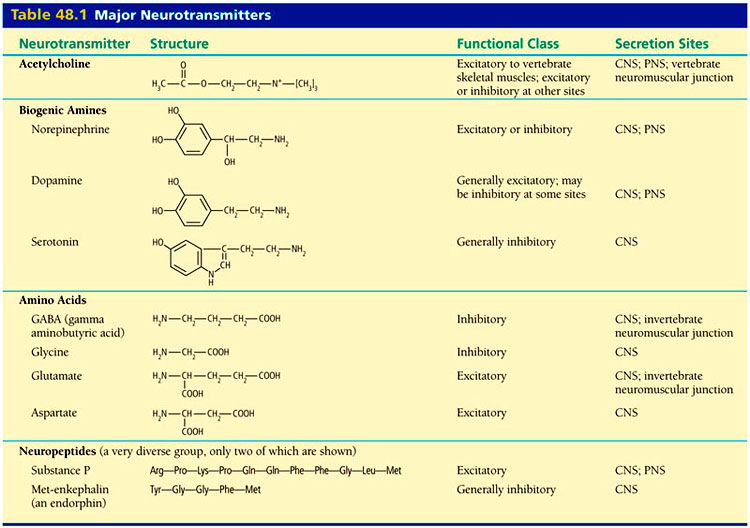
 A dogma from a generation ago taught that each neuron produced one neurotransmitter. It is odd that forty years ago there was already evidence that calcium activity stimulated peripheral sympathetic neurons, which normally release norepinephrine, to switch to release acetylcholine. At that time it was also observed that 80% of early motor neurons in Xenopus laevis embryos were cholinergic but this number was different later without any explanation. There was clearly evidence then for what is now known to be a new type of neuroplasticity involving changes in neurotransmitters.
A dogma from a generation ago taught that each neuron produced one neurotransmitter. It is odd that forty years ago there was already evidence that calcium activity stimulated peripheral sympathetic neurons, which normally release norepinephrine, to switch to release acetylcholine. At that time it was also observed that 80% of early motor neurons in Xenopus laevis embryos were cholinergic but this number was different later without any explanation. There was clearly evidence then for what is now known to be a new type of neuroplasticity involving changes in neurotransmitters.
Like many important scientific discoveries, these strange facts were ignored because they were not consistent with the dogma. In recent years it has become clear that most neurons can make more than one neurotransmitter, as well as manufacturing and secreting many other important factors that modulate the transmission of information. What has not been accepted until now, however, is that an entirely new type of neuroplasticity with changing neurotransmitters has been discovered. This could be a very fundamental discovery affecting future treatment of neurological diseases.
Neuroplasticity Mechanisms
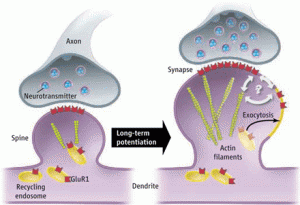 At first, the major mechanism was thought to be long-term potentiation or long-term depression, whereby a single synapse is strengthened or weakened in the process of memory formation. Synapses are now noted to be extremely complex with many moving parts at the same time. Both the pre synaptic and postsynaptic cells are constantly remodeling including vast changes in structure. Also, the matrix between the two cells is very complex and constantly rebuilding. It has also become clear that whenever a memory is reconsidered or “re remembered,” the synaptic connections and the new brain cells incorporated into the circuit are all new.
At first, the major mechanism was thought to be long-term potentiation or long-term depression, whereby a single synapse is strengthened or weakened in the process of memory formation. Synapses are now noted to be extremely complex with many moving parts at the same time. Both the pre synaptic and postsynaptic cells are constantly remodeling including vast changes in structure. Also, the matrix between the two cells is very complex and constantly rebuilding. It has also become clear that whenever a memory is reconsidered or “re remembered,” the synaptic connections and the new brain cells incorporated into the circuit are all new.
In the pre synaptic neuron the axon sends out processes, called boutons, which approach the postsynaptic dendrite. The postsynaptic cells are constantly producing processes called spines that will receive the input from the other cell. It is the head of the spine that is most active, growing and connecting with the bouton. Very complex building and rebuilding of microtubule scaffolding structures determine these changing structures of the membrane. Previous posts have described how these scaffolding molecules appear to be the language of the mind in the neuron.
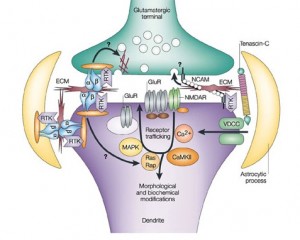 All three regions of the synapse, the pre synaptic and post synaptic neurons and the extracellular space are critical to the function of the transmission of information. Recently, the enormous complexity of the molecules in the extracellular space has been recognized. These neuroligins on the post-synaptic membrane meet the b-neurexins from the presynaptic membrane and shake hands, holding the synapse together. There are actually a wide variety of critical connecting molecules in the synapse. These molecules in the synapse are also regulated by the astrocytes that have been shown to have critical influence on all phases of synapse function (see post).
All three regions of the synapse, the pre synaptic and post synaptic neurons and the extracellular space are critical to the function of the transmission of information. Recently, the enormous complexity of the molecules in the extracellular space has been recognized. These neuroligins on the post-synaptic membrane meet the b-neurexins from the presynaptic membrane and shake hands, holding the synapse together. There are actually a wide variety of critical connecting molecules in the synapse. These molecules in the synapse are also regulated by the astrocytes that have been shown to have critical influence on all phases of synapse function (see post).
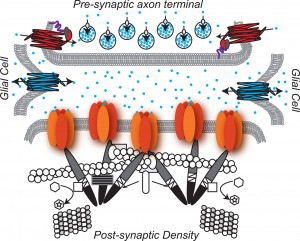 A previous post described recently discovered electrical mechanisms of neuroplasticity, with alterations in the membrane channels that carry the neuronal signals. In addition to the sodium and potassium channels being altered in this “intrinsic neuroplasticity”, calcium channels have also been defined as yet another mechanism that influences the changes in all three ingredients of the synapse.
A previous post described recently discovered electrical mechanisms of neuroplasticity, with alterations in the membrane channels that carry the neuronal signals. In addition to the sodium and potassium channels being altered in this “intrinsic neuroplasticity”, calcium channels have also been defined as yet another mechanism that influences the changes in all three ingredients of the synapse.
The post on astrocytes showed that astrocytes have an entirely different form of electrical transmission where information is sent around the entire brain, a parallel system to the neuron’s axon network. This astrocyte transmission uses a calcium based electrical signal, instead of the sodium and potassium of the neurons. But, recently it was found that even in the neuron, calcium electrical events could have dramatic influence on bringing about a change in neurotransmitters — the totally new form of neuroplasticity described in this post.
It was also noted, that the entire concept of neuroplasticity has changed in the past several years to include wide ranging circuits, all changing in lockstep or in sequence–these neuroplasticity circuits including cortex, cerebellum, and brainstem at the same time with different mechanisms. Many processes, like memory, learning, and response to music – in fact most advanced forms of thought – utilize much of the brain not just a single synapse and utilize this distributed form of neuroplasticity.
Each Cell Has DNA to Make All Neurotransmitters
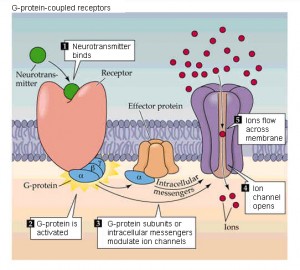 Neurotransmitters have a wide variety of effects. Some change the characteristics of the ion channels in the membrane of the neuron or the postsynaptic neuron. Some trigger signaling cascades from the membrane that can carry all the way into the nucleus. The changes can directly cause an action potential in the postsynaptic neuron. These widely varying mechanisms are triggered by specific receptors and neurotransmitters that fit together as a lock and key. It is, therefore, unusual that a single mechanism spans two cells and the extracellular matrix.
Neurotransmitters have a wide variety of effects. Some change the characteristics of the ion channels in the membrane of the neuron or the postsynaptic neuron. Some trigger signaling cascades from the membrane that can carry all the way into the nucleus. The changes can directly cause an action potential in the postsynaptic neuron. These widely varying mechanisms are triggered by specific receptors and neurotransmitters that fit together as a lock and key. It is, therefore, unusual that a single mechanism spans two cells and the extracellular matrix.
Stem cells are able to use of wider array of genes than those cells that have differentiated into cells for particular organs such as neurons, glia, or heart cells. In the same way, the machinery for all of the neurotransmitters exist in each neuron and are signaled by the extremely complex regulation of DNA and RNA information in forming functional proteins and enzymes.
New Form of neuroplasticity – Changing Neurotransmitters – Dopamine Switch of Light and Mood
An important example of this new form of neuroplasticity is recently described in response to light changes in the length of the day. This example correlates important behavior changes in an animal with the neuroplasticity switch of the neurotransmitter.
The current study shows a switch in the response to the reaction of the day-night cycle. Adult rats showed a neurotransmitter switch when experimentally exposed to a long day (19 hours of day, five of night) and a short day (opposite 19 hours of dark, 5 of light).
In the short day experience, dopamine neurons in the hippocampus increased and in the long day cycles the number of dopamine neurons decreased. In the rat, dopamine has widespread effects on learning, memory, motivation and mood.
I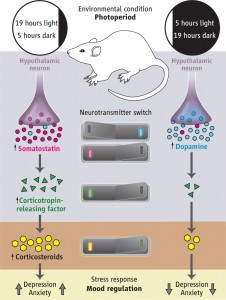 n the short day experience somastatin neurons decreased in the hippocampus and in the long day increased. Somastatin in rats is not as clearly understood but appears to be related to stress. In humans it is found in many brain regions.
n the short day experience somastatin neurons decreased in the hippocampus and in the long day increased. Somastatin in rats is not as clearly understood but appears to be related to stress. In humans it is found in many brain regions.
These two events involved specific neurons changing their neurotransmitter as the length of the day changes. The two different neurotransmitters were coordinated to respond to the amount of day and night. When each of the two different neurotransmitters was active, the behavior of the animal completely changed, including the mood and levels of anxiety. These same alterations in mood, based upon the amount of sunlight and the length of the day, are also seen in human beings with seasonal affective disorder– and in a milder way in most people.
Importantly, these changes correlated with the hypothalamus secreting different amounts of the cortico-releasing factors (CRF). The ratio of dopamine to somastatin changed the amount of CRF and steroids in the blood. Steroids appear to be controlled by the balance of dopamine and somastatin, with both receptors appearing on cells that produce CRF, which triggers the steroid producing cells.
Mechanisms of Neurotransmitter Switch
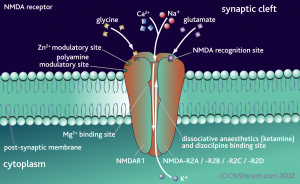 Many mechanisms have been described for the neurotransmitter switch. Electrical activity, neuronal secreted factors, and intercellular interactions can all stimulate the change of the neurotransmitter for a particular neuron, both in the fetus and the adult brain. What is most intriguing is that the postsynaptic neuron also changes the receptors. The regulation appears to be in the synapse.
Many mechanisms have been described for the neurotransmitter switch. Electrical activity, neuronal secreted factors, and intercellular interactions can all stimulate the change of the neurotransmitter for a particular neuron, both in the fetus and the adult brain. What is most intriguing is that the postsynaptic neuron also changes the receptors. The regulation appears to be in the synapse.
The switch can be as extreme as changing from an excitatory neuron (glutamate) to an inhibitory neurotransmitter (GABA). For example, some circuits change when there is a very high level of activity – too much activity. In order to stave off problems related to excessive excitatory activity, which can be damaging, neurons switch to inhibitory neurons to protect the system.
Motor and sensory neurons don’t usually change because it is important to have a consistent function in these systems. Much of the research has been on interneurons where it seems to be more common.
Electrical Stimulation of Neurotransmitter Change
Until this new discovery it was thought that neurons specify their only neurotransmitter from an early age. In the fetus, calcium based electrical signals are important in forming these early synapses.
It has not been appreciated until now that neurons have electrical capacity before there are synapses — while still in the fetus. The duration of the initial action potentials can be very long and these affect the maturation of the sodium and potassium channels. In those neurons newly made in the adult this same process occurs.
When the neurons have large calcium electrical events before any synapses form then this will determine the type of adult neuron it will become.
In the adult, when neurons have already developed synapses, different types of calcium based electrical activity can also stimulate a neurotransmitter change. Before this recent discovery there were other examples of changes in neurotransmitters with light and darkness in the cells of the hypothalamus. Interneurons where noted to fluctuate between secreting dopamine and neuro-peptide Y. These are also dependent upon the particular type of calcium spiking electrical activity. Other examples include neuroplasticity changes in animals when sensory information is blocked in the olfactory process — reducing the number of dopamine neurons while maintaining the GABA neurons.
If the electricity stays constant then the neuron will keep the same neurotransmitter. If not it will change.
Post Synaptic Receptors Also Change With the Pre Synaptic Neurotransmitters
One of the most remarkable aspects of these changes is that both the neurotransmitter changes and the receptor on the next cell changes in a similar way to make a functioning synapse. Clearly if only one of these changed then the transmission could not occur and it would not be relevant.
One recent example occurs in the neuromuscular junction where acetylcholine is the noted neurotransmitter of the neuron and, of course, binds to the acetylcholine receptors on the muscle. But, in the African clawed frog, the muscles have many different kinds receptors prior to the neuron making connections. These pre existing receptors include acetylcholine, glutamate, GABA, and glycine.
However, upon closer examination of the muscle cell there were many other messenger RNA transcripts and proteins that specifically make up the receptors for a wide variety of other receptors – nicotinic, AMPA, NMDA, GABAA,, and glycine. It is possible that many cells make a wide variety of receptor types and then solidify the use when the neuron connects and starts sending the specific neurotransmitter –- glutamate for invertebrate neuromuscular junctions, and acetylcholine for vertebrate junctions.
In the fetus when the calcium spikes stimulate the change in neurotransmitters, they also change the receptor to match. When there is increased calcium electrical spikes then GABA and glycine inhibitory neurotransmitters occur (the example before of lowering the excitability) and the corresponding receptors occur also. When the spiking is low then there is an increase in AMPA and NMDA (excitatory neurons) as well as acetylcholine– again, both neurotransmitters as well as receptors.
The level of calcium electricity, plus a variety of secreted factors, determines the type of neurotransmitter from the pre synaptic neuron and the corresponding receptors in the postsynaptic neuron.
Another example of this occurs in the hypothalamus melanotrope cells that allow animals to change pigment. These neurons have utilized the neurotransmitters NPY and GABA, not dopamine. Studies find no dopamine receptors present. When exposed to light, however, there is a production of dopamine in the neurons. The melanocyte cells then produce receptors to dopamine in order to connect with the new dopamine being used by the neuron stimulating the cell.
Neurological Disorders
There are many possible implications and questions related to these new findings. Are negative neuroplasticity with correlated changes of neurotransmitter and receptor involved in causing disease?
Is it possible that neurological disorders could be caused by this type of neuroplasticity — diseases where specific neurotransmitters are decreased?
Those neurological diseases with a decrease in specific neurotransmitters include dopamine in Parkinson’s’ disorder, acetylcholine in Alzheimer’s and AMPA in fragile X. Could switches of neurotransmitters of existing cells reverse these changes, perhaps though calcium electrical signaling?
Another example involves seizure activity, which leads to release of GABA from normally glutamate producing mossy fibers from the dentate nucleus to CA3 in the hippocampus. After the seizure, the dentate releases GABA and inhibits beta/gamma oscillations in the CA3 area that are involved in memory. This could be the cause of the amnesia after a seizure.
New Type of Neuroplasticity with Changing Neurotransmitters
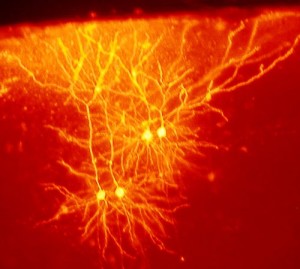 Just as the unidirectional dogma of DNA has been supplanted by a much more complex bidirectional flow in the genome, the dogma of one neurotransmitter per neuron has been shown to be incorrect. This new example of the dramatic ability of the brain to change adds to a growing list of neuroplasticity mechanisms.
Just as the unidirectional dogma of DNA has been supplanted by a much more complex bidirectional flow in the genome, the dogma of one neurotransmitter per neuron has been shown to be incorrect. This new example of the dramatic ability of the brain to change adds to a growing list of neuroplasticity mechanisms.
In this case, secreted factors and calcium electrical spikes can stimulate an alteration in both the neurotransmitter of the pre synaptic neuron and the receptor of the postsynaptic neuron. Neuroplasticity has been shown to occur not in isolation, but as part of distributed brain-wide circuits with a wide variety of mechanisms constantly building and rebuilding connections. Just as research in genetic mechanisms demonstrates increasing complexity, so does the research into the brain’s ability to change itself.
The growing complexity of the whole brain mechanisms add to the difficulty of finding a central place in the brain that would correlate to the unitary subjective experience of the mind. Since these extremely complex changes are occurring in correlation with thinking in millisecond time all over the entire brain, perhaps these mechanisms are more consistent with an interaction of the brain with mind.