 Up until recently, research into circadian rhythms has focused on central brain clocks that synchronize other cells. The three previous posts have described new research that each living cell has its own individual clocks based on genetic feedback loops combined with epigenetic loops. The three posts described the discovery of individual clocks, the relation to metabolic cycles in tissues and other cells, and the way immune cell cycles alter responses based on clocks. This fourth post in the series describes what is known about the central clocks and how they might be related to brain disease.
Up until recently, research into circadian rhythms has focused on central brain clocks that synchronize other cells. The three previous posts have described new research that each living cell has its own individual clocks based on genetic feedback loops combined with epigenetic loops. The three posts described the discovery of individual clocks, the relation to metabolic cycles in tissues and other cells, and the way immune cell cycles alter responses based on clocks. This fourth post in the series describes what is known about the central clocks and how they might be related to brain disease.
With aging, sleep becomes more erratic and circadian cycles can be disrupted. It is possible that circadian clock disruption can increase neurodegeneration. Also, the relationship of central brain rhythms to other tissues and cell rhythms are not totally understood but might be relevant to the question of brain degeneration.
 Ben Franklin noted that sleep routines are important for health—“early to bed, early to rise, makes a man healthy…” How important this type of regularity is to brain health is just being researched with the new circadian cellular genetic networks. Dramatic sleep disturbance can occur in several brain diseases—Alzheimer’s, Parkinson’s, and Huntington’s. Aging alone causes some changes in sleep with less stage three and four deep sleep and more fragmented sleep architecture.
Ben Franklin noted that sleep routines are important for health—“early to bed, early to rise, makes a man healthy…” How important this type of regularity is to brain health is just being researched with the new circadian cellular genetic networks. Dramatic sleep disturbance can occur in several brain diseases—Alzheimer’s, Parkinson’s, and Huntington’s. Aging alone causes some changes in sleep with less stage three and four deep sleep and more fragmented sleep architecture.
The Central Clock in the Hypothalamus
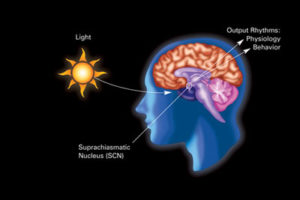
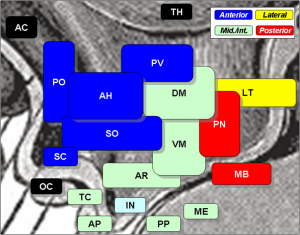 The central clock in the brain is in the hypothalamus and is called the suprachiasmatic nucleus or SCN. The hypothalamus has many centers that direct physiological parameters. For a long time the SCN was considered the only clock that works through neurons responding to sleep/wake and feeding cycles of the animal. SCN signals oscillate and affect other brain cells and hormone production. It has large unusual synapses that produce the clock in the pineal brain region to make melatonin related to sleep. It also connects with para ventricular nucleus (PVN) and the pituitary gland. This cycle stimulates ACTH adrenocorticotropic hormone in the pituitary that triggers cortisone steroids in the adrenal related to day and night. Steroids help us wake up and trigger muscles to get energy. This is all connected to the melatonin cycle.
The central clock in the brain is in the hypothalamus and is called the suprachiasmatic nucleus or SCN. The hypothalamus has many centers that direct physiological parameters. For a long time the SCN was considered the only clock that works through neurons responding to sleep/wake and feeding cycles of the animal. SCN signals oscillate and affect other brain cells and hormone production. It has large unusual synapses that produce the clock in the pineal brain region to make melatonin related to sleep. It also connects with para ventricular nucleus (PVN) and the pituitary gland. This cycle stimulates ACTH adrenocorticotropic hormone in the pituitary that triggers cortisone steroids in the adrenal related to day and night. Steroids help us wake up and trigger muscles to get energy. This is all connected to the melatonin cycle.
The SCN is highly connected to the hypothalamic brain centers for hunger, sleep, temperature, and blood concentration. This also connects with another clock related to eating. These loops and pathways are extremely complicated and not yet well understood.
The SCN has conversations with the gut and other tissues that all have their own clocks as well. These clocks are coordinated related to the time to take in more sugar and the release of insulin and other hormones (glucagon, etc.) Therefore, the SCN’s cycles and genetic loops direct melatonin, steroids, growth hormone, insulin, glucagon, and others.
Brain Signaling with Neuronal Clocks and Molecular Clocks
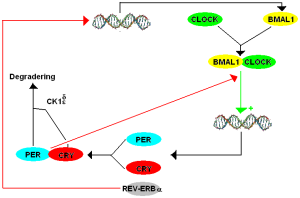
Clocks exist both in molecular form and in brain signaling circuits. Particular protein and gene sets create feedback loops that form a 24 clock of activity. The oscillations in gene production are produced through a series of protein transcription factors such as BMAL1 and CLOCK that bind to particular sections of DNA called motifs throughout the genes. Because they bind to many places, they can affect many different genes in different ways. The negative feedback involves other protein transcription factors PERIOD (PER) and CRYPTOCHROME (CRY) and REVERB. This clock forms 24 hour oscillations of many different types. The positive part of the loop involves thousands of genes producing proteins (transcripts) in particular types of cells. Signals from the central clock in the SCN of the hypothalamus synchronize many of these cycles.
SCN responds to light in the retina and synchronizes the core clock genes in a particular set of neurons. These become oscillating signals that are sent many of other cell nuclei throughout the brain. If the SCN is destroyed, then many of the other tissues lose their rhythms in their gene networks. SCN also responds to the levels of darkness and sends these rhythmic signals as well.
When the SCN is destroyed in mouse experiments, there is disruption of sleep but some of the peripheral clocks continue on their own. These mice sleep unrelated to the time of day. If genes that produce BMAL1 are eliminated, then the SCN stops functioning properly. The very complex relationships among clock genes and sleep are not completely worked out. Mutations in some of the clock genes alter sleep in a variety of ways. Also, in experiments with sleep deprivation, the core clock gene networks are altered. So, sleep and the clock genes influence each other
Clocks in Brain Disease
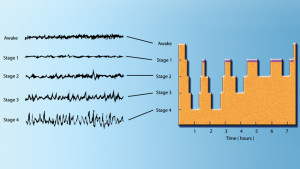 In brain diseases, sleep disturbance can occur before any obvious brain pathology, sometimes many years before. One notable sleep disturbance occurs with behavior during REM sleep (Rapid Eye Movement sleep is often associated with dreaming). During normal REM sleep the body is paralyzed so that the many wild mental experiences of dreams do not lead to physical activity and danger. When behavior occurs during REM, it is called REM behavior disorder (RBD). Almost all of those who develop RBD eventually develop Parkinson’s disease, which is a disease of the abnormal deposition of the protein synuclein. The small percentage of people with RBD who do not develop Parkinson’s also have a rare disease of abnormal versions of the protein synuclein. Animal models of a number of these degenerative brain diseases also have sleep disturbances.
In brain diseases, sleep disturbance can occur before any obvious brain pathology, sometimes many years before. One notable sleep disturbance occurs with behavior during REM sleep (Rapid Eye Movement sleep is often associated with dreaming). During normal REM sleep the body is paralyzed so that the many wild mental experiences of dreams do not lead to physical activity and danger. When behavior occurs during REM, it is called REM behavior disorder (RBD). Almost all of those who develop RBD eventually develop Parkinson’s disease, which is a disease of the abnormal deposition of the protein synuclein. The small percentage of people with RBD who do not develop Parkinson’s also have a rare disease of abnormal versions of the protein synuclein. Animal models of a number of these degenerative brain diseases also have sleep disturbances.
With sleep deprivation experiments, there is an increase in molecules related to inflammation and to damaged neurons in the cerebrospinal fluids of both humans and mice. In mice, stress was also noted in the endoplasmic reticulum that is vital to manufacturing and folding proteins for use; this was noted by a response called the unfolded protein response.
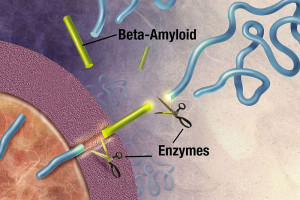 Abnormal amyloid protein fragments form plaques in the Alzheimer brain many years before the clinical symptoms of memory loss occur (b-amyloid – Ab). Neurons release Ab in particular clock rhythms into the space between neurons with fluctuating levels that rise and fall over a 24-hour cycle. What is unusual is that these cycles continue even with total darkness based on cycles inside of neurons related to metabolic activity. This metabolic activity is tied, however, to sleep and waking. These same cycles occur in the cerebrospinal fluid amyloid.
Abnormal amyloid protein fragments form plaques in the Alzheimer brain many years before the clinical symptoms of memory loss occur (b-amyloid – Ab). Neurons release Ab in particular clock rhythms into the space between neurons with fluctuating levels that rise and fall over a 24-hour cycle. What is unusual is that these cycles continue even with total darkness based on cycles inside of neurons related to metabolic activity. This metabolic activity is tied, however, to sleep and waking. These same cycles occur in the cerebrospinal fluid amyloid.
In mouse experiments that attempt to replicate the situation of Alzheimer’s (but not yet in humans) , depriving the animal of sleep greatly increases the amyloid plaque. The opposite occurs with less amyloid when sleep is increased by using drugs that antagonize orexin. Orexin is one of the new peptide signals from the hypothalamus that increases alertness and wakefulness. Blocking orexin causes sleep. Eliminating orexin also increases appetite. While the amount of sleep increase is not dramatic, the decrease in the production of amyloid is.
Poor Sleep and Alzheimer’s: Which Comes First
Taken as a whole, poor sleep is now being considered as a possible increased risk for Alzheimer’s. Since the exact relationship of amyloid to Alzheimer’s is not yet clear or certain, more needs to be done to confirm this. (See posts on Alzheimer’s and on Tau for a discussion of the relationship of amyloid and abnormal tau). There does seem to be evidence that people who are developing amyloid plaque in their brain (before Alzheimer’s memory loss) do develop poor sleep.
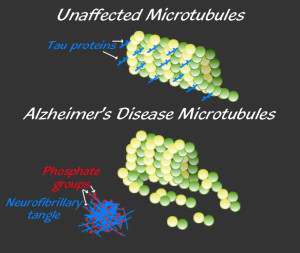 Another fact that is perhaps more ominous is that the existence of tau is closely related to levels of orexin. Tau is perhaps even more tied to the cause of Alzheimer’s, but amyloid plaque and abnormal tau (neurofibrillary tangles) are related in complex ways and probably are both somehow related to the cause of Alzheimer’s. High levels of one form of orexin (orexin-A) related to waking are correlated with abnormal tau (phosphorylated tau) and not with amyloid in the cerebrospinal fluid. Sleep deprivation produced more abnormal tau and damaged synapses.
Another fact that is perhaps more ominous is that the existence of tau is closely related to levels of orexin. Tau is perhaps even more tied to the cause of Alzheimer’s, but amyloid plaque and abnormal tau (neurofibrillary tangles) are related in complex ways and probably are both somehow related to the cause of Alzheimer’s. High levels of one form of orexin (orexin-A) related to waking are correlated with abnormal tau (phosphorylated tau) and not with amyloid in the cerebrospinal fluid. Sleep deprivation produced more abnormal tau and damaged synapses.
As background, it is well known that sleep is highly related to laying down of memories, and to forming and pruning synapses in the brain memory centers. Very poor disrupted deep sleep (Non REM) is highly associated with poor learning and memory and more plaque.
Another vital function of sleep previously described in posts is that of cleaning out proteins from the extra cellular fluid of the brain. In fact, the “glymphatic” cleaning network (from glia and lymphatic) specially removes unwanted accumulation of protein material between neurons each night during sleep. The remarkable new finding last year was that during deep sleep, neurons shrink considerably to increase the volume of the extra cellular space and this cleaning process.
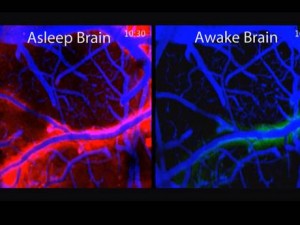 The glymphatic cleaning process is greatly encouraged by astrocytes. Studies show that slow wave sleep increased the fluid between cells in the brain by 60%. This cleaning includes abnormal tau and amyloid species. Sympathetic neurons during waking use nor epinephrine signals, which lowers the glymphatic cleaning. The opposite, that is, blocking norepinephrine receptors in the brain, increases cleaning. Another complicating fact in understanding this process is that sleep deprivation in mice damages the center in the brain that producing nor epinephrine (locus coeruleus).
The glymphatic cleaning process is greatly encouraged by astrocytes. Studies show that slow wave sleep increased the fluid between cells in the brain by 60%. This cleaning includes abnormal tau and amyloid species. Sympathetic neurons during waking use nor epinephrine signals, which lowers the glymphatic cleaning. The opposite, that is, blocking norepinephrine receptors in the brain, increases cleaning. Another complicating fact in understanding this process is that sleep deprivation in mice damages the center in the brain that producing nor epinephrine (locus coeruleus).
Huntington’s disease also has severe sleep problems before other brain changes. Research shows that sleep medications can help this process by regulating the circadian rhythms and helps cognitive ability.
Aging and Clocks
 Circadian rhythms can become less exact with aging. Some of this is because of the SCN signals and also changes in the genetic network of individual cells. Alzheimer’s can particular destroy some of the SCN neurons as well as some other cells in other brain regions. Amyloid can impair BMAL1 in some cells. In Parkinson’s, cells in the blood have altered clocks. Huntington patients have altered SCN neurons as well. Some research shows intact genetic networks in the SCN but damaged electrical properties, which some think is the primary problem.
Circadian rhythms can become less exact with aging. Some of this is because of the SCN signals and also changes in the genetic network of individual cells. Alzheimer’s can particular destroy some of the SCN neurons as well as some other cells in other brain regions. Amyloid can impair BMAL1 in some cells. In Parkinson’s, cells in the blood have altered clocks. Huntington patients have altered SCN neurons as well. Some research shows intact genetic networks in the SCN but damaged electrical properties, which some think is the primary problem.
Circadian rhythms from the SCN have strong effects on the hippocampus and memory and learning. By changing normal sleep and eating cycles in mice alters the synchronization of the SCN and the hippocampus. This causes changes in learning and memory. Because melatonin is altered in this situation, it has been used in research into Alzheimer’s (still question of its effect). Disruption of the vital synchronization with immune function, hormones, and metabolism of sugar and fats have widespread effects.
Jetlag research shifts lights earlier by several hours each day. Mice given “jetlag” from eastward travel (or simulations) cause increased inflammation, decreased new neurons in the hippocampus, and decreased learning. Other changes such as 10 hours of light and 10 hours of dark cause metabolic problems, less dendrites in the cortex and problem behaviors. Flight attendants also had changes in the hippocampus with atrophy.
Alterations in Individual Brain Cell Clocks
 Neurons, astrocytes, microglia, and oligodendrocytes all have individual clocks and can be observed in cell cultures where several hundreds of genes are part of these networks. When BMAL1 is removed from brain cells, dramatic changes occur including large increase in the amount of astrocytes, damage to synapses and connections throughout the brain, decreased new neurons in hippocampus and poor learning, and more seizures.
Neurons, astrocytes, microglia, and oligodendrocytes all have individual clocks and can be observed in cell cultures where several hundreds of genes are part of these networks. When BMAL1 is removed from brain cells, dramatic changes occur including large increase in the amount of astrocytes, damage to synapses and connections throughout the brain, decreased new neurons in hippocampus and poor learning, and more seizures.
It is not clear if these are direct brain effects or related to changes in metabolism since it is all so intertwined. In mice without BMAL1, everywhere except the SCN, there is intact sleep and rhythm synchronization in many tissues. But, there is still a big increase in the number of astrocytes and decreased neurons. The SCN appears to be very important to keep the organism healthy, but how cycles relate to brain disease is not clear and is possibly related to molecular changes in cellular clocks.
Other Cycles Related to Brain Deterioration
 Oxygen free radicals damage neurons and are involved in all brain deterioration. Oxygen cycles are highly related to clock rhythms. Peroxiredoxin 6 oxidizes molecules in most cells in nature from microbes to mammals. The results of this metabolic pathways are called ROS or reactive oxygen species that appear to have 24-hour cycles. These rhythms are in the SCN neurons and appear to be related to neuronal signals and functions.
Oxygen free radicals damage neurons and are involved in all brain deterioration. Oxygen cycles are highly related to clock rhythms. Peroxiredoxin 6 oxidizes molecules in most cells in nature from microbes to mammals. The results of this metabolic pathways are called ROS or reactive oxygen species that appear to have 24-hour cycles. These rhythms are in the SCN neurons and appear to be related to neuronal signals and functions.
Oxidative stress in animals alters the rhythms and causes fragmented sleep as in aging. Elimination of BMAL1 increases oxidative stress. ROS products are coordinated with the genes for this redox process. With greater neuronal activity, more ROS occurs. Disruption of the cycles can cause more ROS and more neuronal damage.
A previous post noted how neurons trigger versions of inflammation symptoms as a type of neuroplasticity. This often works through astrocytes and microglia. Astrocytes have been found to have a very strong clock function. In fact, eliminating BMALI causes extreme activation and production of a large number of astrocytes. Microglia also have less well defined clock functions, but clearly regulating inflammation response. Outside of the brain, macrophages and monocytes responds to time of clock cycles. BMAL1 stimulates REV-ERB causing pro inflammation cytokine production.
Degenerative brain diseases seem to be defined by accumulation of altered or abnormal proteins. The process of degrading these proteins appears to be tied to a clock cycle. These cycles can be disrupted. Autophagy is one major process for removing abnormal proteins. Another is the glymphatic flow that have already been described as highly related to a clock.
Synchronizing with Brain Clocks
 Cellular communication is the basis of all of life’s activity. Signaling related to individual cellular clocks is a new vast area of new research related to the conversations among cells. Each cell has its own clock and also feedback loops that are unique to each type of cell and each organ. These various rhythms impact each other producing cycles of metabolism and immune function.
Cellular communication is the basis of all of life’s activity. Signaling related to individual cellular clocks is a new vast area of new research related to the conversations among cells. Each cell has its own clock and also feedback loops that are unique to each type of cell and each organ. These various rhythms impact each other producing cycles of metabolism and immune function.
The central brain clocks send messages that synchronize some of this activity. Future research about the feedback loops related to clocks at all levels will be vital in determining the effects of food and sleep on disease.