 It was once thought that to prove the cause of an infection, a microbe is found. The microbe is isolated from the unhealthy person. Later, this infection is reproduced in an otherwise healthy animal by injecting that particular microbe. This approach is described in Koch’s Postulates and was the dogma of microbiology for over a hundred years. However, current research about back and forth elaborate intelligent communication between cells, including microbes, has shown that determining the cause of infections is much more complex (see post on the language of cells).
It was once thought that to prove the cause of an infection, a microbe is found. The microbe is isolated from the unhealthy person. Later, this infection is reproduced in an otherwise healthy animal by injecting that particular microbe. This approach is described in Koch’s Postulates and was the dogma of microbiology for over a hundred years. However, current research about back and forth elaborate intelligent communication between cells, including microbes, has shown that determining the cause of infections is much more complex (see post on the language of cells).
Wherever we look in current physiology, it is the elaborate communication between cells that determines outcomes. This signaling is particularly crucial in causing human infections, where constant chatter between microbes, epithelial lining cells, and immune cells determines health and disease. Recent posts have demonstrated elaborate intelligent signaling in the intestine epithelial cell (The Intelligent Intestinal Epitheilial Cell), the skin cell (The intelligent Skin Cell), the choroid plexus cell (the very intelligent choroid plexus cell) and many immune system cells (see regulation of pattern recognition receptors).
 Very recent research shows that even with dangerous bacteria causing serious illness, it is not just that single microbe species creating the infection. Much of the time, it is a collaboration of multiple microbes, and even human cells, that alters the environment to create the illness. Surprisingly, viruses play a large role in these interactions. It is the politics of gut microbe Kingdoms that determines the strength of the infection.
Very recent research shows that even with dangerous bacteria causing serious illness, it is not just that single microbe species creating the infection. Much of the time, it is a collaboration of multiple microbes, and even human cells, that alters the environment to create the illness. Surprisingly, viruses play a large role in these interactions. It is the politics of gut microbe Kingdoms that determines the strength of the infection.
This post will discuss what has been recently discovered about the complex communication between viruses, other microbes and host cells in determining the severity of illness. Future posts will discuss the complex geography and interactions of microbes in the gut and in biofilms.
Microbe Kingdoms in the Human Gut
Viruses, bacteria, fungi, archaea, and eukaryotes (such as protozoa and parasitic worms) all call the human gut their home. Each of these kingdoms interacts wildly with all the others. Viruses while the smallest and with the least genetic material, have dramatic regulatory influences on the other kingdoms. Until recently, it was thought that the virus’ enormous influence occurred by transfer of particular genes, making bacteria more powerful, and by particular immune reactions to these particles (horizontal gene transfer is ubiquitous in bacteria and often determines resistance to antibiotics).
Recently, many new viruses have been found in the gut exhibiting very complex relationships with human cells and with other microbes. This relationship goes both ways. The viruses are critical for the results of the bacterial activity, and the bacteria are critical for the viruses survival. Microbes are critical for the success of four gut virus families—norovirus, picornavirus, retrovirus and reovirus. Other viruses in the gut include rotavirus, herpesviruses and adenoviruses. It is not possible to understand the danger of these viruses without the full view of the supporting microbe communities and their specific interactions. Most information has been related to the bacteria in the gut, and now their complex relations to our immune cells and to viruses are emerging.
 These families of viruses have close relations with the phage viruses associated only with bacteria (phage are part of the constant transfer of information among microbes). They, also, have close relations with fungi and helminthes (parasitic worms).
These families of viruses have close relations with the phage viruses associated only with bacteria (phage are part of the constant transfer of information among microbes). They, also, have close relations with fungi and helminthes (parasitic worms).
Gut viruses travel in water, food and by contact of animals. When they enter the gut, a very complex environment bombards them in different ways as they travel further into the gut. Different regions of the gut have different levels of acid and base, many different kinds of enzymes, antimicrobials and toxins that attack material including viruses and a vast array of different temporary and permanent microbes. Unique microbes live in the mucous that lines the epithelial cells. These have been chosen by the intelligent intestinal epithelial cell (see post) as friendly and helpful to the organism.
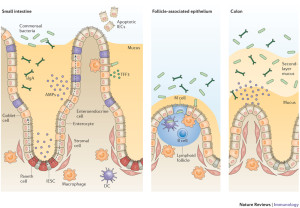 If viruses can penetrate the mucous barrier, they can then possibly enter Microfold or M immune cells that are part of the epithelium. M cells are like immune dendritic cells in that they sit in the intestinal epithelium, take up molecules (antigens) from the microbes in the gut and pass them along to immune cells below to determine what the response will be. This couild allow them to infect the animal. Surprisingly, the virus is able to deal with all of these factors and multiply in the gut, perhaps the most complex ecology they can face.
If viruses can penetrate the mucous barrier, they can then possibly enter Microfold or M immune cells that are part of the epithelium. M cells are like immune dendritic cells in that they sit in the intestinal epithelium, take up molecules (antigens) from the microbes in the gut and pass them along to immune cells below to determine what the response will be. This couild allow them to infect the animal. Surprisingly, the virus is able to deal with all of these factors and multiply in the gut, perhaps the most complex ecology they can face.
Gut Virome
 All of the already mentioned families of viruses in the gut are, also, influenced by other families that have already infected the host or exist in other parts of the body’s surface such as the mucosa of the lungs. Interactions between all of these families influence immunity and healing related both to bacteria and viruses. Some viruses improve immunity to fight other microbes. Cataloguing these interactions has been beyond current science, which focuses on DNA viruses, while many gut viruses use only RNA.
All of the already mentioned families of viruses in the gut are, also, influenced by other families that have already infected the host or exist in other parts of the body’s surface such as the mucosa of the lungs. Interactions between all of these families influence immunity and healing related both to bacteria and viruses. Some viruses improve immunity to fight other microbes. Cataloguing these interactions has been beyond current science, which focuses on DNA viruses, while many gut viruses use only RNA.
There are approximately 100 trillion microbe cells in the gut, many with 10x as many friendly phage viruses (temperate phage are useful for bacteria – see post) both in their genomes and surrounding them. The eukaryote cells, also, have their viruses. In fact, each animal has at least 10 ongoing virus infections at any given time. Human infections consist, at least, with the gut viruses Adenoviridae, Astroviridae, Caliciviridae, Reoviridae, and Picornaviridae, Picobirnaviridae, Annelloviridae, and Circoviridae families. The norovirus and the reovirus are extremely common. Others, like polio are rare. These create many different levels of infections from those with no symptoms to acute illness and severe chronic illness. Some of these infections are life long and can be transmitted to others. Healthy children have many of these, as well. The regulation of this large number of families of viruses is vastly complex.
 Surprisingly, with so many trillions of microbes and viruses, even 100 noro, rota and polio viruses can stimulate an infection. This small number must travel through high acid, dangerous enzymes, dense mucous and many enemy bacteria before they can multiply inside of a human cell. The first infection of each type is much more difficult for the virus, whereas once established it is much easier. Each virus attacks a different type of human cell including immune cells like lymphocytes and myeloids and epithelial intestinal cells. These new baby viruses are then sent into the gut lumen to continue the process elsewhere.
Surprisingly, with so many trillions of microbes and viruses, even 100 noro, rota and polio viruses can stimulate an infection. This small number must travel through high acid, dangerous enzymes, dense mucous and many enemy bacteria before they can multiply inside of a human cell. The first infection of each type is much more difficult for the virus, whereas once established it is much easier. Each virus attacks a different type of human cell including immune cells like lymphocytes and myeloids and epithelial intestinal cells. These new baby viruses are then sent into the gut lumen to continue the process elsewhere.
Bacteria Help Viruses
Because of the great complexity, research is incomplete in the ways bacteria help and hurt the viruses. Also, human viruses don’t attack many other animals that are used in research (mice, for example, don’t vomit or get diarrhea). There is substantial evidence, however, that bacteria do influence viral infections. One virus transmitted in mother’s milk needs bacteria for this transmission. Polio is increased by gut bacteria during transmission from feces to mouth and these bacteria increase its strength. Gut microbes also, increase the number and strength of Reo and rotaviruses. The gut bacteria also increase norovirus. In research, if these viruses are injected directly their numbers don’t increase. They only increase if they pass through the mouth with the benefit of bacteria.
One mechanism of this cooperation includes viruses that attach to the polysaccharides on the surface of bacteria, increasing the virus’ ability to invade human cells. MMTV virus with two lipid layers in its envelope attaches to lipopolysaccharide (LPS) glycan on Gram-negative bacteria, stimulating immune reactions creating immunity to the virus. This process greatly increases the ability to make more viruses and travel further.
Polio has no envelop. It attaches to peptidoglycan and LPS on the microbe surface. This allows it to survive the heat and acid and allows it to slip onto the human cells from the bacteria. Norovirus, also without an envelope, attaches to sugar antigens that are on the surface of human cells and some bacteria. This allows it to infect lymphocytes.
Immune Help For Bacteria and Viruses
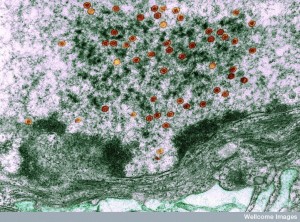 Immune responses to bacteria can have positive effects on viruses. One example includes the development of antibodies to gut bacteria, which make vaccines less effective. A particular interaction uses the pattern receptors TLR5 (see post). Molecules from gut bacteria stimulate a pattern receptor, which then affects the vaccine response to the influenza virus.
Immune responses to bacteria can have positive effects on viruses. One example includes the development of antibodies to gut bacteria, which make vaccines less effective. A particular interaction uses the pattern receptors TLR5 (see post). Molecules from gut bacteria stimulate a pattern receptor, which then affects the vaccine response to the influenza virus.
When antibiotics limit the immune response, they can help the virus. Parasitic worm infections can stop CD8 T cells from attacking viruses. These worms decrease the ability to limit herpes. The worm stimulates interleukin-4 or interleukin-13, which has the effect of producing particular transcription factors promoting the copying of the virus. Thus, it can awaken a dormant herpes virus. It, also, changes the types of macrophages that are produced to chase the virus.
On the other hand, viruses stimulate interferon-1 changing the macrophages. This new type of macrophage stimulates more epithelial cells and helps the healing of the breach in the gut from the infection.
 Bacteria affect the type of signals sent by immune cells to combat viruses. An LPS from a virus stimulates TLR4 with IL-10, which creates tolerance to that particular virus transmission. Another mechanism uses IFN-λ receptors on immune cells, which stops virus infection. This shows that it is not just viruses and bacteria attaching to help each, but rather they affect the immune signaling involved by complex means. In fact, research shows that when immune cells are eliminated, there is better control of the viruses from just a cytokine signal. This shows that lymphocyte signaling is actively helping the virus. There are multiple examples of this help by cytokines with norovirus. Another with rotavirus uses TLR signaling from bacterial molecules, which stimulates cytokines IL-22 and Il-18 to stop virus infection. In all of these cases, it was not general adaptive immune responding, but interruption of a particular cytokine signal.
Bacteria affect the type of signals sent by immune cells to combat viruses. An LPS from a virus stimulates TLR4 with IL-10, which creates tolerance to that particular virus transmission. Another mechanism uses IFN-λ receptors on immune cells, which stops virus infection. This shows that it is not just viruses and bacteria attaching to help each, but rather they affect the immune signaling involved by complex means. In fact, research shows that when immune cells are eliminated, there is better control of the viruses from just a cytokine signal. This shows that lymphocyte signaling is actively helping the virus. There are multiple examples of this help by cytokines with norovirus. Another with rotavirus uses TLR signaling from bacterial molecules, which stimulates cytokines IL-22 and Il-18 to stop virus infection. In all of these cases, it was not general adaptive immune responding, but interruption of a particular cytokine signal.
Complex Genetic interactions
 The cooperation between microbe Kingdoms and the completion of the microbe behavior plays out in the genetic regulation of human cells. In one example, a virus needs a special mutation to create disease. Neither an alteration in the intestinal Paneth cell or a special virus are enough to create the infection. It needs both. The Paneth cell sits in the gut epithelium and produces substances to fight dangerous microbes and has interactions with the immune cells below.
The cooperation between microbe Kingdoms and the completion of the microbe behavior plays out in the genetic regulation of human cells. In one example, a virus needs a special mutation to create disease. Neither an alteration in the intestinal Paneth cell or a special virus are enough to create the infection. It needs both. The Paneth cell sits in the gut epithelium and produces substances to fight dangerous microbes and has interactions with the immune cells below.
Another example is a virus in combination with a mutation of a cytokine IL-10 creating disease, but not either alone. Also, in both of these examples the disease that is created in the intestine requires bacteria as well. It is the interaction between the virus and the bacteria that allow the creation of the intestinal disease.
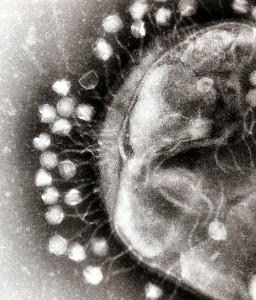 Another way this happens is by virus moving genes between several bacteria. Phages do this all the time. In infections of the colon (colitis), there are fewer families of diverse bacteria, but the ones present are altered by viruses. It is the complex interactions of several bacteria along with viruses that create the environment to support the infection.
Another way this happens is by virus moving genes between several bacteria. Phages do this all the time. In infections of the colon (colitis), there are fewer families of diverse bacteria, but the ones present are altered by viruses. It is the complex interactions of several bacteria along with viruses that create the environment to support the infection.
We will not really understand these complex processes until much greater research is done on the vast amount of RNA viruses in the gut. Also, there is still little research on the interactions with fungi, archaea and many tiny animals (meiofauna). Also, just as immune-nervous system synapses are extremely complex involving as many as five different cells at the same time (see post), these gut virus-bacteria-host cell interactions occur between many different Kingdoms in very complex ways. Other environmental factors, such as diet, are clearly important, but are just being researched.
One counterintuitive conclusion is that antibiotics can both help and hurt gut viruses. Treatment in the future might need to use very particular cytokines—for example flagellin and IFN0λ. The different binding sites on bacteria affect the viruses.
Koch’s Postulates
The early dogma in microbiology was called Koch’s Postulates and it was thought to be valid until recently. It states that to determine the cause of an infection, a single microbe must be found in the ill person, that would not be there in someone healthy. Also, one microbe causes a particular disease. Then, the single microbe is injected into a healthy animal and it causes the disease.
Recently, it has been found that the causes of infections are not so simple and are more often from the complex interactions of many different microbes, both positive and negative. There are, in fact, many situations where multiple microbes are essential for an infection to occur, even though one appears to be prominent and takes the credit.
 In many hospital-acquired infections, there are pre existing permanent colonies of microbes that fight against dangerous microbe. A particular microbe will start the infection in some people but not others. This resistance can occur through many complex mechanisms. In one example, a particular microbe changes the bile acids in the gut and this directly stops Clostridium difficile infection.
In many hospital-acquired infections, there are pre existing permanent colonies of microbes that fight against dangerous microbe. A particular microbe will start the infection in some people but not others. This resistance can occur through many complex mechanisms. In one example, a particular microbe changes the bile acids in the gut and this directly stops Clostridium difficile infection.
Another mechanism is when a particular version of E. Coli alters immune function to help with muscle damage from the gut trauma from infection of another microbe. This strain of E. coli was actually stopping colitis muscle wasting caused by another bacteria. E. coli stopped damage caused by Salmonella by influencing the immune system, which reverses muscle atrophy and stimulates new muscle cells.
 Even more complex interactions are being found. In the example with c. difficile above, one microbe species inhibits the action of another. In fact, six different microbe communities together have an even more positive effect. This type of situation is extremely hard to piece together. One additional microbe species slows it down, but more are better. Up to six other microbes together will stop it. In order for this information to become available, it will be necessary to have much better whole genetic sequencing of all of the many microbes involved.
Even more complex interactions are being found. In the example with c. difficile above, one microbe species inhibits the action of another. In fact, six different microbe communities together have an even more positive effect. This type of situation is extremely hard to piece together. One additional microbe species slows it down, but more are better. Up to six other microbes together will stop it. In order for this information to become available, it will be necessary to have much better whole genetic sequencing of all of the many microbes involved.
The Politics of Gut Microbe Kingdoms
 Intelligent interactions and signaling between wide varieties of cell species determine outcomes of dangerous microbes in the human gut. Surprisingly, viruses are a critical partner in these interactions between bacteria, archaea, worms, fungi, tiny animals and host cells.
Intelligent interactions and signaling between wide varieties of cell species determine outcomes of dangerous microbes in the human gut. Surprisingly, viruses are a critical partner in these interactions between bacteria, archaea, worms, fungi, tiny animals and host cells.
Back and forth signaling occurs constantly between the human barrier cells (epithelium), the many different immune cells and the wide range of different microbe species. Somehow, this intelligent communication can occur between completely different Kingdoms of species and cells. Humans depend on this intelligent communication to avoid disease. Microbes depend upon it to help humans and to create disease.
Future posts will discuss how this communication determines the geography of the stable friendly microbe communities that live throughout the different regions of the small and large intestines (and other human bodily surfaces). Microbe geography is determined by many environmental factors and by intelligent communication of cells.
Where is the mental connection among free ranging cells that allows completely different Kingdoms of species to have intelligent communication?