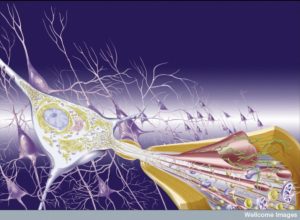
 While mapping connections of neurons has become the holy grail of current neuroscience, it is clear that communication of neurons with many other types cells is, also, vitally important to every aspect of brain function. This includes constant back and forth signaling with astrocytes, microglia, oligodendrocytes, immune cells such as T cells, blood vessel cells such as capillaries, and many other tissue cells. What is also becoming apparent is that the environment around the neuron can greatly affect all aspects of neuronal, and therefore, brain function. These factors can help create the significant degenerative brain diseases including Alzheimer’s, Parkinson’s, ALS and other dementias. These brain diseases have become one of the greatest scourges on earth.
While mapping connections of neurons has become the holy grail of current neuroscience, it is clear that communication of neurons with many other types cells is, also, vitally important to every aspect of brain function. This includes constant back and forth signaling with astrocytes, microglia, oligodendrocytes, immune cells such as T cells, blood vessel cells such as capillaries, and many other tissue cells. What is also becoming apparent is that the environment around the neuron can greatly affect all aspects of neuronal, and therefore, brain function. These factors can help create the significant degenerative brain diseases including Alzheimer’s, Parkinson’s, ALS and other dementias. These brain diseases have become one of the greatest scourges on earth.
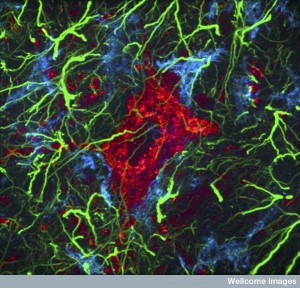 Of all the factors, inflammation of many different types is now shown to be very significant for all aspects of neuronal function. A previous post demonstrated that neurons themselves can create every aspect of inflammation, not just pain. These signals with immune cells occur over regular transmissions of axons, dendrites and synapses. But, they can also occur in special unmyelinated segments of axons that send sideways signals locally. This type of neuro inflammation is used as a form of neuroplasticity, or signaling, to peripheral cells.
Of all the factors, inflammation of many different types is now shown to be very significant for all aspects of neuronal function. A previous post demonstrated that neurons themselves can create every aspect of inflammation, not just pain. These signals with immune cells occur over regular transmissions of axons, dendrites and synapses. But, they can also occur in special unmyelinated segments of axons that send sideways signals locally. This type of neuro inflammation is used as a form of neuroplasticity, or signaling, to peripheral cells.
But, an entirely different, negative effect of inflammation can alter how neurons’ function in their milieu. Glial cells have constant interactions with the immune cells in each region and use a large number of cytokines for communication. This update describes recent research into the relationship of glial cells and inflammation that could be vital for degenerative brain diseases.
Neuro Degenerative Disease
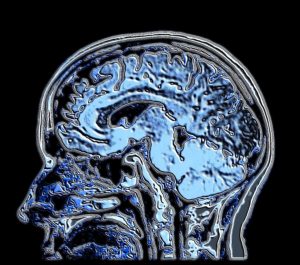 Normal brains deteriorate for various reasons in later life. Age appears to be one important factor for several of the degenerative disorders of the brain, although some can occur much earlier. These diseases have also become more prominent as the life expectancy increases because of advances in the treatment of cancers, infectious diseases and heart disease, the other largest number of ill people. There is no effective treatment currently for dementia and the pathway of a cure is not yet known.
Normal brains deteriorate for various reasons in later life. Age appears to be one important factor for several of the degenerative disorders of the brain, although some can occur much earlier. These diseases have also become more prominent as the life expectancy increases because of advances in the treatment of cancers, infectious diseases and heart disease, the other largest number of ill people. There is no effective treatment currently for dementia and the pathway of a cure is not yet known.
Neurons form the best known and arguably the most significant brain networks. Although astrocyte networks are just as large and are recently beginning to be researched. With almost a hundred billion neurons, each with up to 10,000 connections, the number of neuronal synapses is astronomical. These connections are damaged in neurodegenerative diseases in many different ways, some of which are not understood. Blood vessels are damaged by many different processes and when damaged, can create bleeds or blocked vessels in the brain, killing regions of neurons and other cells. Decreased blood sugar, oxygen, and physical damage all kill regions of blood vessels and neurons.
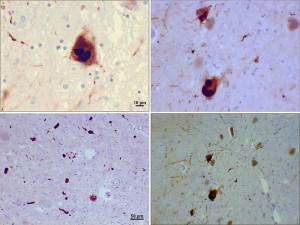
Major degenerative brain diseases include Alzheimer’s, Parkinson’s, frontal temporal lobe dementia (FTLD) and amyotrophic lateral sclerosis. Other brain diseases such as Huntington’s also cause brain damage. Infections, such HIV, and autoimmune diseases like multiple sclerosis also damage the brain. Some of these are considered primary brain diseases, such as Alzheimer’s and others secondary, such as multiple sclerosis. Most of these diseases damage the central nervous system—brain and spinal cord—and not the peripheral nerves. Unfortunately, anyone who suffers from multiple sclerosis could experience painful symptoms in various areas of their body and this can be hard for them to control. They also tend to lose control of some of their bodily functions, like bladder excretion, which leads to incontinence. Luckily, urologists out there like https://www.advancedurology.com/ can offer treatment methods for issues like this that those with multiple sclerosis can practice. Those who suffer with pain don’t have many options on easing it with medication. That’s why some sufferers prefer to take something similar to bc weed express to help minimize any pain that they are experiencing. On some occasions, this may not prove to be effective and that is when you should consider contacting a medical professional who can give you a relevant prescription to help with the pain.
In each of these diseases, very specific regions and circuits of neurons are destroyed producing particular constellations of symptoms. Dopamine neurons are damaged in Parkinson’s disease and motor neurons in ALS. Many of these diseases have specific mis folded proteins, such as amyloid, tau and synuclein. These have been discussed in several previous posts. Amyloid is a molecule that is widely functional in the many different parts of the body and brain. At times, an altered amyloid state occurs with arrays of a specific molecular pattern called beta sheets. These accumulate outside of cells in the hippocampus in Alzheimer’s and also in some other regions in Parkinson’s. These are formed from parts of a molecule called amyloid precursor protein that is cut into pieces.
Another molecule tau that is part of the microtubule structures in the neuron forms clumps called neurofibrillary tangles in Alzheimer’s’. a-synuclein forms clumps called Lewy bodys in dementia from Parkinson’s.
 A small percentage (?5%) of each disease has a clear genetic stimulus. Mutant proteins are correlated with these diseases. These same abnormal proteins might be involved in the larger percentage of the diseases as well.
A small percentage (?5%) of each disease has a clear genetic stimulus. Mutant proteins are correlated with these diseases. These same abnormal proteins might be involved in the larger percentage of the diseases as well.
Previous posts have shown that regions involved in each degenerative disease can have neuronal circuits where pathology appears to be transmitted, possibly in the form of prion like abnormal proteins. However, transmission between animals has not been clearly found in any. Specific regions might be connected in other ways and each becomes vulnerable in sequence as the brain ages. How these protein clumps cause disease has not been clear and some think that there are other primary causes. It is possible that the original precursor molecules might be involved, rather than the abnormal clumps.
Recent findings are, also, consistent with problems in the processes that get rid of debris, as well as problems with mitochondria inside cells which produce energy for all cells. Autophagy is one of the major processes that clears debris and this may be defective. The process whereby mitochondria turn over and clear damage appears to be significant. Also metabolism involving mitochondria produces oxidative molecules that are powerful and destructive. The normal pathways that contain these byproducts of metabolism are also defective.
What is Neuro Inflammation?
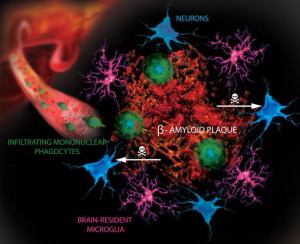 In some brain diseases, the particular type of inflammation is clear. This occurs in multiple sclerosis where lymphocytes and macrophages derived from monocytes are common in brain tissues. The blood brain barrier is compromised and glial cells are very active in unusual ways.
In some brain diseases, the particular type of inflammation is clear. This occurs in multiple sclerosis where lymphocytes and macrophages derived from monocytes are common in brain tissues. The blood brain barrier is compromised and glial cells are very active in unusual ways.
In Alzheimer’s, Parkinson’s and ALS, astrocytes and microglia are altered with much less obvious inflammation and cytokines. Changes in glial cells are similar in all of these different diseases and are also present in traumatic brain injury and strokes. At first these changes in astrocytes and microglia were considered to be due to reaction to destruction of neurons.
Now, however, evidence points to the fact that inflammation itself can further the diseases. One piece of evidence is a study that showed those who used NSAIDs anti inflammation medications for many years tend to have less degenerative brain disease. Later studies using NSAIDs, steroids, and other anti-inflammatory medications to treat Alzheimer’s didn’t show any effect. Other studies appeared to show that defects in neurons were most prominent. This included transplants of dopamine neurons in Parkinson’s disease.
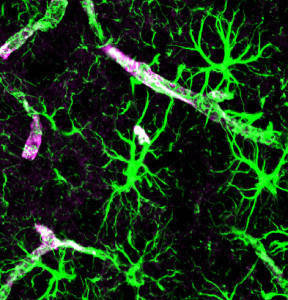
However, continued research with genetic mutations in animal models implicated inflammation again. A gene related to ALS showed close correlation with immune pathways in other cells than neurons. Research with microglia demonstrated increased survival. This survival was based on microglia not changing from the normal form that fights inflammation to a pathological version that increases inflammation. This same gene affected astrocytes, which also stopped microglia from changing into a pro inflammatory form. This showed that interactions between astrocytes and microglia increased brain degeneration.
Another research finding in Parkinson’s disease also supported the model of inflammation being a primary cause. A gene in astrocytes related to a-synuclein produced Parkinson’s symptoms. Then another study showed that dementia from Parkinson’s disease had very specific clumps of both astrocytes and oligodendrocytes (that make myelin) as well as the more traditional clumps of a-synuclein called Lewy bodies.
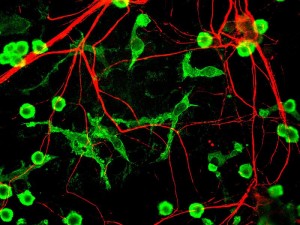 The biggest problem in this new line of research is that microglia and astrocytes when they are used in research outside of live brains do not appear the same as the natural type. In fact, they change into a shape that resembles the pathological type in the research apparatus. Therefore, they have to captured and analyzed in a very short time to actually see if they are pathological. Another big problem is that astrocytes and microglia exist in a three dimensional matrix, which means that two dimensional research models and cultures don’t show what is occurring.
The biggest problem in this new line of research is that microglia and astrocytes when they are used in research outside of live brains do not appear the same as the natural type. In fact, they change into a shape that resembles the pathological type in the research apparatus. Therefore, they have to captured and analyzed in a very short time to actually see if they are pathological. Another big problem is that astrocytes and microglia exist in a three dimensional matrix, which means that two dimensional research models and cultures don’t show what is occurring.
Because of these issues, current research that promotes the idea that special inflammation in the brain furthers degenerative brain disease needs more general definitions. Currently, research points to a constellation of glial cell involvement, changes in the blood brain barrier, and possible inflammation from outside the brain. It remains possible that treatment for these brain diseases could be based on these three factors. The other important point is that this type of inflammation is different from the type in the gut or skin responding to microbes. When this type of data from outside the brain is considered for the brain, mistakes are made in conclusions.
Genetic Associations
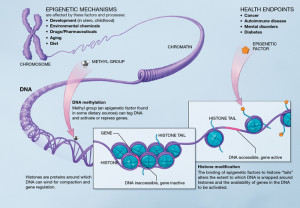 One genetic disorder called Nasu-Hakola disease was found to be based only on a genetic change in microglia. This showed an onset of dementia in midlife, although very rare, could definitely be caused by abnormalities in microglia. This same genes (TREM2) have now been related to three other dementias—Alzheimer’s, Parkinson’s, and FrontoTemporal Dementia.
One genetic disorder called Nasu-Hakola disease was found to be based only on a genetic change in microglia. This showed an onset of dementia in midlife, although very rare, could definitely be caused by abnormalities in microglia. This same genes (TREM2) have now been related to three other dementias—Alzheimer’s, Parkinson’s, and FrontoTemporal Dementia.
There is still much debate about the significance of these findings as related to the causes of brain degeneration. The strongest evidence is in Alzheimer’s where 20 other genes in microglia are related. APOE, the most well-known abnormal gene related to Alzheimer’s, is mostly in astrocytes and the abnormal type of microglia.
Dementia from Multiple Sclerosis
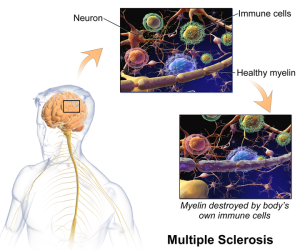 The type of multiple sclerosis that relapses frequently appears to be a unique sub group. People are normal in between these sequential bouts of illness. It is also unusual in that it appears in different parts of the brain in different episodes. This has been related to autoimmune responses that destroy myelin. In this relapsing type of MS, eventually the relapses stop and the nature of the disease changes to a chronic progressive stage of worsening symptoms.
The type of multiple sclerosis that relapses frequently appears to be a unique sub group. People are normal in between these sequential bouts of illness. It is also unusual in that it appears in different parts of the brain in different episodes. This has been related to autoimmune responses that destroy myelin. In this relapsing type of MS, eventually the relapses stop and the nature of the disease changes to a chronic progressive stage of worsening symptoms.
A current view of this progressive MS is that multiple bouts of myelin destruction in particular places can eventually kill neurons. At first myelin is repaired and then the ability to repair is lost. Inflammation in the meninges also occurs seen as the disease progresses and after death. There is reason to believe that these regions of inflammation support destruction of myelin.
Another disease that is somewhat similar to MS does not have this course. In auto antibody mediated astrocytopathy or NMO there is no progressive last phase in any subset. This disease has been correlated with a particular channel called aquaporin-4.
Inflammation can be Positive or Negative
 There is now substantial research associating inflammation and brain degeneration. This research is in mice, worms, zebrafish and human stem cells. One study shows a gene that produces a critical inflammation molecule affects microglia and particular types of inhibitory synapses. These neuronal effects extend to excitatory and inhibitory neurons in altered circuits and are correlated with changes in immune complement molecules.
There is now substantial research associating inflammation and brain degeneration. This research is in mice, worms, zebrafish and human stem cells. One study shows a gene that produces a critical inflammation molecule affects microglia and particular types of inhibitory synapses. These neuronal effects extend to excitatory and inhibitory neurons in altered circuits and are correlated with changes in immune complement molecules.
Such research shows that an immune genetic process from microglia can alter a particular circuit. The way that microglia alter the circuit is through their normal function of pruning synapses. As with most research, there are still questions about the conclusions. It implies, along with other research, that genes related to inflammation in microglia are associated with Alzheimer’s, Parkinson’s and ALS. For now, none of these have resulted in treatments. While more and more gene associations are being picked up by GWAS (large searches among genes in populations), it is still very difficult to find the exact mechanisms for specific genes.
Thus far, there has been less research on glia cells in the periphery as opposed to the brain and spinal cord. These are known to be affected by both inflammation and specific microbes in the gut. Events occurring outside of the brain can influence the brain through defective blood brain barriers, which appear to occur in these degenerative diseases.
Update on Inflammation and Brain Disease
 There is still much uncertainty about the relationship of inflammation and degenerative brain disease. This line of thinking is only twenty years old and started with the finding that abnormal microglia are involved in brain inflammation and related to degenerative brain diseases. This question involves very complex research examining the milieu surrounding neurons and synapses, not just neurons. Inflammation outside the brain can, also, affect the brain. Blood brain barriers also appear to be impaired in major degenerative brain diseases.
There is still much uncertainty about the relationship of inflammation and degenerative brain disease. This line of thinking is only twenty years old and started with the finding that abnormal microglia are involved in brain inflammation and related to degenerative brain diseases. This question involves very complex research examining the milieu surrounding neurons and synapses, not just neurons. Inflammation outside the brain can, also, affect the brain. Blood brain barriers also appear to be impaired in major degenerative brain diseases.
Research is just now unearthing the great complexity of different kinds of inflammation. Immune cells produce many unique kinds of inflammation for different purposes. A previous post showed how some inflammation can help or hurt the brain after traumatic brain injury. While enormously complex and challenging, this line of research is vital to future understanding of dementia.