 New research on electrical synapses greatly complicates plans to map the brain. Recent findings show electrical synapses are critical throughout the brain and interact in complex ways with chemical synapses making the function of the brain much more convoluted. Almost all of the current research on synapses and brain wiring has been on chemical synapses. Now, electrical synapses are shown to be critical in initial wiring of the fetal brain, in repairing neurons, and in synchronous oscillations, among many other critical functions. In fact, chemical synapses cannot survive without them and electrical synapses are critical for chemical synapse function.
New research on electrical synapses greatly complicates plans to map the brain. Recent findings show electrical synapses are critical throughout the brain and interact in complex ways with chemical synapses making the function of the brain much more convoluted. Almost all of the current research on synapses and brain wiring has been on chemical synapses. Now, electrical synapses are shown to be critical in initial wiring of the fetal brain, in repairing neurons, and in synchronous oscillations, among many other critical functions. In fact, chemical synapses cannot survive without them and electrical synapses are critical for chemical synapse function.
 Previous posts described other forms of brain electrical activity, such as synchronous waves and local field potentials in the regions surrounding neurons. In addition, a post showed that astrocyte networks are critical for chemical synapses. But, recent findings on electrical synapses demonstrate that they are, in fact, equally important as chemical synapses in brain development and adult brain function. This post will discuss the recent data that shows how much electrical synapses greatly complicate our understanding of brain function.
Previous posts described other forms of brain electrical activity, such as synchronous waves and local field potentials in the regions surrounding neurons. In addition, a post showed that astrocyte networks are critical for chemical synapses. But, recent findings on electrical synapses demonstrate that they are, in fact, equally important as chemical synapses in brain development and adult brain function. This post will discuss the recent data that shows how much electrical synapses greatly complicate our understanding of brain function.
Communication between Neurons has become More Complex
 Most research into brain structure focuses on synapses where neurons rapidly communicate with each other with electrical signals along the axon triggering vesicles with specific neurotransmitters that convey a specific meaning to the next neuron. In the other, less researched type, the electrical synapse directly transmits electricity through the synapse to the next cell by way of a cluster of channels between the two cells. In fact, these two forms of synapse communication are absolutely necessary for each to work in all brain regions.
Most research into brain structure focuses on synapses where neurons rapidly communicate with each other with electrical signals along the axon triggering vesicles with specific neurotransmitters that convey a specific meaning to the next neuron. In the other, less researched type, the electrical synapse directly transmits electricity through the synapse to the next cell by way of a cluster of channels between the two cells. In fact, these two forms of synapse communication are absolutely necessary for each to work in all brain regions.
Many Forms of Transmission in the Brain
There have been a variety of new forms of information transmission described but it is too early to tell how significant they are. These include diffusion of neurotransmitters in extra cellular space, exosome vesicles transported from one cell to another and cell-to-cell nanotubes. Electricity in the brain can come from many different sources (see post on electricity in the brain.) Electrical fields are described around the neuron and in the intracellular space that influence nearby neurons or entire groups of neurons. Yet another is with synchronous brain waves.
Oscillations
 Studies on brain waves demonstrate that neurons in two regions oscillate with synchronous beta waves describing color and two different regions oscillate beta waves for spatial orientation. These regions are sharing information in the form of waves at the same time that chemical synapses are sharing information. Theta resonance has been described in some cortex regions. In contrast some inhibitory neurons have been noted to have a resonance in other ranges.
Studies on brain waves demonstrate that neurons in two regions oscillate with synchronous beta waves describing color and two different regions oscillate beta waves for spatial orientation. These regions are sharing information in the form of waves at the same time that chemical synapses are sharing information. Theta resonance has been described in some cortex regions. In contrast some inhibitory neurons have been noted to have a resonance in other ranges.
Local Field Potential
 All electrical currents in a section of the brain combine to form the voltage potential that is the sum of all the activity. This activity is different at each point and forms an electrical field. The local field potential, LFP, is the sum of all electricity in a region of extracellular space. A previous post described that extracellular gradients can, also, correlate with mental states.
All electrical currents in a section of the brain combine to form the voltage potential that is the sum of all the activity. This activity is different at each point and forms an electrical field. The local field potential, LFP, is the sum of all electricity in a region of extracellular space. A previous post described that extracellular gradients can, also, correlate with mental states.
Electrical currents contribute to LFP from synapses, calcium spikes, action potentials, and after potential of the spike.
 Most importantly, the specific shapes of neurons and the rhythmic timing affect these currents. The common pyramidal cell in parallel columns has very thick dendrites creating strong flow of ions in the extracellular space, which are additive. Symmetrical neurons in the thalamus and cortex have equal sized dendrites and electricity is not additive. Folding of the cortical gyrus creates different electrical effects. Inhibitory neurons create gradients between the cell body and dendrite.
Most importantly, the specific shapes of neurons and the rhythmic timing affect these currents. The common pyramidal cell in parallel columns has very thick dendrites creating strong flow of ions in the extracellular space, which are additive. Symmetrical neurons in the thalamus and cortex have equal sized dendrites and electricity is not additive. Folding of the cortical gyrus creates different electrical effects. Inhibitory neurons create gradients between the cell body and dendrite.
Electrical Synapse History
Recent findings suggest that of all these new mechanisms discovered, electrical synapses are equal to chemical synapses in importance and sophistication.
Since it is more difficult to study electrical transmission than chemical, it was thought that it occurred mainly in cold-blooded vertebrates, not mammals. Now it is clear chemical and electrical synapses exist throughout the human brain. They were first described in the retina, olive and olfactory bulb, but now everywhere. The very significant inhibitory neurons, critical for cognition, are prominent users of electrical synapses. But, even more important is that it now appears that both types of synapses are necessary for the function of either.
In evolution, chemical transmission came before electrical communication. Chemical signals are widely used between cells in any community of bacteria or multi cellular organism. It is interesting to note that in evolution invertebrates developed identical structures (innexins and pannexins) from entirely different sets of genes.
Connexin Channels in Clusters Form Gap Junctions
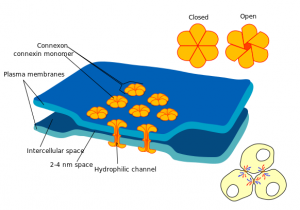 Electrical signaling uses gap junctions, a structure made of many clustered channels connecting the two cells. This allows electrical signals to flow both ways between the two coupled neurons. It also allows some small molecules such as calcium and cyclic AMP to flow through.
Electrical signaling uses gap junctions, a structure made of many clustered channels connecting the two cells. This allows electrical signals to flow both ways between the two coupled neurons. It also allows some small molecules such as calcium and cyclic AMP to flow through.
The gap junction is very small, 3 nanometers, (synapses that use neurotransmitters usually are 20 to 40 nanometers) and is very fast and bidirectional. These synapses don’t amplify the signal the way chemical synapses may with addition of multiple inputs. With the very rapid response electric synapses are often found in loops for rapidly needed defensive measures. Electrical synapses, also, trigger synchronous waves and can cause increase in the currents of the local field potential in the extracellular medium.
Two connexin six part molecules, one from each cell, form the channel. Vertebrate neurons make five known connexins from a family of 20 genes. The retina is the most studied junction gap. The most widely used connexin is CX36.
The critical feature of the gap junction is that the electrical signal goes both ways. This allows many neurons to be connected in a large system. These neurons can share electricity without action potential spikes from the axon. Gap junctions connect a large group of neurons together and couple them as they accumulate depolarizations that are not sufficient for an action potential. This allows all the coupled neurons to fire at one time in 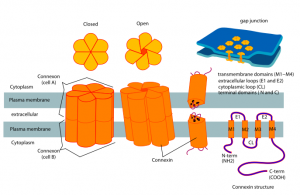 synchronous firing.
synchronous firing.
Another unique property is lateral excitation — the ability of sensory neurons to excite many of its neighbors allowing a stronger signal and more sensitivity to sensory input.
Escape Response: These lateral gap junctions form specific small networks of neurons that consist of both motor and sensory neurons and are used for very important single movements, often a critical escape mechanism. One example is the tail-flip escape response of a fish. In humans there are cerebellar interneuron networks that spread responses rapidly through gap junctions. These transmissions have no delay and are very reliable.
Neuroplasticity of Electrical Synapses
Electrical synapses, also, show neuroplasticity; they use very complex, dynamic mechanisms that are constantly remodeling and replacing connexin channel subunits. The strength of the channel is altered by neuroplastic mechanisms  much as the subunits and receptors are altered in chemical synapses. (see neuroplasticity post). Just as glutamate receptors are manufactured and altered to strengthen synapses, connexins also are rebuilt in a neuroplasticity mechanism. There are also connexin modulators, such as dopamine, that influence gab junction neuroplasticity.
much as the subunits and receptors are altered in chemical synapses. (see neuroplasticity post). Just as glutamate receptors are manufactured and altered to strengthen synapses, connexins also are rebuilt in a neuroplasticity mechanism. There are also connexin modulators, such as dopamine, that influence gab junction neuroplasticity.
In chemical synapses there are many scaffolding proteins, including the extremely complex post-synaptic density consisting of 1000 interlocking proteins that are critical for neuroplasticity. There are similar multi protein scaffolding structures in each cell that support the connexin channel and its modifications. These mechanisms are very complex and just now being discovered.
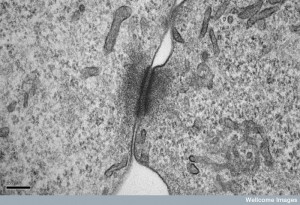
Large complex proteins help create and insert the pieces for the new connexin channels. It is now known that there are many different types of structures in gap junctions, and both sides of the connection can be different. Different properties of the channels are related to two different connexin structures coming from the two different cells. The different structures can determine which way the electrical signals will flow for that message and that connection. This differential flow helps groups of sensory neurons to cooperate, as in the auditory system.
The complex protein structure, which is similar to the post-synaptic density of chemical transmission, is called the semi-dense cytoplasmic matrix. This matrix adds molecules to the channels, which also is a way of modifying its communication.
Electrical Synapses in the Fetal Brain
Gap junction electrical transmission is very important in early fetal development as a way of forming networks of cooperating and coordinated  activity in neurons for the developing brain. The pattern of electrical synapses form an architectural blue print for the later development of neuronal networks of chemical synapses. The electrical synapses, also, aid migration and circuit formation and the pruning of unnecessary chemical synapses. These transient electrical synapses are prominent and give way to the more prominent chemical synapses in later brain development. Gap junctions are critical for formation of asymmetrical brain structures in the embryo. During brain development before birth, electrical synapses, also, use molecules to communicate among neurons.
activity in neurons for the developing brain. The pattern of electrical synapses form an architectural blue print for the later development of neuronal networks of chemical synapses. The electrical synapses, also, aid migration and circuit formation and the pruning of unnecessary chemical synapses. These transient electrical synapses are prominent and give way to the more prominent chemical synapses in later brain development. Gap junctions are critical for formation of asymmetrical brain structures in the embryo. During brain development before birth, electrical synapses, also, use molecules to communicate among neurons.
As the spinal cord develops, lumbar motor neurons communicate with gap junctions, leading to synchronous activity with multiple neurons as the motor system develops into a single neuronal system for each muscle by neurons competing for the job. Gap junctions are critical for the transition from multiple to single fiber control of muscles.
In fact, the gap junction molecules are needed for the later development of almost all networks of chemical synapses. As the coupling decreases in the motor neurons, glutamate synapses form. As movement starts and glutamate neurons operate, the gap junctions in these motor neurons decrease.
Similarly, in the olfactory system and the neo-cortex, gap junctions are critical for development. In the development of the cortex, excitatory neurons are created in columns, where nearby cells are first connected with electrical synapses to hold them in place. As the structure solidifies with electrical synapses, then specific chemical synapses appear in the correct column circuits.
Later, in the hippocampus, glutamate LTP also needs the electrical synapses to function.
While electrical synapses are necessary for the development of chemical synapses, the opposite is also true. Structures from future chemical synapses have a great influence on the development of the first electrical synapses. Even before the chemical connections are formed, receptors later used in chemical transmission are needed to form the electrical synapses. An interplay of GABA and glutamate receptors is necessary to form electrical synapses. Later, more glutamate activity reduces electrical coupling that formed much of the structure. It appears that glutamate NMDA activity regulates electrical junctions.
Electrical Synapses in Adults
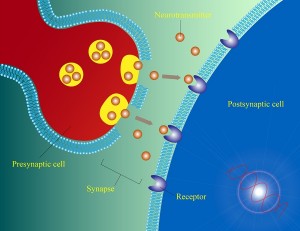 Neurotransmitters are secreted from presynaptic neurons into the synapse space and then bind to receptors in the postsynaptic neuron, which produces a cascade of signaling in the post synaptic cell. A nearby cell can release other neuromodulators, such as dopamine, that alters the strength and transmission that that synapse.
Neurotransmitters are secreted from presynaptic neurons into the synapse space and then bind to receptors in the postsynaptic neuron, which produces a cascade of signaling in the post synaptic cell. A nearby cell can release other neuromodulators, such as dopamine, that alters the strength and transmission that that synapse.
These modulators also affect electrical synapses on those two cells. The effect of dopamine can either be to increase or decrease the electrical synapse coupling through D1 and D5 receptors. The mechanisms are phosphorylation and de-phosphorylation of connexin molecules. Other systems that regulate electrical coupling have been found using norepinephrine, serotonin, histamine and nitric oxide. Some neurons have both chemical and electrical synapses in the same synapse region—called mixed synapses. In one system cannabinoid receptors determine the dopamine modulation of the electrical coupled neurons.
Electrical Synapses In the Eye and Ear
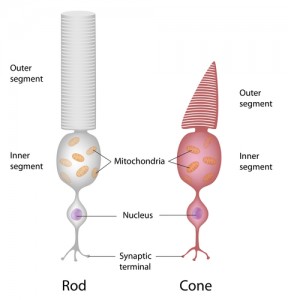 In rod and cone neurons of the eye, dopamine stimulates D2 receptors, which increases electrical coupling between both types of sensory cells. In another system, rods and cones send information to bipolar cells, the rod using electrical synapses, regulated by dopamine and nitric oxide. The retinal circuits are altered by the electrical synapses.
In rod and cone neurons of the eye, dopamine stimulates D2 receptors, which increases electrical coupling between both types of sensory cells. In another system, rods and cones send information to bipolar cells, the rod using electrical synapses, regulated by dopamine and nitric oxide. The retinal circuits are altered by the electrical synapses.
Another mechanism for rapid chemical transmission of amino acids in the auditory system uses mixed synapses. Chemical and electrical synapses in the high frequency range (500Hz) stimulate neuroplasticity and long term potentiation. Both the glutamate and electrical synapses are affected by this neuroplasticity. It appears, also, that the glutamate along with an increase in calcium stimulates the electrical neuroplasticity. An important protein, CaMKII, which is found in the post synaptic density, is critical for the alterations that occur in the connexin electrical channels and appears to have many different effects.
 Recent research shows that the interaction of chemical and electrical synapses occur throughout the brain in different ways. A single neuron can have different types of electrical synapses, making this much more complex. For example in the inferior olive, each cell can couple with forty other cells in different ways.
Recent research shows that the interaction of chemical and electrical synapses occur throughout the brain in different ways. A single neuron can have different types of electrical synapses, making this much more complex. For example in the inferior olive, each cell can couple with forty other cells in different ways.
Another critical function of electric synapses is producing synchronous firing of large groups of coupled neurons. This mechanism allows the post synaptic densities of a particular glutamate neuron to alter and modulate electrical coupling throughout a network of neurons that are all coupled. This produces synchronous firing of the neuronal network.
Electrical Synapses In Inhibitory Neurons
An entirely different mechanism operates with inhibitory neurons. Here the cell increases the electrical conductance near the synapse. This alteration in the electrical properties of the region near the synapse circumvents the electrical currents of the gap junctions. This mechanism can greatly alter and diminish coupling by “shunting” the electrical currents before they can go through the gap junction. Both chemical and electrical synapses, in fact, create GABA networks of interneurons critical for cognition. This electrical coupling appears to be related to synchronized gamma oscillations, that some believe are critical for consciousness, but also occur in some diseases.
Electrical Synapses In Disease
Since electrical synapses are so critical to development of the entire nervous system, it is logical to consider that dysfunction in gap junctions might cause disease.
Alterations in connexins have been associated with juvenile epilepsy.
It was stated that electrical synapses are particularly important in interneurons in the neocortex, thalamus and hippocampus. They contribute to the gamma frequency synchronous oscillations that appear to be very important for consciousness and for the bonding of neurons that creates memories (30 to 80 Hz). Alterations in these oscillations have been considered to be relevant in schizophrenia, Parkinson’s and autism.
After brain injury, the many electrical synapses from fetal development reappear and realign neuronal networks. After peripheral injury, spinal motor neurons are recoupled as they were in the fetus. It is possible that this stabilizes the situation until repair can occur by re connecting the synapses that were broken.
 In stroke caused by lack of blood flow and traumatic brain injury there are increased connexins and gap junctions. This change occurs 2 hours after the injury, stimulated by the large outpouring of glutamate in the damaged region. The interactions of the chemical and electrical synapses keep neurons from dying while repair is ongoing, (just as in the fetus).
In stroke caused by lack of blood flow and traumatic brain injury there are increased connexins and gap junctions. This change occurs 2 hours after the injury, stimulated by the large outpouring of glutamate in the damaged region. The interactions of the chemical and electrical synapses keep neurons from dying while repair is ongoing, (just as in the fetus).
But, it is also shown that gap junctions can cause negative effects in the damaged brain. In this situation there is rapid communication of the damaged nerves with the functioning neurons that are nearby possibly spreading the injury.
Electrical Synapses Are Critical for Chemical Synapse Function
Electrical synapses are very widespread in the brain. Mixed synapses of both chemical and electrical signals are critical to the function of each type of synapse. The faster electrical synapses are critical, as are the neuroplasticity triggered by the chemical synapses. Electrical synapses are especially common in the hippocampus including the inhibitory fiber dendrites.
Electrical synapses are critical for brain development and the creation of brain circuits. In the adult, they are able to alter the hard-wired networks of neurons. Just as the wiring of the brain with chemical based synapses is dependent on the structure created by the electrical synapses, so, also, glutamate chemical signaling modulates the increase and decrease of electrical synapses. After neuronal injury, electrical synapses appear and hold neuronal structures together while repair occurs in the chemical wiring synapses.
 We are just scratching the surface of the vast complexity of the living brain. Electricity and molecular interactions are both working together constantly in the spike along the axon with rapidly changing ion channels, in the electric current of the local field potential, in the flow of electricity through connexin channels, and in synchronous brain waves.
We are just scratching the surface of the vast complexity of the living brain. Electricity and molecular interactions are both working together constantly in the spike along the axon with rapidly changing ion channels, in the electric current of the local field potential, in the flow of electricity through connexin channels, and in synchronous brain waves.
Ions beget electric currents, which in turn stimulates chemical actions. We cannot separate chemicals and electricity or chemical synapses and electric synapses, just as we cannot separate matter and energy. Mental events have constant interplay with both.