cell death of tumor cells without hurting normal cells.
 It is not clear how cannabinoids can stimulate the production of new neurons and glia stem cells, but kill glia tumor cells.
It is not clear how cannabinoids can stimulate the production of new neurons and glia stem cells, but kill glia tumor cells.- A new cannabinoid treatment for multiple sclerosis was just approved. This is the first treatment of the fundamental cause of a major degenerative disease, rather than a symptom.
- THC has been used previously for vomiting and weight loss in cancer and AIDS patients.
- Cannabinoids are involved in the cortisol stress response and the hypothalamus-pituitary-adrenal system.
- They stimulate rapid response to brain injury by triggering some of the processes that originally built the brain.
- Cannabinoids are altered in aging and are deeply involved in brain inflammation and degenerative brain diseases.
Lipids and Creation of Cannabinoids
Cannabinoids are signaling molecules in the brain that are made from lipids cut from the neuronal membrane. Previous posts described the critical process of lipids in the brain including omega 3 and 6 molecules.
Lipids have been discussed in previous posts on the complexity of membranes, and the signaling molecules of lipoproteins. A previous post described the extremely elaborate system of fatty acid derivatives that are critical signaling molecules. Lipids, or fats, are most of the weight of the brain. Lipids are signaling molecules like nucleic acids, amino acids, RNAs and proteins. The CB cannabinoid receptor is one of the most common receptors in the brain that uses the mechanism called G proteins, along with many other neurotransmitters.
Two specific fatty acids, DHA and ARA, attach to phospholipids for storage and await release by special enzymes. Along with the well-known derivative of ARA, prostaglandin, perhaps, the most important derivatives of ARA and DHA are cannabinoids that are critical for neuron and glia function.
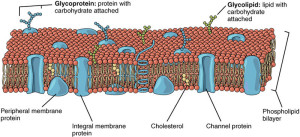
Most often, cannabinoids travel from the postsynaptic neuron to the pre synaptic in retrograde fashion and regulate processes in the synapse. Cannabinoids and their receptors are manufactured by neurons, astrocytes and microglia. These bind to the CB1 and CB2 receptors, which regulate release of glutamate, GABA, monoamines, opioids and acetylcholine.
Neurotransmitters dopamine, acetylcholine, serotonin and?glutamate NMDA stimulate enzymes that cut ARA and DHA ?from their attachment to membrane lipoproteins in neurons? and glia. It is stimulated ?whenever these molecules are needed, such as in brain? development, memory in adults, ischemia, brain injury and inflammation.
Brain Waves and Seizures
To avoid seizures, the inhibitory GABA interneuron network is crucial to regulation of circuits. The cannabinoids are critical in regulating these GABA neurons. Recent research shows that neurons are constantly involved in dynamic oscillations.
Specific brain wave frequencies (alpha, beta and gamma) are found to have very distinct functions in the brain communicating either locally or long distances. Each area produces specific rhythms. The hippocampus, (the critical center of memory and learning) uses the slow theta (5 to 10 herz – per second) for learning and when at rest the fast 120 to 200 Hz sharp waves. These long range sharp waves are important for the consolidation of memory, that is the communication of the short term hippocampal memory to a longer term storage of the recent new memories.
All regions appear to have significant and particular oscillations that communicate with other parts of the brain wirelessly, along with the wired synaptic connections. Critical oscillations occur between the cortex, thalamus and striatum.
The cannabinoid system modulates all of this wireless communication. Many brain diseases have abnormal rhythms, prominently seizure disorders. But, other diseases with significant altered rhythms are Parkinson’s, Alzheimer’s, and depression. In seizures there are high frequency spikes as well as abnormalities in between the seizures. It is possible that specific regions of cannabinoid receptors are critical for these diseases and could be a path to treatment.
Specificity of Cannabinoids and Receptors
There are a variety of ways that cannabinoids operate in very particular places and timing. They are surgical and powerful in very precise effects that control very small regions of hyperactivity. They operate on very specific synapses and in many specific time frames.
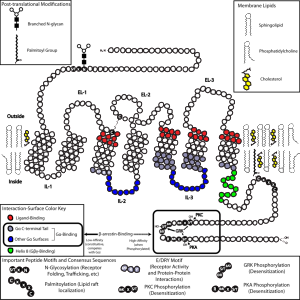
Cannabinoid receptors operate through the system of G proteins that connect in the membrane to a cascade of molecules triggering genetic networks. G proteins are a large family of huge receptor molecules that are triggered by molecules outside the membrane and then signal a cascade inside the neuron to the nucleus activating genetic networks.
Cannabinoid CB1 receptors are widespread in brain, especially the hippocampus. CB2 receptors are mostly on immune cells. CB1 target interneurons especially. Although there are a large numbers of receptors, they are very specific. An example is that of the 19 types GABA interneurons in one small region of the hippocampus, only one has CB1 (CCK). CCK type interneurons, also, have subtypes. They are not on any parvalbumin PV neurons in the cortex. PV neurons produce many important rhythms theta, gamma and ripple.
But, in other regions the specificity is different. In the basal ganglia, PV neurons use CB1. Also, there are great differences based on specific connections. Anandamide is a partial agonist of CB1 and 2-AG a full agonist. 2-AG is the major signal that goes backwards at synapses. A major class of glutamate receptors are involved in stimulating the production of more cannabinoid receptors in excitatory synapses in the cortex. Many important neurotransmitters trigger more 2-AG—acetylcholine, oxytocin, vasopressin, norepinephrine and 15 others.
2-AG is metabolized into arachidonic acid and glycerol by astrocytes and neurons. This maintains a very specific use of the cannabinoid since it doesn’t spread as some other neurotransmitters do. In fact, the use of the cannabinoids are very regulated and limited to very specific areas and very specific timing. These cannabinoids regulate most other types of synaptic signals. This regulation occurs even very far from where the 2-AG is being manufactured.
How Cannabinoids Control Other Neurotransmitters
This control occurs in three major ways—phasic, tonic and with long-term depression.
 One is a transient effect, (phasic) where the cannabinoid signal temporarily stops the other signal. This occurs through triggering increased calcium or through G protein pathways. In particular this depression of activity is highly related to the ability of neurons to form brain waves. What is unusual about this form of signaling is that there are no vesicles, but rather more 2-AG is formed as needed. This type of synthesis particularly occurs when there is spiking of activity.
One is a transient effect, (phasic) where the cannabinoid signal temporarily stops the other signal. This occurs through triggering increased calcium or through G protein pathways. In particular this depression of activity is highly related to the ability of neurons to form brain waves. What is unusual about this form of signaling is that there are no vesicles, but rather more 2-AG is formed as needed. This type of synthesis particularly occurs when there is spiking of activity.
As mentioned above, 2-AG is rapidly eliminated and the spiking starts again in seconds. The timing of this is based on the extent of the activity. Also, this depression of activity is related to the particular frequencies. This mechanism is particularly relevant when there is increased sudden activity, such as in seizures.
The second type of control (tonic) occurs through the level of baseline cannabinoid activity that is maintained. This activity can silence synapses completely for period of time. If the CB1 is blocked, GABA inhibitory synapses revert to full signaling. This effect is not related to any extracellular cannabinoids, but rather a very specific and targeted effect. Also, this inhibition works only with very particular neuron subtypes (CCKBC but not CCKSCAC).
 Another form of depression is not the episodic or tonic, but rather a lasting long-term depression neuroplastic change in the synapse. If there is repetitive electrical activity, then cannabinoids are involved in altering the synapse in a long lasting way through 2-AG. This requires increased calcium and specific neurotransmitter activity, such as with NMDA and dopamine2. There are very specific pathways (calcineurin) and specific types of vesicles carrying neurotransmitters (RAB3) also involved. This mechanism produces lasting decrease in neurotransmitter signaling affecting the excitation.
Another form of depression is not the episodic or tonic, but rather a lasting long-term depression neuroplastic change in the synapse. If there is repetitive electrical activity, then cannabinoids are involved in altering the synapse in a long lasting way through 2-AG. This requires increased calcium and specific neurotransmitter activity, such as with NMDA and dopamine2. There are very specific pathways (calcineurin) and specific types of vesicles carrying neurotransmitters (RAB3) also involved. This mechanism produces lasting decrease in neurotransmitter signaling affecting the excitation.
Regulation of Brain Waves
Cannabinoid receptors have complex effects on brain waves, both inhibitory and stimulating. Sometimes, they do both at the same time. The neurotransmitter acetylcholine is critical to theta and nested gamma oscillations in the hippocampus. These can increase or decrease GABA cannabinoid activity. The type of neuronal electrical activity then determines which effect wins out.
Cannabinoids, also, have a more direct effect on the oscillations. Neurons that do not use cannabinoids stimulate gamma rhythms. Cannabinoids stimulates another neuron that stops these rhythms. CB1 effects through GABA inhibitory neurons have effects on cognition described in previous posts—memory effects and protection of aging effects. Dysfunction in these processes are involved in cognitive symptoms in several diseases.
Regulation of Epilepsy by Cannabinoids
 Stress affects both the cannabinoid system and epilepsy. These alterations in the cannabinoid signaling are very specific as mentioned above. They are specific to particular types of neurons. In research of seizures caused by fever, everse signaling of cannabinoids increased by phasic short term plasticity. Another type of seizure research shows tonic inhibition. These changes occur in the basal ganglia in Huntington’s and the striatum and hippocampus in a model of fragile X syndrome, both of which can have seizures.
Stress affects both the cannabinoid system and epilepsy. These alterations in the cannabinoid signaling are very specific as mentioned above. They are specific to particular types of neurons. In research of seizures caused by fever, everse signaling of cannabinoids increased by phasic short term plasticity. Another type of seizure research shows tonic inhibition. These changes occur in the basal ganglia in Huntington’s and the striatum and hippocampus in a model of fragile X syndrome, both of which can have seizures.
Specific timing of cannabinoid effects occurs with different alterations before, during and after the seizures. Because of these differences, treatment of seizures by cannabinoids is going to be complex.
Patients with epilepsy have specific neuronal changes in the hippocampus related to CB1. These impair control of oscillations in epilepsy. There is both depression of activity and then increase of active in different neurons in the dentate nucleus—increase of GABA and decrease of glutamate.
Decreasing Circuit Activity by Cannabinoids
There are many different kinds of epilepsy, some caused by genetic alterations of sodium channels and some by damage to the brain. Not all of them can be studied, since it is unethical to start seizures in humans. Seizures can be caused by electric shocks, fevers, kainic acid, pilocarpine, pentylenetetrazole, and low magnesium.
One that can be studied is in the temporal lobes by injecting specific chemicals into mice brains. This first seizure can be minutes to hours, but it sets up a focus that will have seizures in an ongoing way with periods without seizures. This research finds that glutamate in excess triggers feedback loops with more glutamate. Glutamate, also, stimulates cannabinoids to decrease the circuits that are hyper excitable. The first glutamate triggers manufacture of more 2-AG in the synapse triggering CB1 receptors that decrease the over active circuits avoiding seizures.
After brain trauma, both cannabinoids are increased to protect against possible seizures. Seizures once started “kindle” a small focus region that then produces seizures in the future. In established long-term epilepsy, the cannabinoid system does not work properly, possibly from less 2-AG production or decreased amount of CB1 receptors. Research is consistent with decreased cannabinoid activity in epilepsy. But, even with a smaller amount of CB1 receptors, they can help by using medications that stimulate them. Cannabinoids, therefore, can be used to treat epilepsy. Several medications that stimulate CB1 act as anticonvulsants in animal models of chronic seizures. Also, several medications that go against the CB1 receptor increase seizures.
The results are variable with some drugs not working with certain chemically induced seizures. These chemicals have multiple complex effects that can cause the seizures outside of the cannabinoid system protection.
A new research advance is that CB1 receptors can be seen on PET scans (positron emission tomography) in live humans. These studies show that in patients with temporal lobe epilepsy there are different effects in different regions. CB1 increases on the opposite side of the brain in the temporal region, but it decreases in several regions of the cortex. Strangely, the amount of cannabinoid receptors in the opposite side correlated with the severity of the epilepsy. This shows that chronic epilepsy has complex changes in cannabinoids in the brain.
Effects of Plant and Synthetic Cannabinoids on Brain Waves and Seizures

Many of the brain wave frequencies appear to be vital to thinking processes. In particular theta and gamma are very important. Those waves that communicate between the hippocampus and the pre frontal cortex are critical to memory and learning. Cannabinoids from plants affect brain oscillations by decreasing some oscillations including those involved with the hippocampus. It is, however, not clear how this relates to the effects of endogenous cannabinoids.
The effects of CB1 receptors have complex effects that are both increasing and decreasing particular frequencies of brain waves. For example they increase oscillations from the thalamus to the cortex by way of nigro-striatal pathways. But, in many other circumstances there is a general decrease. Cannabis smoking can briefly affect cognition and EEGs. Chronic smokers may have decreased gamma waves from sensory nerves. In adolescent mice, synthetic THC decreases gamma waves in the cortex. Therefore, alterations in cognition that occur with intoxication may be related to alterations of brain waves.
Temporal lobe epilepsy is perhaps the most difficult form of seizures to treat in humans. Standard drugs have widespread effects across the entire brain, not just the specific region involved in the seizure. This leads to more side effects. Cannabinoids as we have described are extremely specific in the region that they affect causing less of these side effects. The cannabinoids operate only the very particular regions and time scales where there are seizures with production called upon by the electrical activity. This allows the neurons to trigger the manufacture of the cannabinoids and receptors based on when and where there is abnormal electrical activity. This is why treatment with cannabinoids might not have the same side effects.

There is some evidence that the entire cannabinoid system is negatively altered in very chronic epilepsy. So, medications that stimulate the system might be very helpful. Drugs can affect the breakdown of cannabinoids, or the production of either CB1 receptors or the synthesis of 2-AG.
A downside can be cognitive problems because of altering the function of the hippocampus network through stimulatory neurons. The hippocampus has cannabinoid effects on both inhibitory and Therefore, altering only some part of the cannabinoid system might be best, such as only the stimulatory neurons to maintain hippocampus function.
More study is needed because plant based cannabinoids can help or do the opposite.
One ingredient of marijuana is cannabidiol (CBD). When the plant is bred to have a particularly high amount of cannabidiol, it has been more helpful in some types of epilepsy. The effects of cannabidiol are very complex and not well understood. However, many people have figured out how to use CBD crystals for the most beneficial health effects. It doesn’t have major effects on CB1, which tetrahydrocanabinol (THC) does. The CB1 effects are complex. Cannabidiol seems to affect other cannabinoid systems and may affect particular cells. If you are interested in using CBD then, you might also be interested in discounts on CBD edibles to help save you some money.
Another approach is to stop the creation of new seizure foci by disrupting the normal cannabinoid process that helps start seizures. Research with seizures from high fevers are being looked at in this regard. But, this didn’t work with seizures induced by chemicals.
Cannabinoids and Seizures
Natural cannabinoids in the brain form a system that regulates a vast number of different brain processes. One major effect is to dampen hyper activity in neuronal circuits that could become seizures. This works particularly well because of the unique way that cannabinoids and CB1 receptors can be triggered in very particular places and in very particular time sequences. This allows them to modulate brain waves throughout the brain.
Several of plant based cannabinoids are effective in helping certain kinds of seizures. But, their different effects in different parts of the brain are very complex and not well understood. Cannabinoids often help prevent seizures, but can, also, do the opposite.
The future is bright for this very important field of research.