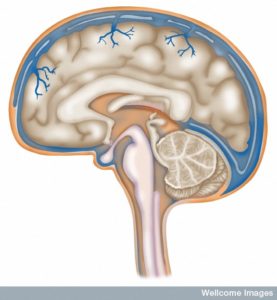
 Small lining cells along the border of the brain’s ventricles are the gateway and supervisor of the relationship between the brain and the rest of the body, including the immune system. The choroid plexus cells produce cerebral spinal fluid, which bathes the brain and provides a cushion. But, it, also, performs a vast amount of other much more complex functions. The fluid filled ventricles are a critical source of sophisticated communication between brain and immune cells.
Small lining cells along the border of the brain’s ventricles are the gateway and supervisor of the relationship between the brain and the rest of the body, including the immune system. The choroid plexus cells produce cerebral spinal fluid, which bathes the brain and provides a cushion. But, it, also, performs a vast amount of other much more complex functions. The fluid filled ventricles are a critical source of sophisticated communication between brain and immune cells.
The choroid plexus has many different functions. It eliminates waste products from brain tissues. It controls the travel of immune T cells in the CSF that promote cognition and fight infections. It regulates the travel of critical microglia and neural stem cells that create new brain cells of all types. It is the major determinant of critical neuroplasticity time windows during brain development. It is vital for the health of the brain regulating the immune surveillance and the maintenance and pruning of synapses.
 In reality, there is no separation between the wired brain of the nervous system sending rapid signals great distances and the wireless brain of the immune system signaling with cytokines and traveling to hard to reach places. Both respond to infections and to stress and mental events. The nervous system, also, produces wireless cytokine signals to trigger the movement of immune cells and pain circuits. The immune system does much more than fight infections; it, also reacts to stress and depression and triggers increased and decreased cognition, fatigue, anorexia, and pain. Glial cells fight infections in the brain and stimulate, regulate and prune synapses for better cognition.
In reality, there is no separation between the wired brain of the nervous system sending rapid signals great distances and the wireless brain of the immune system signaling with cytokines and traveling to hard to reach places. Both respond to infections and to stress and mental events. The nervous system, also, produces wireless cytokine signals to trigger the movement of immune cells and pain circuits. The immune system does much more than fight infections; it, also reacts to stress and depression and triggers increased and decreased cognition, fatigue, anorexia, and pain. Glial cells fight infections in the brain and stimulate, regulate and prune synapses for better cognition.
Much of the coordination of the wired and wireless components of the brain occurs at the window into the brain—the choroid plexus—the center of the communication between the two brains. Choroid cells determine which cells can enter the brain and which are needed. Choroid cells are perhaps the most intelligent of all cells in the brain.
Cerebrospinal Fluid (CSF)
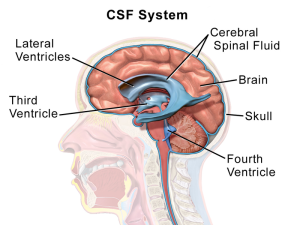 CSF fluid is secreted by the choroid cells into the lateral ventricles; it flows to the third and fourth ventricles and then to the spinal cord. It is taken up in the sub arachnoid space into lymph near the cribiform plate and the newly discovered sinus lymphatics to lymph nodes and the blood. The chroid plexus secretes up to 500 ml of fluid per day into the ventricles.
CSF fluid is secreted by the choroid cells into the lateral ventricles; it flows to the third and fourth ventricles and then to the spinal cord. It is taken up in the sub arachnoid space into lymph near the cribiform plate and the newly discovered sinus lymphatics to lymph nodes and the blood. The chroid plexus secretes up to 500 ml of fluid per day into the ventricles.
The CSF has been very stable during evolution in all vertebrates, but is more complex in more evolved brains. Only recently, with increased knowledge of cellular signaling, has it been understood how complex its contents are and how critical these are to maintain the brain’s function. The CSF provides pressure, which is necessary to build and maintain the brain. If the pressure is too low, the brain can’t grow or function, and it is too high tissue is destroyed (hydrocephalus).
The Choroid Plexus
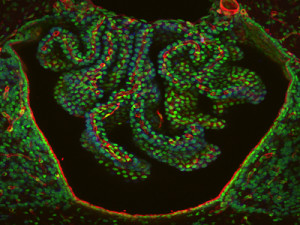 The special choroid tissue exists in parts of multiple places in the four ventricles. It develops first in the fourth ventricle, then the two lateral ventricles and finally in the third. Each of these different versions of the choroid plexus are derived from different types of fetal tissue and vary from species to species.
The special choroid tissue exists in parts of multiple places in the four ventricles. It develops first in the fourth ventricle, then the two lateral ventricles and finally in the third. Each of these different versions of the choroid plexus are derived from different types of fetal tissue and vary from species to species.
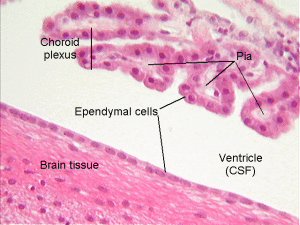
The choroid has a single layer of epithelial cells that line the fluid filled ventricles. This is similar to the single layer of cells lining the gut (see post on the very intelligent intestinal epithelial cell). The gut epithelial cell monitors very complex signaling between trillions of microbes above and many different immune cells below. The choroid cells monitor brain and immune signaling and are bounded by the CSF fluid above (apical side) and many small capillary blood vessels and connective tissue below (the stroma on the basal side).
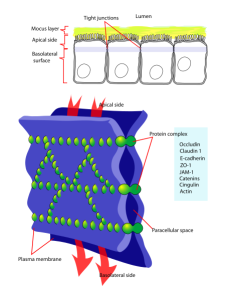 This single line of cells is tightly bound together to form the blood brain barrier (BBB) that maintains a critical separation between the CSF and the blood, allowing only specific molecules and cells to pass through. This function starts in the fetal period. The BBB limits the flow of molecules between the blood and the brain. There are a variety of different transporter molecules that change during development and regulate material at each stage of development with various molecules.
This single line of cells is tightly bound together to form the blood brain barrier (BBB) that maintains a critical separation between the CSF and the blood, allowing only specific molecules and cells to pass through. This function starts in the fetal period. The BBB limits the flow of molecules between the blood and the brain. There are a variety of different transporter molecules that change during development and regulate material at each stage of development with various molecules.
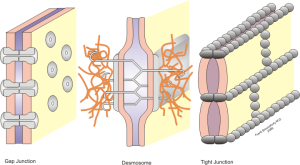
The barrier is based on a complex use of three different junctions holding the epithelial cells tightly together. These are, perhaps, the most closely bound cells in the brain. Each cell has three distinct types of junctions on the sides of the cells to stop any leakiness. The closest to the fluid is the tight junction. Then, there is the adherence junction and third, the desmosome. These all involve many genetic signals for adhesion molecules.
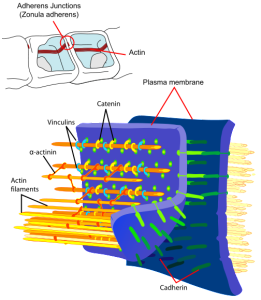 The tight junctions connect two parts of the cell membranes, making it impermeable to fluid. The adherens junction are made up of protein complexes connecting the two cells including their actin cytoskeleton. The desmesome is another specialized adhesion complex that forms spots of adhesion randomly placed on the lateral sides of the cells.
The tight junctions connect two parts of the cell membranes, making it impermeable to fluid. The adherens junction are made up of protein complexes connecting the two cells including their actin cytoskeleton. The desmesome is another specialized adhesion complex that forms spots of adhesion randomly placed on the lateral sides of the cells.
 Sympathetic and parasympathetic nerves regulate the blood flow and the secretion of CSF. Blood comes from the anterior choroidal artery of the internal carotid or middle cerebral artery and the posterior choroidal artery from the posterior cerebral artery. The cerebellar posterior inferior also supplies another section.
Sympathetic and parasympathetic nerves regulate the blood flow and the secretion of CSF. Blood comes from the anterior choroidal artery of the internal carotid or middle cerebral artery and the posterior choroidal artery from the posterior cerebral artery. The cerebellar posterior inferior also supplies another section.
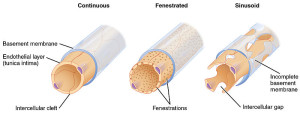
The capillaries are unusual in that they are fenestrated with pores about 70nm in diameter. The pore is covered by a diaphragm, which has radial fibril, which allows small molecules and some proteins to pass. They allow rapid flow of water from blood to the epithelial cells to make more CSF. Some of the materials diffuse through and some are actively transported with vesicles.
Choroid cells are the gateway for all immune cells to enter the brain. The choroid has resident macrophages and dendritic cells in the tissue and special immune Kolmer epiplexus cells in the ventricle near the epithelial lining cell. During development, microglia enter the brain through the choroid plexus and they stay closely tied to the choroid. Both the microglia and choroid are involved in regulation of neuron stem cells, neuroplasticity and circuit pruning and function.(see post on microglia).
When necessary, the choroid can open the gateway and allow many other types of immune cells into the CSF and brain. In fact, the choroid assesses the particular situation and calls for very specific needed cells. Immune cells are first let into the tissue from blood across the fenestrations. They travel along the bottom sides of the endothelial cells into tissue and the ventricle fluid. Complex adhesion molecules aid this travel from the blood into the CSF—IL-13, IL-10, along with other cytokines.
Healthy CNS has 90% T cells, 5% B cells, 5% monocytes and a very small amount of dendritic cells. It is now clear that there are many patrolling cells in the CSF. See the post on T cell functions, such as signaling from the CSF for either normal cognition or during infection, the “sick feeling” with reduced cognition. Extreme reactions of these cells can cause multiple sclerosis.
Choroid Epithelial Cells Maintained By Genetic Networks
 The vital choroid epithelial cells come from neuro-epithelial stem cells. The tissue stroma are derived from other mesenchymal lines. The stem cells that make choroid cells suppress their own production of neurons by multiple specific interacting genetic networks and a large number of different counteracting transcription factors.
The vital choroid epithelial cells come from neuro-epithelial stem cells. The tissue stroma are derived from other mesenchymal lines. The stem cells that make choroid cells suppress their own production of neurons by multiple specific interacting genetic networks and a large number of different counteracting transcription factors.
Complex feedback loops determine how many neurons versus how many choroid cells are produced. This complex process uses different mechanisms and different lineages in each of the ventricles.
Like neurons, active genetic signaling is needed to maintain the status of these very important and intelligent cells. See the post on how complex genetic networks in exact timing sequences are necessary for neurons to be maintained on a daily basis for their entire long lifetime.
A malfunctioning choroid plexus can cause many diseases. It can have cysts, bleeding, increased tissue and tumors. Blockage of the fluid can cause hydrocephalus, where too much fluid pressure kills brain cells. The blood has three carriers for thyroid hormone, but CSF and choroid have one. Therefore, an abnormality of this one carrier can cause decreased thyroid and brain disease. The choroid plexus clears out amyloid and other mis folded proteins by at least three different mechanisms. In Alzheimer’s the choroid shrinks and is less efficient at this clearance, which occurs by direct transport, direct absorption and special chaperone actions. Atrophy of the choroid, also, occurs in strokes, multiple sclerosis, and psychiatric diseases.
Stages of Choroid Epithelial Cells
 Choroid cells develop in a series of steps. The cells first appear without their final orientation in multiple uneven layers and shapes with the nucleus in the center and no special villi and fenestrations. Then, they change into columns with nuclei that are closer to the ventricles with more organized villi in each cell. They eventually become cubes with nuclei at the base near the tissue with much more complex multiple folds in the larger villi that make more surface area.
Choroid cells develop in a series of steps. The cells first appear without their final orientation in multiple uneven layers and shapes with the nucleus in the center and no special villi and fenestrations. Then, they change into columns with nuclei that are closer to the ventricles with more organized villi in each cell. They eventually become cubes with nuclei at the base near the tissue with much more complex multiple folds in the larger villi that make more surface area.
Nuclei move to the bottom away from the microvilli to make room for a large amount of transport machinery that is built during development. Also, the villi become more complex with more surface area. These changes are orchestrated by elaborate genetic network signaling, as well as the development of many more mitochondria and a much more elaborate endoplasmic reticulum. There are no organelles in the microvilli. In humans, the many microvilli increase the surface area to 200 square centimeters, able produce the large amount of fluid each day. With age, microvilli become smaller and there is less CSF.
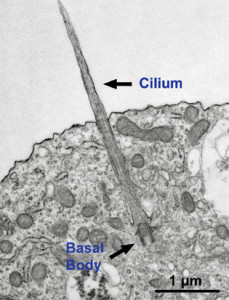 Each choroid cell has multiple cilia with complex microtubule structures. A previous post described the critical functions of cilia in all cells, but especially cells in the human brain. It is commonly known that cilia are used by cells to either travel or to move fluid and they do accomplish this in the choroid.
Each choroid cell has multiple cilia with complex microtubule structures. A previous post described the critical functions of cilia in all cells, but especially cells in the human brain. It is commonly known that cilia are used by cells to either travel or to move fluid and they do accomplish this in the choroid.
The primary cilia, however, is a very complex structure (see post primary cilium) that is an antenna and cognitive signaling center for the cell, as well as helping the flow of the fluid in the ventricles. Primary cilium have vast amounts of receptors and send a wide range of signals. Specialized receptors exist in the primary cilia in the eye and ear and other places. In the choroid these unique cilia have unique abilities to sense chemicals and osmolarity in the CSF fluid.
Choroid Cells Communicate with Blood and Tissue Cells
The one epithelial cell keeps up a two way signaling dialogue with the tissue (stromal cells from a different mesenchymal lineage), just as the one layered epithelial cells in the intestine. These stromal cells have unique features as well, different from other cells in their lineage, including fenestrated capillaries. These lining cells of the small blood vessels have a special diaphragm that allows water and small molecules to cross out of the blood.
It is not at all understood how two independent lines of cells in the fetus closely coordinate their development. Like the outcome of the choroid plexus, this is a close collaboration of the brain and the body outside the brain. In the fetus, epithelial cells form sheets that cover the large amount of developing blood vessels and make close contact from early in development. They appear to produce factors regulating each other. Epithelial cells produce molecules that stimulate blood cells. Other signals control the size of the vessels and placement of the fenestrations. Three types of cells signal about further development—epithelial, stromal and blood vessel.
Choroid’s Elaborate Functions
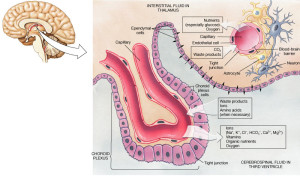 The plexus produces the cerebral spinal fluid and has special transport and barrier functions to determine what is transported between blood and CSF. There are special transporters for ions at both top and bottom (apical and basal) of the epithelial cells. Aquaporins allow water from blood based on osmotic gradients. Other specialized transport occurs for proteins, nutrients and molecules for metabolism. One particular complex carbohydrate, myo inositol, is metabolized into various signaling molecules and has a special transport system that is highly regulated by potassium channels (see post on complexity of potassium channels). Abnormal myo-inositol can cause significant brain and behavioral problems.
The plexus produces the cerebral spinal fluid and has special transport and barrier functions to determine what is transported between blood and CSF. There are special transporters for ions at both top and bottom (apical and basal) of the epithelial cells. Aquaporins allow water from blood based on osmotic gradients. Other specialized transport occurs for proteins, nutrients and molecules for metabolism. One particular complex carbohydrate, myo inositol, is metabolized into various signaling molecules and has a special transport system that is highly regulated by potassium channels (see post on complexity of potassium channels). Abnormal myo-inositol can cause significant brain and behavioral problems.
Choroid cells make special proteins and secrete them into the CSF. Special thyroid transporters are manufactured. Proteins are transported in exosomes (see post on exosomes); others interact with special proteins in the membrane. Vesicles transport critical receptor proteins and small RNAs including microRNAs that stimulate and repress neural stem cells.
The CSF contains many different types of molecules. As well as proteins, it has lipids, hormones, microRNAs, cholesterol, metabolites and others. Cells in the cerebral cortex have long primary cilium that extend into the CSF with special receptors. Signals alert cortical stem cells to either divide frequently or not. In fact, the CSF has a very large amount of molecules that are just being discovered that have very significant effects related to development all over the brain. The construction of any region of the brain is dependent upon cross talk from multiple sources through the CSF. The choroid is really a critical wireless signaling center for the entire brain with the choroid plexus as the conductor.
CSF Molecular Gradients for Elaborate Brain Communication
The choroid responds instantly to situations in all parts of the brain calling for specialized immune cells. It has not been clear how this can happen. Recently, elaborate communication has been discovered, with cytokines and using molecular gradients throughout the CSF. These signals and gradients provide rapid elaborate communication from all parts of the brain to and from the choroid.
It was recently discovered that the protein makeup of the lateral and fourth ventricles are different. In fact, animal studies showed that two different choroid plexus regions have completely different tissues with different genetic networks operating and different functions, even though they look similar under a microscope. Each region has very different signaling cascades operating. There are more than 200 entirely different proteins in these two regions. The composition is, also, different at different ages.
Perhaps most significantly, gradients were discovered within each ventricle and between the various ventricles. Gradients have been found in the cerebellum that are critical to its construction.
Choroid Stimulates Neuroplasticity and New Neurons in Sub Ventricular Zone
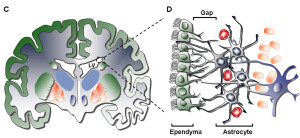
It has recently become clear that the choroid plexus is a major determinant of neuroplasticity and the production of new neurons in both the ventricular sub ventricular zone (V-SVZ zone) and the sub granular zone of the hippocampus. These are the two large areas already known to make substantial amounts of neurons each day for learning and memory.
The V-SVZ zone is built as circular centers with the stem cells in the middle of the circle and ependymal epithelial cells radiating from the center like a pinwheel. The ependymal cells have multiple cilia for movement. The stem cells in the middle have large primary cilia (see post) that extend into the CSF to receive and send signals from the fluid molecules and gradients. At the bottom of the cell, the cell hugs the blood vessels for other signals from the blood.
The center stem cells make new baby neurons (immature precursors), which become neuroblasts (young neurons). Signaling instructs the neurogenesis process via CSF to the cilium and the blood to the cell from the basal contact region. This unique structure allows the creation of the new neurons that become new memories (see post on how new neurons restructure memory). It is supported by multiple different cytokines that stimulate production of adhesion molecules for the cellular movement and migration.
Some cytokines are necessary to maintain this unique niche where the neurons are created. Other neurotrophin molecules support the creation of the new neurons. Signals create repulsion to makes the new neurons start their migration to the hippocampus where they will become new brain cells involved in new memory circuits. Another set of forces sends neurons along the rostral migratory stream to the olfactory system. All of this activity depends on thousands of different regulated gene networks.
Signaling related to this creation of new neurons occurs from many different parts of the brain. What is very interesting about the choroid system is that for any event in the brain where help is needed, regardless how far it is, the choroid will immediately adjust and send signals to call for specific immune cells. This far-flung communication that appears almost instantaneously uses cytokine signaling through the CSF and the blood stream at the same time to reach any spot in the brain.
Other effects of the signaling at the choroid and the CSF regulate critical periods in the brain development, such as the development of binocular vision through regulation of the interneurons. It is critical for the creation of the map of sensory inputs.
Choroid Part of Brain Cleaning Apparatus
As well as secreting signals, the choroid is vital for clearing poisons and debris from the brain such as mis folded proteins that are part of neurodegenerative diseases. The choroid is part of the newly discovered “glymphatic” (for glia lymphatic) system that cleans the brain. Astrocytes are critical to the system and have end feet controlling blood flow. But, they also use aquaporin 4 channels of the end feet to allow CSF fluid near blood vessels to flow in the brain extracellular space.
Pulsation from arteries contributes to convective flow of CSF and interstitial fluid. Waste is pushed through interstitial space along the astrocytes. A recent study showed that during sleep, neurons shrink by 40%, increasing the flow and making the process much more effective at removing waste. This flow goes to near the venous system and eventually enters into the blood for removal. Between 50% and 80% of random proteins and other molecules in solution are cleared by this method from the extracellular space. The removal of the mis folded amyloid and other proteins are very significant to avoid dementia.
The Very Intelligent Choroid Plexus Epithelial Cell
 The choroid plexus is the center of activity between the nervous and immune systems—the wired and wireless aspects of brain. The choroid cells secrete hundreds of very specific factors. They alone determine which cells will enter the brain and call for the specific types needed for each situation in any region.
The choroid plexus is the center of activity between the nervous and immune systems—the wired and wireless aspects of brain. The choroid cells secrete hundreds of very specific factors. They alone determine which cells will enter the brain and call for the specific types needed for each situation in any region.
The choroid plexus with its single layer of extremely intelligent epithelial cells determines what molecules go in and out of the brain. Each cell communicates constantly with many brain and immune cells using cytokines and the unique molecular gradients of the cerebrospinal fluid.
This one cell is the window between the brain and the immune systems. Previous posts have described very intelligent behavior for T cells (T Cells and Cognitive Function) and microglia (Are Microglia the Most Intelligent Brain Cell). The choroid epithelial cell orchestrates the actions of both of these very remarkable cells. How can anyone not be impressed by the intelligence of this single choroid plexus epithelial cell?