It is unfortunate that the word “depression” is used in many different ways and is at the same time chosen to represent a brain illness. There are, undoubtedly, many subtypes of the illness depression and the current clinical definition includes a list of mental and physical symptoms, some of which are severe—sleep disorder, lack of interest in usual activities, excessive guilt and rumination, lack of energy and fatigue, lack of concentration, change in appetite with weight loss or gain, a change in the level of movement either with agitation and retardation and suicidal ideation. Other important symptoms include pain and generally feeling lousy for long periods.
It is unfortunate that the word “depression” is used in many different ways and is at the same time chosen to represent a brain illness. There are, undoubtedly, many subtypes of the illness depression and the current clinical definition includes a list of mental and physical symptoms, some of which are severe—sleep disorder, lack of interest in usual activities, excessive guilt and rumination, lack of energy and fatigue, lack of concentration, change in appetite with weight loss or gain, a change in the level of movement either with agitation and retardation and suicidal ideation. Other important symptoms include pain and generally feeling lousy for long periods.
With such a long list of possible symptoms, there are many subtypes which include some of these symptoms, but not all. With eight discrete symptoms in the definition, there are hundreds of possible variants, but less in clinical experience. It is striking that many of these symptoms occur, also, with medical illness and infections—the “sick feeling” described in previous posts triggered by T cells. Also, a self-limiting form occurs naturally with grief.
Recent research shows that one particular subtype of depression—perhaps as much as a third to a half of cases—is closely related to the occurrence of inflammation, and it is possible that this group might respond to different kinds of treatment than the others. This post will describe the reciprocal relationship of depression and inflammation.
Wired Nervous System Brain and Wireless Immune System Brain
 A previous post described the nervous system and immune system as the two components of the brain—one wired for rapid signals and one wireless for hard to reach places. But, even more recent research shows that the traditional brain cells—neurons, microglia, astrocytes, oligodendrocytes—can, also send cytokine wireless signals like the many immune cells. The truly integrated relationship of the immune and nervous systems is now being discovered.
A previous post described the nervous system and immune system as the two components of the brain—one wired for rapid signals and one wireless for hard to reach places. But, even more recent research shows that the traditional brain cells—neurons, microglia, astrocytes, oligodendrocytes—can, also send cytokine wireless signals like the many immune cells. The truly integrated relationship of the immune and nervous systems is now being discovered.
The immune system doesn’t just fight microbes and disease with inflammation. It, also, resists psychological stress and social isolation. The central nervous system doesn’t just direct sensation, movement, and cognition, but it, also, produces and regulates every aspect of inflammation as a form of neuroplasticity (see post on neuro inflammation). Neurons can, also, stimulate specific mast and dendritic cells to fight microbes. Immune communication is with cytokines, cell to cell contact, and production of antibodies. The nervous systems sends rapid signals, but, also, local secretion of cytokine signals.
Pain is, also, part of the new picture with pain fibers sending signals in both directions that can alter immune response by stimulating immune blood cells into action and changing the flow of blood. By severing a nerve related to arthritis, inflammation is decreased. There are very complex feedback loops between immune cells and neurons. Some lymphocytes secrete dopamine and are affected by dopamine. They are stimulated by dopamine and in turn use dopamine to signal to other cells.
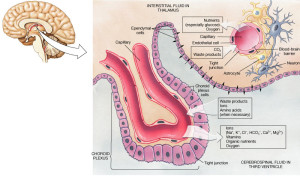 Supposedly, the brain was off limits to immune cells. It is now known that this isn’t true. The choroid cells provide the gate keeping and intelligence to summon immune cells to stimulate cognition and to fight infections. (See post on the Very Intelligent Choroid Plexus Epithelial Cell.)
Supposedly, the brain was off limits to immune cells. It is now known that this isn’t true. The choroid cells provide the gate keeping and intelligence to summon immune cells to stimulate cognition and to fight infections. (See post on the Very Intelligent Choroid Plexus Epithelial Cell.)
Microglia rapidly protect against microbes, either by themselves or by calling in other immune cells. But, they also protect against synapses that aren’t needed by constantly regulating and pruning neuronal synapses and connectivity. Microglia regulate stem cells that produce neurons and eliminate neurons if there are too many. (See post on microglia) Immune cells help rebuild axons and complement helps prune synapses. Immunoglobulin molecules help guide axons as adhesion molecules.
The cytokine interleukin 6, is tied to hunger and the ability to burn fat and lose weight. The ‘sick feeling” includes fatigue, pain, and lack of interest and is triggered by microglia in the brain. Microbes trigger a vagus signal to the brain picked up by microglia triggering sick feeling.
Inflammation and Depression
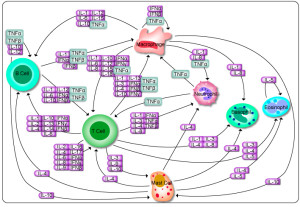 Inflammation increases depression and depression increases inflammation. In the subtype of depression associated with inflammation, excessive cytokines are stimulated in response to extreme stress, as well as increased reactions to actual infections. Many other emotional and physical factors contribute to inflammation including obesity, metabolic syndromes and severe psychosocial stressors. The altered responses with depression include extreme inflammatory reactions, which can cause pain and poor sleep. Inflammation is, also, increased by lifestyles with no exercise and very poor diet. All of this creates a vicious cycle with increased depression and inflammation.
Inflammation increases depression and depression increases inflammation. In the subtype of depression associated with inflammation, excessive cytokines are stimulated in response to extreme stress, as well as increased reactions to actual infections. Many other emotional and physical factors contribute to inflammation including obesity, metabolic syndromes and severe psychosocial stressors. The altered responses with depression include extreme inflammatory reactions, which can cause pain and poor sleep. Inflammation is, also, increased by lifestyles with no exercise and very poor diet. All of this creates a vicious cycle with increased depression and inflammation.
Those people who have significant amounts of inflammation have the strongest responses to treatments that fight inflammation such as omega-3, celecoxib, and exercise, as well as medications that counteract the effects of cytokines. Those patients that have both inflammation and depression appear to need treatment for both.
 There is greater inflammation in diabetes, cardiac disease, arthritis, asthma, allergic disease and pain syndromes. In all these diseases, there is, also, an increase of depression. There are strong correlations of inflammatory cytokines such as several interleukins and c-reactive protein, with depression. These include interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-a), IL-1b, the soluble IL-2 receptor, the IL-1 receptor antagonist (IL-1ra), and C-reactive protein (CRP).
There is greater inflammation in diabetes, cardiac disease, arthritis, asthma, allergic disease and pain syndromes. In all these diseases, there is, also, an increase of depression. There are strong correlations of inflammatory cytokines such as several interleukins and c-reactive protein, with depression. These include interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-a), IL-1b, the soluble IL-2 receptor, the IL-1 receptor antagonist (IL-1ra), and C-reactive protein (CRP).
Likewise, patients with depression have increases in these same cytokines. This correlation occurs in children and adults. About a third of depressed patients have these changes in inflammatory markers.
Cytokines can affect neurotransmitters dopamine, serotonin and glutamine and neuronal activity. (See post on neuroinflamation). These cytokines can come from immune cells not in the brain but affect the brain (like T cells signaling in CSF for sick feeling). They can alter tryptophan metabolism to make less serotonin and instead increase kynurenine, a different metabolite of tryptophan, which can precipitate depression symptoms. This occurs when those with hepatitis C are treated with the cytokine interferon. Kynurenine is associated with depression with heart disease and is associated with cognitive changes in schizophrenia and Alzheimer’s.
Effects of Cytokines in Depression and Inflammation
Cytokines can increase oxidation, damaging neurons and glial cells in the PFC and amygdala. They can increase glutamate causing neurotoxicity and decreasing BDNF, which is related to the function of neurons and the creation of new neurons. These cytokine changes can alter functions of neurons. They particularly alter the response to stress and motivation. They can alter the HPA causing changes in glucocorticoid adrenal hormones a well-known sign of depression.
Normally, glucocorticoid hormones decrease inflammation, but resistance can develop from inflammation. When the normal defense against inflammation is damaged, it can increase without regulation through multiple signaling pathways involving nuclear factor-kB and NF-kB. These increase depression further. This resistance affects CRH and other cytokines involved in the regulation of the glucocorticoids.
A major effect of these alterations is decreased creation of new neurons in the hippocampus, the center of memory and learning. The hippocampus has many glucocorticoid receptors and BDNF is very involved in stimulating new neurons for learning and memory. When the glucocorticoids are disregulated, it leads to less dendrites, less BDNF and less new neurons. It is now well known that the hippocampus shrinks in many cases of depression and that the treatment with antidepressant medications starts up the creation of new neurons bring the hippocampus back to normal. In depression, cognitive ability is slowed.
There are a wide range of different responses to these very complex networks involving immune cells, neurons, neurotransmitters and cytokines. Some people do not have depression from inflammation. The complex mechanisms are not yet worked as to why these individual differences occur. Some develop depression with low-level inflammation. This can produce a decrease of function of the parasympathetic system and an increased response to threat or fight and flight reactions from social situations. It can produce abnormal activity in the amygdala and anterior cingulate and a smaller hippocampus with less memory.
 Inflammation causes immune cells to signal to produce sickness behavior such as fatigue, lack of interest, lack of appetite and decreased cognition. It can increase negative mood as well. These are all part of depression. The sick feeling also includes increased sensitivity to pain, another close relationship to immune signaling. Research shows cytokines can increase mood or increase fatigue, pain and increase inflammation.
Inflammation causes immune cells to signal to produce sickness behavior such as fatigue, lack of interest, lack of appetite and decreased cognition. It can increase negative mood as well. These are all part of depression. The sick feeling also includes increased sensitivity to pain, another close relationship to immune signaling. Research shows cytokines can increase mood or increase fatigue, pain and increase inflammation.
When cytokines are used for cancer treatment, they are well known to cause depression in half of the patients. In particular, interferon treatment can cause all of the depressive symptoms, especially changes in sleep and appetite and lack of interest. These results are worse with people who already have had depression or have high levels of inflammation. Patients with depression caused by these cytokines do not respond as well to antidepressant medications. In a recent study with a cytokine antagonist, only those patients with high CRPs showed decreased depression.
Gut Microbes, Inflammation and Depression
 Previous posts have shown that microbes in the gut mediate some of these effects. Stress and poor diet create changes in the gut, altering the microbes and increasing the ability of microbes to enter the body from the gut causing more inflammatory reactions. Current research shows that the brain doesn’t only respond to microbe threats through the immune reactions, but other threats as well including adversity and psychosocial stress.
Previous posts have shown that microbes in the gut mediate some of these effects. Stress and poor diet create changes in the gut, altering the microbes and increasing the ability of microbes to enter the body from the gut causing more inflammatory reactions. Current research shows that the brain doesn’t only respond to microbe threats through the immune reactions, but other threats as well including adversity and psychosocial stress.
Gut microbes are critical in producing many different types of signals related to immune cells and the brain. Stressors affect the microbe composition and behavior and signals from the gut microbes alter the brain responses. These have been demonstrated to be related to obesity, mood and inflammation.
 A recent unusual finding is that depression can alter the leakiness of the gut lining cells (see The Very Intelligent Intestinal Epithelial Cell). This allows microbes and endotoxins to enter the system producing inflammation. Depressed patients have more antibodies to gut microbes. Patients with alcoholism and depression have even greater leakiness.
A recent unusual finding is that depression can alter the leakiness of the gut lining cells (see The Very Intelligent Intestinal Epithelial Cell). This allows microbes and endotoxins to enter the system producing inflammation. Depressed patients have more antibodies to gut microbes. Patients with alcoholism and depression have even greater leakiness.
Some research is beginning to look at treatment of depression and inflammation through gut brain signaling with the vagus nerve, hormones and spinal cord. Probiotics, antibiotics and supplement research is ongoing as well. In some animal studies probiotics reduced leakiness after severe stress. It is possible that they can decrease depression by lowering inflammation and decreasing adrenal hyperactivity. In animal research, extreme stress alters the adrenal stress system and the microbe composition.
Childhood Abuse and Neglect
Mistreatment of children can cause adult depression if there is additional stress later. These depressions are often the most difficult to treat and have high inflammatory markers—CRP, IL-6 and TNF-alpha levels even in old age. These adults have changes in the hypothalamus-pituitary-adrenal function and autonomic stress responses. They have decreased parasympathetic function producing reduced heart rate responses and inflammation. Trauma victims have corticoid hormone abnormalities. The receptors in the hippocampus have epigenetic tags related to inflammation and increase inflammation activity based on stress. When stress occurs in the present, they had more than twice the interleukin levels in response.
Sickness Behavior
Pain and poor sleep, also, are closely related to inflammation and depression.
Normally, sickness feelings and behavior benefit an animal by reducing activity and allowing healing to occur for infections. But, they can, also, produce longer lasting depression symptoms. The post on neuro-inflammation showed that neurons can produce all symptoms of inflammation, not just pain.
Pain by itself produced inflammation activity. The more pain, the more inflammation response and the more there were other depression symptoms. Pain and depression are very closely tied. Many patients with depression have pain and many pain patients develop depression. If the depression is treated, then the pain is reduced as well.
Sleep alteration is a primary symptom of depression. Those with very poor sleep have a much higher chance of developing depression. When sleep is poor, cells produce many signals and cytokines that cause inflammation and this further stimulates depression. Blocking cytokines helped sleep in some case.
The more sleep and pain, the more inflammation and depression causing more of the same vicious cycle increasing all four. Poor eating and fatigue further exacerbates all of these symptoms.
Diet with Inflammation and Depression
 There is increasing evidence that metabolic syndrome caused by excessive fatty and refined sugar diets cause liver disease and inflammation. Mediterranean diet reduced inflammatory markers in these patients. In a six-year study of depressed elderly patients, inflammation did not increase with Mediterranean diet but did increase for those without the diet.
There is increasing evidence that metabolic syndrome caused by excessive fatty and refined sugar diets cause liver disease and inflammation. Mediterranean diet reduced inflammatory markers in these patients. In a six-year study of depressed elderly patients, inflammation did not increase with Mediterranean diet but did increase for those without the diet.
Fasting or food restriction can decrease both depression and inflammation. Even without weight change, fasting lowers inflammation markers. But, this research is confounded by the fact that less eating goes along with depression and can reduce inflammation.
Depressed patients have decreased omega-3 fatty acids. EPA is more anti-inflammatory than DHA. Low omega-3 is correlated with increased inflammatory markers. EPA reduced the amount of depression in those treated with interferon. Those with pre existing depression and inflammation often respond better. Omega protects best for those who had an inflammation challenge.
Exercise has been helpful to decrease some depression and produces decreased inflammatory markers. Those with pre existing inflammation benefited the most in decreasing depression. Exercise didn’t decrease inflammation in those who didn’t have significant inflammation before the study.
There is a reciprocal relationship between obesity and depression, both increasing the other. While some depressed people stop eating, many increase eating and develop the metabolic syndrome. Obesity induces chronic inflammation with high markers. Fat tissue increases cytokines that produce cortisol that increases more fatty tissue. Fat increases the response to stress with more inflammation. Stress, depression, and obesity cause more inflammation and stronger inflammation reaction, which causes more depression. The greater the amount of inflammation present, the greater the future inflammatory response will be.
Stress, Depression and Inflammation
Not only childhood stress causes depression, but other life stresses can as well. Depressed people respond worse to stress, and have more stressors. The depression interferes with their social life causing more isolation, stress and trauma.
In mice, experimental stress increased inflammation markers. Stressed mice had greater inflammation response to microbe endotoxin. High fat meals caused intestinal leaking, increased endotoxin and increased reactivity. The same response to endotoxin occurs with social stress. The worst reactions occur with both endotoxin and stress. Social stress increased gut leaking from fatty meals and produced much more inflammation and markers. The greater the social stress the greater the inflammation response to endotoxin.
Increased cytokines alter behavior related to social stressors. Those with the most reactivity had the most depression. If the cytokine was blocked with a medication, the psychological reactivity was less. Differences of inflammation markers in individuals predict how they will respond to social and psychological stress.
During depression, stress and microbes both cause greater inflammation response. With continued stress, the exaggerated responses continue. The greater the inflammatory response, the more negative are the psychological and physical responses.
Depression Treatment
Current research includes use of anti inflammatory medications for depression (celecoxib). Treatment of depression which lowers inflammation works in those depressed people who, also, have substantial inflammation and pain.
One strange finding is that placebos worked better than anti inflammatory drugs and omegas for those depressed patients without inflammation (could anti inflammatory medications work opposite in these patients?). There are probably many subtypes of those with inflammation and those without. EPA worked better than placebo with patients who had higher inflammation markers. But, worked best when all of the markers were higher.
Both cognitive behavior therapy and meditation practices have been shown to decrease inflammatory markers. Treatment of pain and sleep helps decrease inflammation and depression. CBT helps sleep and can reduce inflammation and pain.
Very recent research shows correlations with inflammation and other mental illnesses as well. Other mental illnesses have increased inflammation markers but in different patterns—schizophrenia, bipolar and depression. Genetic studies show some overlap of these three different illnesses. When acute, patients have increased IL-6 and IL-2. In major depression and schizophrenia the cytokines increase acutely, decrease with treatment and then later rise again. These two illnesses, also, had increases of SIL-2R and IL-1. IL-6 was most increased for all three and is related to acute and chronic inflammation.
Reciprocal Relationship of Inflammation and Depression
 It is not possible to separate the brain and the immune systems. They work together constantly for both psychological, emotional and physical factors. About a third to half of depressed patients have high correlation with inflammation and pain.
It is not possible to separate the brain and the immune systems. They work together constantly for both psychological, emotional and physical factors. About a third to half of depressed patients have high correlation with inflammation and pain.
Being a community of intelligent cellular communities, with coordinated activity of immune cells, microbes and brain cells, it becomes increasingly difficult to understand how this can all be directed or affected.
Mental events stimulate activity in all of these different cells at the same time–immune cells, brain cells, bodily organ cells and microbes. How can this be explained? Isn’t it reasonable to consider mind as an innate aspect of all of these cells.