 Even after thousands of studies, the genetic basis of psychiatric illness is not at all clear. The relationship of mental events and genetic networks is extremely complex. A previous post discussed social interactions triggering genetic networks. Genetic networks are triggered by mental events and a vast number of other scales simultaneously.
Even after thousands of studies, the genetic basis of psychiatric illness is not at all clear. The relationship of mental events and genetic networks is extremely complex. A previous post discussed social interactions triggering genetic networks. Genetic networks are triggered by mental events and a vast number of other scales simultaneously.
Many studies have demonstrated that mental illness does occur more frequently in families and that there is more illness the closer relatives are to patients. One major problem in finding genetic links to psychiatric illness is that diagnoses are inexact and not based on biological markers, but on clinical syndromes—lists of symptoms. Each diagnosis probably represents different subtypes that are not yet understood. For example, if depression is diagnosed by four of eight specific symptoms, there are many different combinations of four that can occur. See a previous post discussing one subtype of depression related to inflammation biomarkers. Also, see previous post on use of MRIs to describe mental events for more information on the lack of biological markers in psychiatry.
 It has become clearer in recent years that many illnesses do not occur because of simple mutations in genes; genes are the DNA that codes for specific proteins, which makes up 1.5% of the total DNA. The large international study called ENCODE (see post on Encyclopedia of DNA Elements) demonstrated that the 98.5% of the DNA that does not code for proteins is quite complex. At least 20% (thirty times more than the coding genes) produce regulatory RNAs of various types—small and large non-coding RNAs. But, this number could be as high as 50%.
It has become clearer in recent years that many illnesses do not occur because of simple mutations in genes; genes are the DNA that codes for specific proteins, which makes up 1.5% of the total DNA. The large international study called ENCODE (see post on Encyclopedia of DNA Elements) demonstrated that the 98.5% of the DNA that does not code for proteins is quite complex. At least 20% (thirty times more than the coding genes) produce regulatory RNAs of various types—small and large non-coding RNAs. But, this number could be as high as 50%.
The other 50% of the total DNA is made up of jumping genes, from viruses and other sources that constantly try to move, multiply and alter chromosomes. Cells must control jumping genes through complex intra nuclear immune processes. Jumping genes have been critical in producing new strands of usable proteins in evolution. There are many reasons for the limitations of genetic research in psychiatric illness.
Complexity of Genetic Networks
 The complexity of the genetic processes is much greater than previously thought. Mutations are most often in the large regions of regulatory DNA. Regulatory RNAs produced in one spot can, in fact, influence many different genes in different location and in different ways. Studies with cancer show that not one, but many mutations are necessary to create specific cancerous cells. And these mutations are different in each individual, which makes treating cancer much more complex. This same situation may be occurring with psychiatric illness where multiple different mutations are needed to produce an illness and each case might be different.
The complexity of the genetic processes is much greater than previously thought. Mutations are most often in the large regions of regulatory DNA. Regulatory RNAs produced in one spot can, in fact, influence many different genes in different location and in different ways. Studies with cancer show that not one, but many mutations are necessary to create specific cancerous cells. And these mutations are different in each individual, which makes treating cancer much more complex. This same situation may be occurring with psychiatric illness where multiple different mutations are needed to produce an illness and each case might be different.
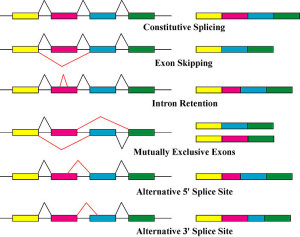
Even more significant, perhaps, is the fact that genetic networks have multiple layers of regulation that are only vaguely understood. These layers all operate simultaneously and interact in unknown ways. The shapes of DNA have great impact. The place of the DNA in the nucleus is a factor. Tags on both the histones and the DNA itself are critical in determining which DNA can be used in specific circumstances. Many different epigenetic tags have now been described for both histones and DNA. Research shows that alternative RNA splicing is greatly enhanced in humans and in particular in the human brain. It is now clear that RNA alternative splicing produces a large number of unique proteins that have been instrumental in development of the human brain. Also, there are many thousands of other factors, both proteins and RNAs that regulate all aspects of the DNA transcription and RNA translation process.
These are some of the reasons why there is still very little known about the genetics of psychiatric illness. What follows is a summary of what is known.
Mutations Causing Psychiatric Illness
 When studying the genome for specific diseases, some mutations have been found in specific genes (1.5% of the DNA) but this is rare. Most mutations are in the vast regulatory regions. Surprisingly, as more people’s DNA is decoded, it is found that each individual has many very unique mutations. Most of these mutations don’t have any apparent impact. At the same time with psychiatric illness there are a large number of different mutations in people with the same illness. There are small groups with slight effect and very rare mutations with much greater affect. Each illness appears to have great variation and much overlap, as well. The recent studies are humbling because of how little we can learn from them about the great complexity of brain circuits and functions.
When studying the genome for specific diseases, some mutations have been found in specific genes (1.5% of the DNA) but this is rare. Most mutations are in the vast regulatory regions. Surprisingly, as more people’s DNA is decoded, it is found that each individual has many very unique mutations. Most of these mutations don’t have any apparent impact. At the same time with psychiatric illness there are a large number of different mutations in people with the same illness. There are small groups with slight effect and very rare mutations with much greater affect. Each illness appears to have great variation and much overlap, as well. The recent studies are humbling because of how little we can learn from them about the great complexity of brain circuits and functions.
Since the causes of mental illness are not known and for a long time were not considered biological, but rather “mental,” these genetic studies do not yet answer the questions of definitions for specific diagnoses. Further, there is clinical and genetic overlap between the major categories of depression, bipolar and schizophrenia. However, there are now genetic clusters that are definitely associated with schizophrenia, autism spectrum, major depression and bipolar disorder. And there is evidence that these illnesses are at least partially hereditary.
GWAS
The recent technique for comparing large numbers of people’s gene differences is called GWAS (genome-wide association studies). GWAS compare individual nucleotide variations by computer among thousands of people. When many people (perhaps 5% of the population) have the same variation, they are called common alleles or common variants.
The great majority of variants found in GWAS are in regulatory DNA that can influence many different genes. The complexity of the regulatory region is barely understood; for a long time, it was considered “junk DNA”. “Junk” referred to DNA that is randomly gathered from genetic processes, rather than a specific code for a specific RNA purpose. Now much more of the DNA is found to not be junk. But, little is understood about these many thousands of regulatory small and large RNAs (see post), GWAS studies often assume that if a regulatory DNA strand is near a gene then it has something to do with it. Unfortunately, this assumption used for calculations is not always the case. Regulatory DNA, particularly large non coding RNAs (lncRNA), can affect entire regions of a chromosome.
There are very rare variations, called “copy number variants” or CNVs that have been found to be related to autism and schizophrenia.
Another new technology codes all of the proteins for a given individual, called whole exome sequencing or WES. This has been accomplished for thousands of people. There are now, also, altered proteins found by this technique for some autism and schizophrenia.
The amount that an illness is inherited is estimated by the number of these specific variants that exist in the population of ill people. This, of course, is based on our current very incomplete knowledge and assumptions. It is not known whether inheritance is based on many small variants or several large variants. With this WES analysis, it is now understood that the genetic effects causing psychiatric disease involve a very large number of small variations, each with very little effect. This makes everything much more complex. This is, also, the case for other major common complex diseases. If these diseases have greater heritability, then it would be based on mutations or factors that are not known.
It must be understood that mutations are only one way that genetic networks trigger major changes. Previous posts have described the critical effect on the development of alternate splicing on the human brain. Also, epigenetic markings on the histones and the DNA itself have major impact.
Another complication is that as more people with an illness are studied, such as anxiety and depression, more variations and subtypes in symptoms and situations are included in the data and the less specific the findings are.
Rare CNVs account for a very small percentage of autism cases. Several major variants are related to increased risk of schizophrenia but some of these are, also, related to autism, epilepsy and developmental intellectual disability. There are few in bipolar, depression, obsessive-compulsive disorder, attention deficit and anxiety. Some people have gone as far as saying that anxiety and depression may cause OCD-like behaviors. It is not yet known if there is an actual link between these factors, but there are ways to help treat people who may display these symptoms. Have a look at this information provided by The Renewed Freedom Center to learn more about this potential correlation and what types of treatments you can take to help you feel better.
Genetic Studies That Provide Some Information
 Studies of schizophrenia have come closest to finding specific genes. Early studies showed some possibilities, but the recent larger research did not.
Studies of schizophrenia have come closest to finding specific genes. Early studies showed some possibilities, but the recent larger research did not.
Very rare variants have been found in Tourette syndrome, with heritability of 21%, but this was completely unrelated to obsessive compulsive disorder, which was surprising at the time. In Tourette studies, one family has a rare dominant mutation, but in all other cases it was multiple small foci.
Even though drug abuse, anxiety and depression appear to have a strong inherited component, there have been no major variants found.
In autism studies, the particular group of co regulated genes implies that variants are related to the structure of the chromatin, the particular protein inhibitors and stimulators and the effects of neurogenesis. These are all vastly complicated subjects that don’t supply understandable genetic correlations.
Overlap in Diagnoses and Genetics
 Although the specific variants are not clear, studies have determined that there is overlap in the risk for specific illnesses, for example bipolar disorder and schizophrenia. This is not surprising, since there are many patients in that overlapping area that are called schizoaffective because they cannot be defined clearly in one group or the other and have characteristics of both illnesses. This overlap is noted in both the analysis of the multiple small loci and all of the GWAS studies.
Although the specific variants are not clear, studies have determined that there is overlap in the risk for specific illnesses, for example bipolar disorder and schizophrenia. This is not surprising, since there are many patients in that overlapping area that are called schizoaffective because they cannot be defined clearly in one group or the other and have characteristics of both illnesses. This overlap is noted in both the analysis of the multiple small loci and all of the GWAS studies.
Another source of overlap in the genetic risk is found in mutations in genes expressed in the fetus for Fragile X syndrome of mental retardation, autism spectrum disorder, developmental intellectual disability, schizophrenia and epilepsy. In these illnesses there are no real specific rare large CNVs. This research implies that it is possible that there are major large-scale mutations (unknown) that affect the very specific and extremely complex processes of brain development that then express these very different illnesses.
The variations that have been found do not just overlap with different disease states, but they overlap with normal people as well. In fact, very recent studies are finding a very large number of unique alleles in normal people that do not result in any known disease. One dramatic example of overlap with normal people, for example, finds a cluster of multiple variants that overlaps with bipolar disorder and schizophrenia on the one hand and creative normal on the other.
It has not been possible to find clusters that affect family members and patients that are below the amount needed to stimulate the illness. Even analyzing only very severe illness is not helpful in all cases. These associated normal variants appear as complex in their own way as the genetic information of the illnesses.
Animal models are being used to study many psychiatric and neurological illnesses, but their value has been questioned. A model of autism spectrum in mice has shown some promise. Some studies have been attempting to study neurons in culture, but this is complicated by the fact that there are thousands of different species of neurons.
Current Genetic Evidence
 There appears to be thousands of genes related to each disorder. If there are a large number of subtypes or individual unique versions of an illness, then in depth study will have to be done for each small group. Autism does appear to be a large group of many very rare diseases. Different rare variants appear to be prominent in small groups. This does not seem to be the case with most of the other illnesses.
There appears to be thousands of genes related to each disorder. If there are a large number of subtypes or individual unique versions of an illness, then in depth study will have to be done for each small group. Autism does appear to be a large group of many very rare diseases. Different rare variants appear to be prominent in small groups. This does not seem to be the case with most of the other illnesses.
There is no evidence of particular individual major genetic variants for most of the other diseases—schizophrenia, bipolar, substance abuse, anxiety and depression.
Many psychiatric diagnoses have been influenced by the fact that certain drugs are effective in treatment, such as antidepressants. But, in fact specific drugs may work in only 30 to 40% of people. Each drug may work in different subgroups, but there aren’t enough studies comparing drugs to know. There are a substantial number of resistant cases to all current medications that may be different subtypes. Also, there are many diseases where medications help multiple different subtypes—examples are pain medications and anti inflammation medications.
Do different clusters of genetic variants all affect particular switches in the brain and cause the illness as a final common pathway? This would mean that a particular molecule or gene might not be the way that psychiatric diseases can be studied. Genes operate in extremely complex networks of many levels of factors including a vast number of protein and RNA enhancers and inhibitors, stimulated by many variables such as metabolic sensors. Final common pathways could trigger epigenetic tags. Epigenetics now has discovered more than forty different types of tags for DNA and histones, not just the simple methyl and acetyl of a decade ago. The shape of the chromatin is now a new factor. Long non-coding RNAs have vast effects at great distances in the genome.
In autism and schizophrenia there is some evidence suggesting shared common final molecular pathways. In autism there are at least three pathways identified. One is the regulation of the development of the cortex and its specific networks of synapses. A second is the relationship of the structure of the chromatin (the protein structure that protects and regulates the operations of DNA—histone) to stimulation of the creation of new neurons in the hippocampus (see post). The third is regulation of the process where proteins are made from RNA, such as alternative splicing (see post). These various pathways may lead to the clinical findings of decreased or increased activity in particular brain circuits.
Evidence, also, shows that schizophrenia converges on the same pathways related to the fetal development of the cortex. However, these pathways are vast and complex and the particulars are not known.
Autism variants have been found in many rare genetic diseases, which, also, have pathology in unique sets of bodily organs. This is not the case in schizophrenia or bipolar disorder, although bipolar subtypes are triggered later in life with treatment of specific medical disorders and with particular types of medications.
Another major complication is that it is not known how to find all of the species of neurons, which now number in the thousands.
Perhaps the biggest problem is that there is little known about the function of the clusters of genes that have been identified. Each will take very intense research for many years. For comparison, the gene related to Huntington’s disease has been known for 25 years and the mechanism is not yet understood. Huntington’s is a clear case where one gene has great significance and it still can’t be figured out. In psychiatric illness, there are thousands of interacting genes, each one of which is not understood.
Limitations of Genetic Research in Psychiatric Illness
Two major obstacles hinder study of the genetics of psychiatric illness. The first is the lack of biological understanding of psychiatric diagnoses. Diagnoses are made by clinical syndromes (lists of related symptoms) not specific molecular biomarkers or brain regions noted by imaging. In fact, there are major issues in using imaging to describe mental events (see post on Limitations of MRIs for Understanding Behavior). The second major issue is the vast complexity of genetics, especially for the brain. Multiple layers of regulation are just being described. Mutations are more often in the vast regulatory regions, not the “genes”. But, even when finding mutations, most with illness have unique individual sets of mutations. It would take tens of thousands of patients to possibly identify correlations, which is not currently feasible.
While biological factors appear to be very significant in psychiatric illness, this is not the entire story. With increasing data showing that mind most probably interacts with the brain rather than the brain creating the mind, mental factors will again become a focus of diagnosis and treatment. In fact, a large recent study of treatment for schizophrenia shows that psychotherapy is critically important, not just medications. This study showed that better treatment outcomes did occur with substantial psychotherapies as part of the treatment. This is really not news for those in psychiatry. Psychotherapy was known to be very effective in helping a wide variety of patients in the years before the modern development of medications; but many of these patients, also, needed medications. When modern medications were available, psychotherapy was eliminated in the treatment of schizophrenia and diminished in many other illnesses, mainly for financial reasons. The combination is definitely better that either alone.
The same vast complexity that is found in all aspects of neuroscience is present in attempting to find biological descriptions of psychiatric illness and its genetic underpinnings. Modern research is now more consistent with mind interacting with organs, cells, organelles and molecules. This research implies that in the future the pendulum in psychiatry will swing back to include both molecular biology of the brain and its interactions with mind.

